ESGO/ESTRO/ESP Endometrial Risk (2021) Calculator: Streamline Risk Stratification at the Point of Care
The ESGO/ESTRO/ESP 2021 guidelines introduced an updated risk classification system that integrates both clinicopathologic and molecular parameters. This guide explores the practical application of these criteria using digital decision support.
1. Introduction to the ESGO/ESTRO/ESP 2021 Framework
Endometrial cancer is the most common gynecologic malignancy in high-income countries, and its incidence continues to rise worldwide. After surgical staging, the critical next question is deceptively complex: which patients need adjuvant therapy, and how much? The answer hinges on accurate risk stratification — a task that requires clinicians to synthesize FIGO stage, histological type, tumor grade, lymphovascular space invasion (LVSI) status, and, when available, molecular classification data into a single prognostic risk group.[1], [2], [3]
The ESGO/ESTRO/ESP 2021 guidelines introduced an updated risk classification system that integrates both clinicopathologic and molecular parameters, representing a major advancement in personalized adjuvant treatment planning for endometrial carcinoma. At OncoToolkit, we have built a free, mobile-responsive ESGO/ESTRO/ESP Endometrial Risk (2021) calculator that translates this complex framework into instant, transparent clinical decision support — designed for use during MDT meetings, point-of-care rounds, and trainee education.[4], [5], [6]
2. Defining the ESGO/ESTRO/ESP 2021 Endometrial Risk Classification
The ESGO/ESTRO/ESP 2021 risk classification is a consensus-based prognostic framework published jointly by the European Society of Gynaecological Oncology, the European Society for Radiotherapy and Oncology, and the European Society of Pathology. It stratifies patients with endometrial carcinoma into five prognostic risk groups — Low, Intermediate, High-Intermediate, High, and Advanced/Metastatic — each linked to specific adjuvant treatment recommendations.[1], [2], [6]
The system operates in two parallel tracks: one for when molecular classification is known (incorporating POLE mutation status, mismatch repair deficiency, and p53 status), and one for when molecular classification is unknown or unavailable, relying solely on traditional clinicopathologic variables. Our calculator on OncoToolkit focuses on the molecular-unknown pathway, which remains the everyday reality for many institutions globally where comprehensive molecular testing is not yet routinely available.[7], [2], [6]
2.1. Core Variables for Risk Assessment
The four clinicopathologic inputs that drive the classification when molecular status is unknown are:
- FIGO Stage (IA, IB, II, III, IVA, IVB) — reflecting depth of myometrial invasion, cervical involvement, and distant spread.
- Histological Type — Endometrioid versus non-endometrioid (serous, clear cell, undifferentiated carcinoma, carcinosarcoma, mixed).
- Tumor Grade (for endometrioid tumors) — Grades 1 and 2 are classified as low-grade; Grade 3 as high-grade, per binary FIGO grading.[1]
- Lymphovascular Space Invasion (LVSI) — Graded as negative/focal versus substantial. Substantial LVSI is defined as multifocal or diffuse LVSI, or the presence of tumor cells in five or more lymphovascular spaces.[1]
2.2. Risk Group Definitions: Molecular Classification Unknown Pathway
| Risk Group | Criteria (Molecular Unknown) | |---|---| | Low | Stage IA endometrioid, low-grade (G1/G2), LVSI negative or focal | | Intermediate | Stage IB endometrioid low-grade with LVSI negative/focal; OR Stage IA endometrioid high-grade with LVSI negative/focal; OR Stage IA non-endometrioid without myometrial invasion | | High-Intermediate | Stage I endometrioid with substantial LVSI (any grade/depth); OR Stage IB endometrioid high-grade (any LVSI); OR Stage II | | High | Stage III–IVA with no residual disease; OR Stage I–IVA non-endometrioid with myometrial invasion and no residual disease | | Advanced/Metastatic | Stage III–IVA with residual disease; OR Stage IVB |
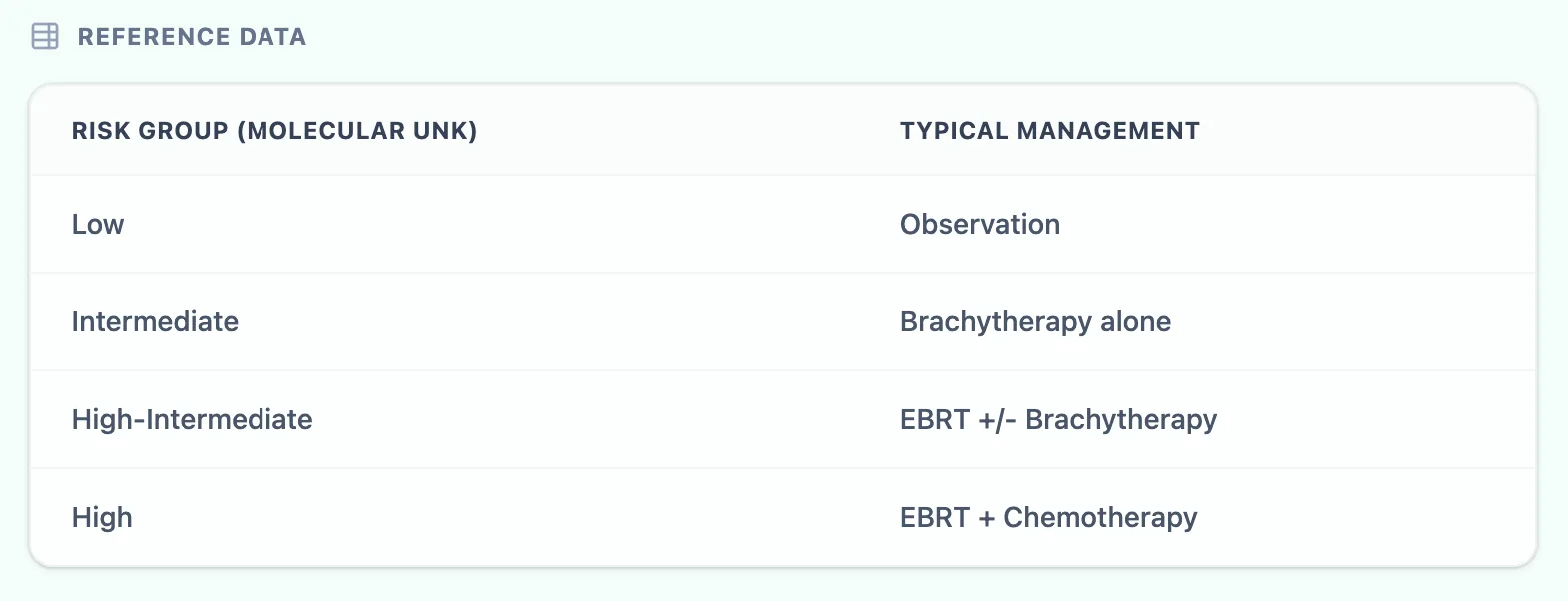
Figure 1. The reference data table within the OncoToolkit calculator maps each risk group (Molecular Unknown) to its typical adjuvant management strategy — from observation for low-risk patients to EBRT plus chemotherapy for high-risk disease. This quick-glance reference is available alongside every calculated result.
3. Clinical Significance: Linking Risk to Adjuvant Therapy Decisions
3.1. Optimizing Adjuvant Treatment Strategies
Adjuvant treatment recommendations for endometrial carcinoma are directly tied to the prognostic risk group. The 2021 guidelines provide clear, evidence-graded recommendations for each category:[1]
- Low Risk → No adjuvant treatment (Level I, Grade A).
- Intermediate Risk → Vaginal brachytherapy to reduce vaginal recurrence; observation is acceptable, especially for patients under 60 years.[8]
- High-Intermediate Risk → EBRT is recommended, particularly for substantial LVSI and/or stage II disease. Additional adjuvant chemotherapy may be considered for high-grade tumors and/or substantial LVSI.[1]
- High Risk → EBRT with concurrent and adjuvant chemotherapy, or sequential chemotherapy and radiotherapy. Chemotherapy alone is an alternative.[9], [1]
3.2. Reducing Cognitive Load and Calculator Fatigue in MDTs
In a typical MDT meeting, a gynecologic oncology team may review 10–20 new or follow-up endometrial cancer cases. For each, the team must mentally cross-reference FIGO stage, grade, histology, and LVSI against a multitiered risk table — often while discussing surgical findings, imaging, and patient comorbidities simultaneously. This cognitive burden is a well-documented source of decision-making fatigue in cancer MDTs.[10], [11]
Without digital support, common pain points include misremembering whether a particular Stage IB, Grade 3 endometrioid tumor falls into the intermediate or high-intermediate group, and uncertainty about how substantial versus focal LVSI shifts the risk assignment.
On our platform, the calculator resolves these ambiguities in seconds. It is optimized for mobile devices, meaning clinicians can run a quick classification from a phone during ward rounds or while preparing for an MDT — without needing to pull up the full guideline PDF.
4. Clinical Evidence and Validation of the 2021 Risk Groups
4.1. The Rule-Based Algorithm: Math and Logic Behind the Tool
Unlike point-based scoring systems that sum weighted coefficients, the ESGO/ESTRO/ESP 2021 classification uses a rule-based decision algorithm — a hierarchical mapping of clinicopathologic features to discrete risk categories. The logic is essentially a decision tree: given a specific combination of FIGO stage, histological type, grade, and LVSI status, the patient is assigned to exactly one risk group. There is no continuous score or regression equation; the output is categorical.[6]
This design reflects the guideline's foundation in consensus expert interpretation of trial-level evidence, rather than a single derivation cohort. The decision boundaries are anchored to adjuvant treatment thresholds established in landmark randomized trials (PORTEC-1, PORTEC-2, PORTEC-3, GOG-249, GOG-258).[9], [12], [13]
4.2. Key Validation Data from Landmark Clinical Trials
The prognostic validity of these risk groups has been confirmed in multiple independent cohorts:
- PORTEC-1 and PORTEC-2:
These landmark trials established that patients in what is now classified as the intermediate-risk group have low recurrence rates manageable with vaginal brachytherapy alone, without the morbidity of full pelvic EBRT. Ten-year results of PORTEC-2 confirmed that vaginal brachytherapy was non-inferior to EBRT for high-intermediate risk disease, with significantly better quality of life.[12]
- PORTEC-3 (10-year analysis):
For high-risk endometrial cancer, chemoradiotherapy improved 10-year overall survival and recurrence-free survival compared to radiotherapy alone. The benefit was most pronounced in stage III disease and serous carcinomas. A post-hoc molecular analysis showed that only the p53-abnormal subgroup derived clear survival benefit from added chemotherapy, underscoring the importance of integrating molecular data when available.[14], [15], [16]
- Implementation cohort (N=604, Helsinki):
When the 2021 risk groups were applied to an unselected cohort of 604 endometrial cancer patients with molecular classification, the five risk groups showed significantly distinct disease-specific survival curves. Molecular classification caused a risk-group shift in approximately 6–7% of patients — primarily upshifting p53-abnormal tumors to high risk and downshifting POLE-mutated tumors to low risk.[2]
4.3. Refining the Prognosis: When Molecular Classification is Available
The calculator on OncoToolkit explicitly notes that molecular classification (POLE, p53, MMR) significantly refines these groups. In the 2021 guidelines, POLE-mutated endometrial carcinoma of any stage (I–II) is considered low risk regardless of grade or histology, while p53-abnormal tumors with myometrial invasion are automatically high risk. MMRd and NSMP tumors generally remain in their clinicopathologic risk group, though nuances exist.[7], [17], [2], [6]
For clinicians working at centers where molecular testing is being implemented, the calculator provides the clinicopathologic baseline risk group, and the result screen prompts consideration of molecular data as a refinement layer.
4.4. Clinical Limitations and Implementation Caveats
- The molecular-unknown risk groups may over-treat some patients (e.g., POLE-mutated tumors classified as high-intermediate by clinicopathologic features alone) and under-treat others (e.g., occult p53-abnormal tumors within the low-risk category).[2]
- The classification was developed primarily from European trial populations. While endometrial cancer biology is broadly similar across populations, differences in histotype distribution and access to molecular testing should be considered when applying the framework in non-European settings.[4]
- The 2021 guidelines have been updated in 2025 to incorporate the revised FIGO 2023 staging system and expanded molecular categories. The OncoToolkit calculator reflects the 2021 framework, which remains the most widely validated and implemented version globally.[18], [19]
- LVSI grading (focal vs. substantial) can have inter-observer variability, which may affect risk assignment at the boundary between intermediate and high-intermediate groups.[1]
5. How to Use the ESGO/ESTRO/ESP Endometrial Risk Calculator
At OncoToolkit, the ESGO/ESTRO/ESP Endometrial Risk (2021) calculator is designed for speed and transparency. Here is how it works, step by step.
5.1. Step 1: Inputting Patient Clinical Data
The input form presents four dropdown menus, each mapped directly to the guideline's classification variables:[6]
- FIGO Stage — Select from IA (< 50% invasion), IB (≥ 50% invasion), II, III, IVA, or IVB.
- Histological Type — Endometrioid or Non-endometrioid (serous, clear cell, undifferentiated, carcinosarcoma, mixed).
- Tumor Grade — Appears conditionally when endometrioid histology is selected; choose Grade 1, 2, or 3.
- LVSI — Negative/Focal or Substantial.
The interface uses conditional logic: if a non-endometrioid histology is selected, the grade field is hidden because grade does not apply to those histotypes in this classification.
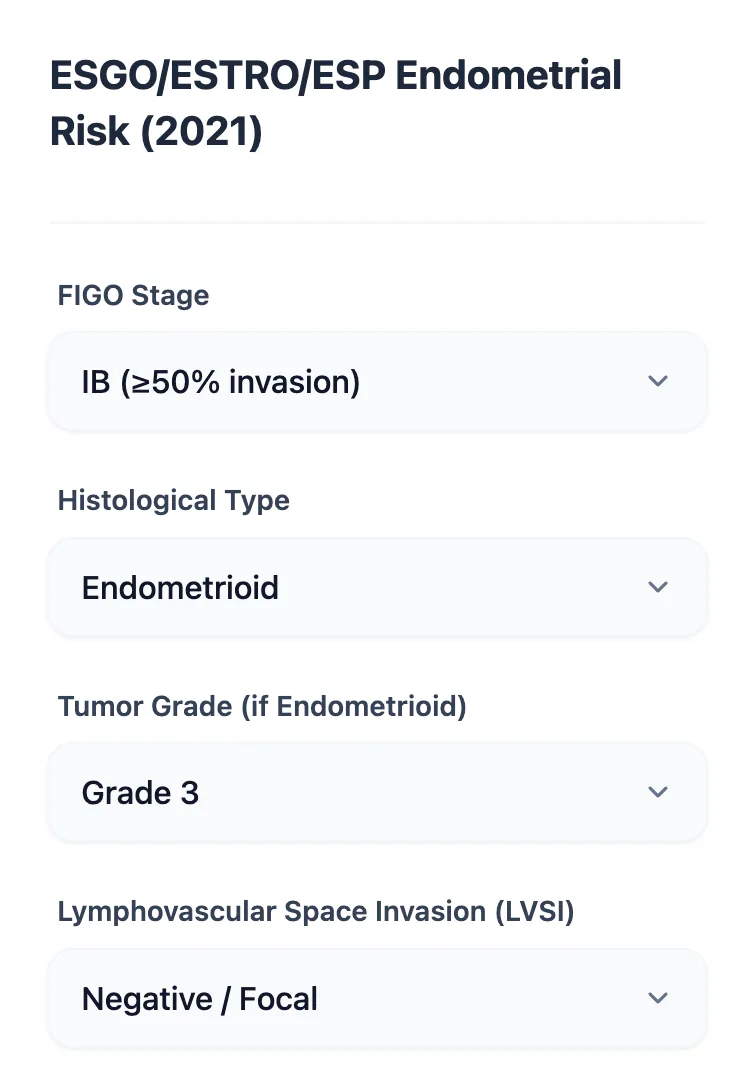
Figure 2. The calculator input form showing a sample case: FIGO Stage IB (≥50% invasion), Endometrioid histology, Grade 3, with LVSI Negative/Focal. The clean dropdown interface is designed for rapid data entry — ideal for MDT meetings or bedside use on a mobile device.
5.2. Step 2: Interpreting the Risk Stratification Results
After submitting the inputs, the calculator displays:
- The Risk Group — Prominently shown (e.g., "High-Intermediate Risk") with a color-coded risk bar spanning from Low Risk (green) through Intermediate (yellow) to High Risk (red).
- The Treatment Recommendation — A concise summary of the guideline-endorsed adjuvant approach for that risk group (e.g., "EBRT (Adjuvant Chemotherapy considered for G3)").
- A Molecular Refinement Note — A reminder that molecular classification (POLE, p53, MMR) can significantly alter the risk assignment, prompting the clinician to seek or review molecular data.

Figure 3. The result screen for the example case (Stage IB, Endometrioid, Grade 3, LVSI Negative/Focal) shows a "High-Intermediate Risk" classification. The recommendation includes EBRT with adjuvant chemotherapy considered for Grade 3 tumors. The visual risk bar and molecular classification prompt support transparent, guideline-aligned decision-making.
5.3. Step 3: Reviewing Evidence and Clinical Background
Below the result, clinicians can expand a "Clinical Context & Background" section that explains the guideline source, the primary reference citation, and the formula logic underpinning the classification. This transparency is intentional — at OncoToolkit, we believe clinicians should always be able to see why the tool arrived at a particular result, not just what it is.[6]

Figure 4. The clinical background panel cites the primary source (Concin et al., Int J Gynecol Cancer 2021) and displays the underlying formula logic. This evidence layer builds trust by making the calculator's reasoning fully transparent to the treating physician.
6. Supporting Clinical Care, Education, and Research
6.1. Enhancing Routine Decision Support and MDT Preparation
The most immediate use case is staging at diagnosis and MDT preparation. When a pathology report arrives with the four key variables — stage, histology, grade, and LVSI — the calculator provides an instant risk classification that aligns the entire team around a guideline-endorsed starting point for adjuvant treatment discussion. This is especially valuable in high-volume centers where MDT coordinators prepare dozens of cases per week.[11]
In point-of-care rounds, the mobile-responsive design allows residents or fellows to confirm risk group assignments at the bedside while counseling patients, reducing the risk of miscommunication between surgical, radiation, and medical oncology teams.
6.2. Educational Utility for Oncology Trainees
For gynecologic oncology fellows and residents, the calculator serves as an interactive teaching tool. Trainees can:
- Input various combinations of stage, grade, histology, and LVSI to build intuition about which features drive risk group assignment.
- Explore edge cases — for example, understanding why Stage IA, Grade 3, endometrioid with negative LVSI is intermediate risk, while the same tumor with substantial LVSI becomes high-intermediate.
- Use the reference table and clinical background panel to connect calculator outputs to the underlying trial evidence.
6.3. Standardization for Research and Quality Improvement
Consistent risk classification is essential for multi-site research and quality improvement (QI) initiatives. The calculator standardizes the application of ESGO/ESTRO/ESP criteria across clinicians, reducing inter-observer classification variability. Teams engaged in retrospective chart reviews or prospective registry studies can use OncoToolkit's calculators to ensure uniform staging across their cohorts.
6.4. Integrated Gynecologic Oncology Tools on OncoToolkit
Clinicians working in gynecologic oncology may also find these related tools useful:
- Mayo Criteria (Endometrial) — Identifies low-risk endometrial cancer patients in whom lymphadenectomy may be safely omitted.[21]
- Sedlis Criteria (Cervical) — Identifies intermediate risk factors for adjuvant radiation post-radical hysterectomy in cervical cancer.[20]
- FIGO GTN Risk Score — Stratifies risk for gestational trophoblastic neoplasia to guide single-agent versus multi-agent chemotherapy.[20]
7. Clinical FAQ: Addressing Common Implementation Questions
Can the ESGO/ESTRO/ESP 2021 risk classification be used when molecular testing is unavailable?
Yes — this is precisely the scenario our calculator is built for. The 2021 guidelines provide a complete parallel track of risk group definitions for when molecular classification is unknown. While the guidelines encourage molecular testing in all endometrial carcinomas, they explicitly recognize that many centers worldwide do not yet have routine access to testing.[4], [1], [6]
How does the ESGO/ESTRO/ESP 2021 classification differ from the older ESMO 2016 system?
The 2021 update introduced molecular classification as a parallel pathway, refined LVSI grading to focal versus substantial, and redefined certain risk boundaries to align with the latest trial evidence.[1], [2]
What is the most common mistake when classifying endometrial cancer risk with this system?
The most frequently observed error is misgrading LVSI — specifically, failing to distinguish substantial from focal LVSI. The distinction has direct treatment implications (brachytherapy alone vs. EBRT ± chemotherapy).[1]
Does the calculator account for the updated FIGO 2023 staging system?
The current OncoToolkit calculator reflects the 2021 ESGO/ESTRO/ESP risk groups, which are based on FIGO 2009 staging. While FIGO 2023 is emerging, the 2021-based calculator remains the most broadly validated and implemented version globally.[18], [3], [6]
8. Conclusion: Implementing Precision Risk Stratification
Accurate risk stratification is the foundation of personalized adjuvant treatment in endometrial cancer. Whether preparing for an MDT, counseling a patient after surgery, or teaching trainees the nuances of the ESGO/ESTRO/ESP classification, the OncoToolkit calculator offers a fast, transparent, and guideline-aligned tool at the point of care.
Try it with several example cases — vary the stage, grade, histology, and LVSI to build a feel for how the classification boundaries work. Bookmark the calculator for your next MDT meeting, and explore OncoToolkit's full library of gynecologic oncology decision-support tools.[20]
Disclaimer: This calculator is a clinical decision-support tool and does not replace clinical judgment. All treatment decisions should be made in the context of the individual patient's clinical scenario, institutional protocols, and multidisciplinary team discussion.
Ready to Simplify Your Risk Stratification?
Access the ESGO/ESTRO/ESP Endometrial Risk (2021) calculator for instant, evidence-based results.
Use the Risk Calculator
Free to use. No registration required.
References
- ESGO-ESTRO-ESP Guideline for the management of Endometrial Carcinoma. Source
- Clinicopathologic vs. Molecular Integrated Prognostication of Endometrial Carcinoma by European Guidelines. Source
- Endometrial Cancer: 2023 Revised FIGO Staging System. Source
- 836 Implementation of the 2021 molecular ESGO/ESTRO/ESP risk groups in endometrial cancer. Source
- The emerging role of molecular pathology in directing the systemic treatment of endometrial cancer. Source
- ESGO/ESTRO/ESP Endometrial Risk (2021) | OncoToolkit. Source
- Polemut Endometrial Cancer. Source
- Adjuvant treatment algorithm based on recent ESGO/ESTRO/ESP guidelines. Source
- Final results of the international randomized PORTEC-3 trial. Source
- Do multidisciplinary cancer care teams suffer decision-making fatigue? Source
- The Importance of the Multidisciplinary Team in Endometrial Cancer. Source
- Ten-year results of the PORTEC-2 trial. Source
- Adjuvant chemoradiotherapy versus radiotherapy alone (PORTEC-3). Source
- The PORTEC-3 trial 10-year analysis. Source
- Adjuvant Chemoradiotherapy vs Radiotherapy for High-Risk Endometrial Cancer. Source
- Molecular Classification of the PORTEC-3 Trial. Source
- ESGO-ESTRO-ESP Endometrial Cancer Guidelines - MaNGO Group. Source
- ESGO-ESTRO-ESP guidelines: update 2025. Source
- ESGO–ESTRO–ESP guidelines for management - July 2025. Source
- OncoToolkit | Oncology Calculators. Source
- Mayo Criteria (Endometrial) | OncoToolkit. Source