DIPSS (Myelofibrosis) Calculator: Dynamic Risk Stratification
Estimate myelofibrosis survival and risk in seconds with the DIPSS calculator. Optimize transplant and JAK inhibitor decisions at the point of care.

Quick Navigation
1. Introduction: The DIPSS Myelofibrosis Calculator
Primary and secondary myelofibrosis (MF) are clinically heterogeneous myeloproliferative neoplasms with survival ranging from near‑normal life expectancy to rapid progression and leukemic transformation. Hematology teams must integrate constitutional symptoms, cytopenias, leukocytosis, splenomegaly, and molecular data—often in high‑pressure environments such as transplant clinics and multidisciplinary meetings. At OncoToolkit, we’ve built a DIPSS (Myelofibrosis) calculator to convert this complexity into a fast, transparent, point‑of‑care risk estimate that can be recalculated dynamically across the disease course. 1, 2, 3, 4, 5
The Dynamic International Prognostic Scoring System (DIPSS) remains one of the most widely used prognostic tools for myelofibrosis and is a core reference in major guidelines and expert reviews worldwide. Our online calculator implements the original DIPSS model, clearly displays the scoring logic, and links out to related tools such as IPSS, DIPSS‑Plus, and transplant risk calculators so clinicians can build a comprehensive, calculator‑driven view of each patient. 3, 6, 1
2. What is the DIPSS Score? Clinical Definitions
2.1 From IPSS to DIPSS: Evolution of Scoring
The original International Prognostic Scoring System (IPSS) was developed for primary myelofibrosis (PMF) at diagnosis, using five baseline clinical variables to stratify patients into four risk groups. DIPSS (Dynamic IPSS) uses the same variables but allows recalculation at any time, giving more weight to anemia, and is therefore more aligned with real‑world practice where patients evolve over years. 6, 7, 1, 3
DIPSS considers the following factors: 8, 1, 6
- Age > 65 years
- Hemoglobin < 10 g/dL
- White blood cell (WBC) count > 25 × 10⁹/L
- Peripheral blood blasts ≥ 1%
- Presence of constitutional symptoms (weight loss, fevers, night sweats)
Each variable contributes adverse points that are summed from 0 to 6 and translated into four risk categories with distinct survival expectations. 6, 8
2.2 DIPSS Risk Categories and Survival Expectations
The original IWG‑MRT cohort and subsequent validations demonstrated clear separation of survival curves across DIPSS strata: 9, 3, 8, 6
| DIPSS risk group | DIPSS score | Approximate median survival* |
|---|---|---|
| Low | 0 | Prolonged; often > 15 years |
| Intermediate‑1 | 1–2 | Around 7–14 years |
| Intermediate‑2 | 3–4 | Around 3–4 years |
| High | 5–6 | Approximately 1–2 years |
*Estimates paraphrased from multiple cohort studies; individual outcomes vary and depend on treatment era, comorbidities, and access to transplantation or targeted agents. 2, 1, 8, 6
Clinical Insight: Figure 1. The clinical background section on OncoToolkit summarizes the DIPSS variables, outlines the additive point system, and paraphrases key validation data so clinicians and trainees can understand how the score was derived.
3. Why DIPSS Matters in Modern Myelofibrosis Practice
3.1 Central Role Across MF Subtypes
DIPSS was originally derived in PMF but has been widely applied to post‑polycythemia vera (post‑PV) and post‑essential thrombocythemia (post‑ET) MF, often together with MYSEC‑PM and other secondary MF tools. In routine practice, many centers still use DIPSS/DIPSS‑Plus as the backbone of risk‑adapted strategies, overlaying molecular and cytogenetic information to refine decisions. 7, 12, 1, 3
DIPSS informs several critical decisions:
- Allogeneic hematopoietic cell transplantation (allo‑HCT): Intermediate‑2 and high‑risk DIPSS categories generally signal a level of disease risk where transplant is strongly considered in suitable candidates. 13, 14, 1
- JAK inhibitor therapy: Intermediate‑1 to high‑risk patients with symptomatic splenomegaly or cytokine‑driven symptoms are common candidates for JAK inhibitors, with DIPSS providing prognostic framing for expected survival and treatment goals. 1, 10, 13
- Supportive versus intensive approaches: DIPSS helps clinicians differentiate patients who may benefit from intensive strategies from those where best supportive care, symptom management, and transfusion support are prioritized. 2, 1
- Clinical trial eligibility: Many MF trials specify DIPSS or DIPSS‑Plus risk strata in inclusion criteria or stratification schemas. 15, 10, 1
3.2 Reducing Cognitive Load with Digital Support
On paper, applying DIPSS requires remembering point assignments, manually summing scores, and mapping to survival tables—all while managing other prognostic models (IPSS, DIPSS‑Plus, MIPSS70, MYSEC‑PM, GIPSS). In practice, this can lead to: 16, 7, 1
- Miscounting points (especially the double weighting of anemia).
- Confusing IPSS and DIPSS thresholds.
- Inconsistent documentation across clinicians and time points.
OncoToolkit’s calculator is mobile‑responsive, fast, and visually streamlined, so hemato‑oncology teams can enter five binary parameters and obtain a clearly labeled risk group and survival estimate in seconds—on ward rounds, in transplant clinics, or during MDT meetings. This reduces “calculator fatigue” and helps standardize risk communication across the team. 4, 5
4. Clinical Evidence and Validation of the DIPSS Model
4.1 Model Development and Scoring Mathematics
DIPSS was developed using Cox proportional hazards regression in a large international PMF cohort, with variables selected and weighted according to their independent hazard ratios for overall survival. Anemia (hemoglobin < 10 g/dL) emerged as the strongest predictor and was therefore assigned 2 adverse points, whereas age > 65 years, WBC > 25 × 10⁹/L, circulating blasts ≥ 1%, and constitutional symptoms each carried 1 point. 8, 9, 1, 6
Our calculator faithfully implements this additive model:
- Age > 65 years – 1 point
- Constitutional symptoms – 1 point
- Hemoglobin < 10 g/dL – 2 points
- WBC > 25 × 10⁹/L – 1 point
- Circulating blasts ≥ 1% – 1 point
The resulting score (0–6) is mapped to risk groups using the same cutoffs as the original DIPSS publication, and the interface displays this mapping in an embedded reference table for transparency. 9, 6, 8
4.2 Key Validation Studies and Clinical Endpoints
Multiple studies have validated DIPSS and DIPSS‑Plus:
- The original IWG‑MRT study in PMF demonstrated robust prognostic separation for overall survival and leukemia‑free survival across DIPSS categories. 6, 8, 9
- DIPSS was later extended to create DIPSS‑Plus, adding three variables—unfavorable karyotype, platelet count < 100 × 10⁹/L, and red‑cell transfusion dependence—to further refine risk estimates. 17, 18
- Reviews and real‑world series confirm that DIPSS and DIPSS‑Plus remain practical tools even in the era of JAK inhibitors, with prognostic value retained when combined with genomic scores such as MIPSS70 and GIPSS. 19, 15, 16, 1
- Studies in transplant settings have shown that DIPSS risk, alongside factors like splenomegaly and pre‑transplant JAK inhibitor use, predicts post‑allo‑HCT outcomes and can guide timing of transplantation. 14, 20
Endpoints typically evaluated include:
- Overall survival
- Leukemia‑free survival / transformation to acute myeloid leukemia
- Transplant‑related mortality and relapse in allo‑HCT cohorts
- Symptom burden and quality‑of‑life measures in JAK inhibitor studies
4.3 Asian and Global Data Applicability
While early cohorts were heavily Western, more recent work has examined MF prognostic models in Asian populations:
- Retrospective series from Chinese and other Asian centers have demonstrated the prognostic impact of splenomegaly, cytopenias, and DIPSS‑related variables, supporting applicability in these populations. 11, 21
- Genomic scores such as GIPSS and MIPSS70 have also been tested in Asian cohorts, showing good discrimination when combined with clinical factors like DIPSS. 22, 11
OncoToolkit’s DIPSS calculator is therefore positioned as a global tool, while reminding clinicians to interpret survival estimates in context of local practice patterns, access to transplantation, and supportive care resources.
4.4 Limitations and Thoughtful Clinical Use
Important caveats when using DIPSS include: 3, 7, 1
- It does not directly incorporate mutational data (e.g., ASXL1, SRSF2, EZH2, IDH1/2), which are central to MIPSS70 and newer models.
- Application to post‑PV/ET MF is common but not identical to using MYSEC‑PM, which was specifically developed for secondary MF.
- Laboratory variability (e.g., blast counting technique, hemoglobin calibration, WBC thresholds) can influence whether patients cross DIPSS cutoffs.
- Treatment era effects mean survival in contemporary JAK inhibitor and transplant practice may differ from original cohorts.
Our calculator highlights these issues in the clinical context section to encourage nuanced interpretation rather than rigid adherence.
5. How the OncoToolkit DIPSS Calculator Works
5.1 Intuitive Input Form and Data Entry
The calculator interface is designed for rapid use by hematologists, hemato‑oncologists, and hematopathologists.
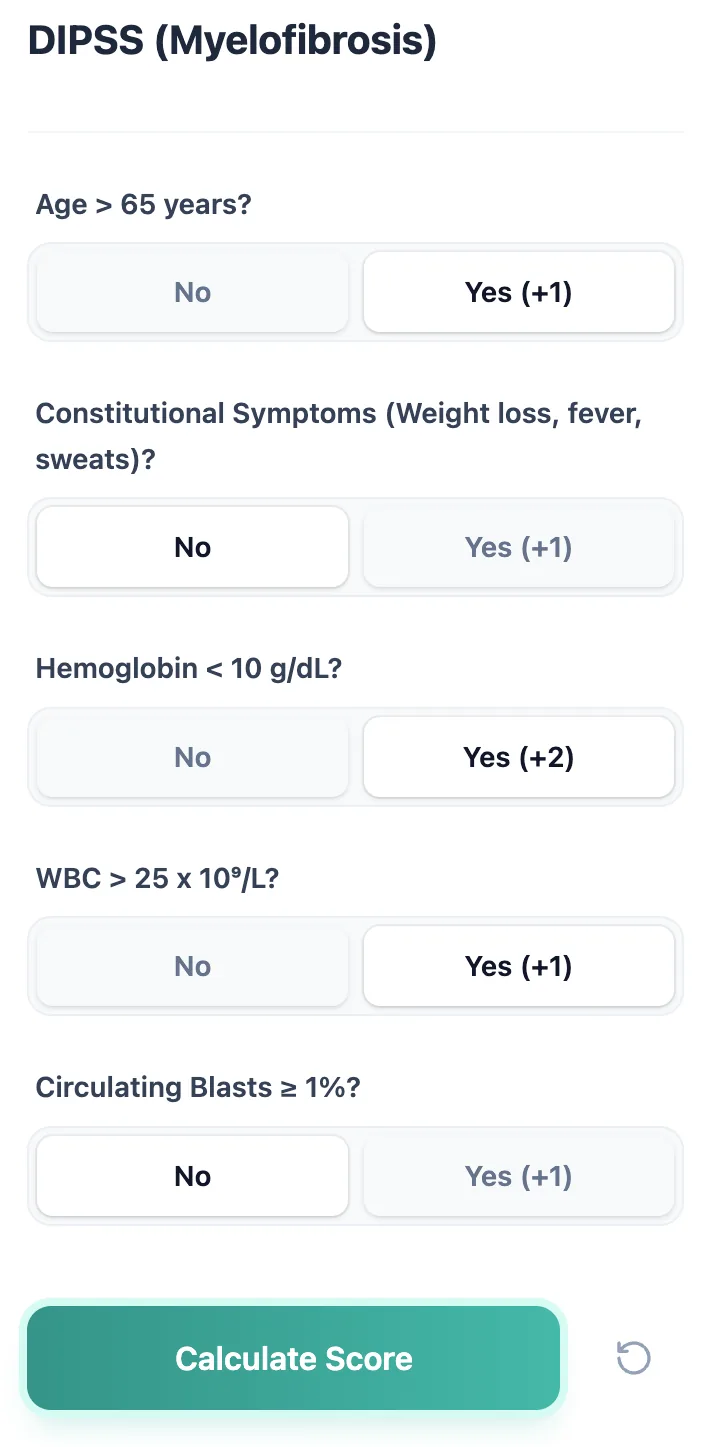
Figure 2. The input form presents each DIPSS variable as a clearly phrased yes/no question with the corresponding point value indicated, reducing errors and supporting quick bedside use.
Users simply answer five binary questions:
- Age > 65 years?
- Constitutional symptoms (weight loss, fevers, sweats)?
- Hemoglobin < 10 g/dL?
- WBC > 25 × 10⁹/L?
- Circulating blasts ≥ 1%?
Tooltips and brief explanations clarify typical definitions (e.g., what constitutes “constitutional symptoms”), helping to standardize data entry across clinicians.
5.2 Calculation Outputs and Results Interpretation
After clicking “Calculate Score”, the algorithm sums points and maps the total to risk groups and survival estimates.
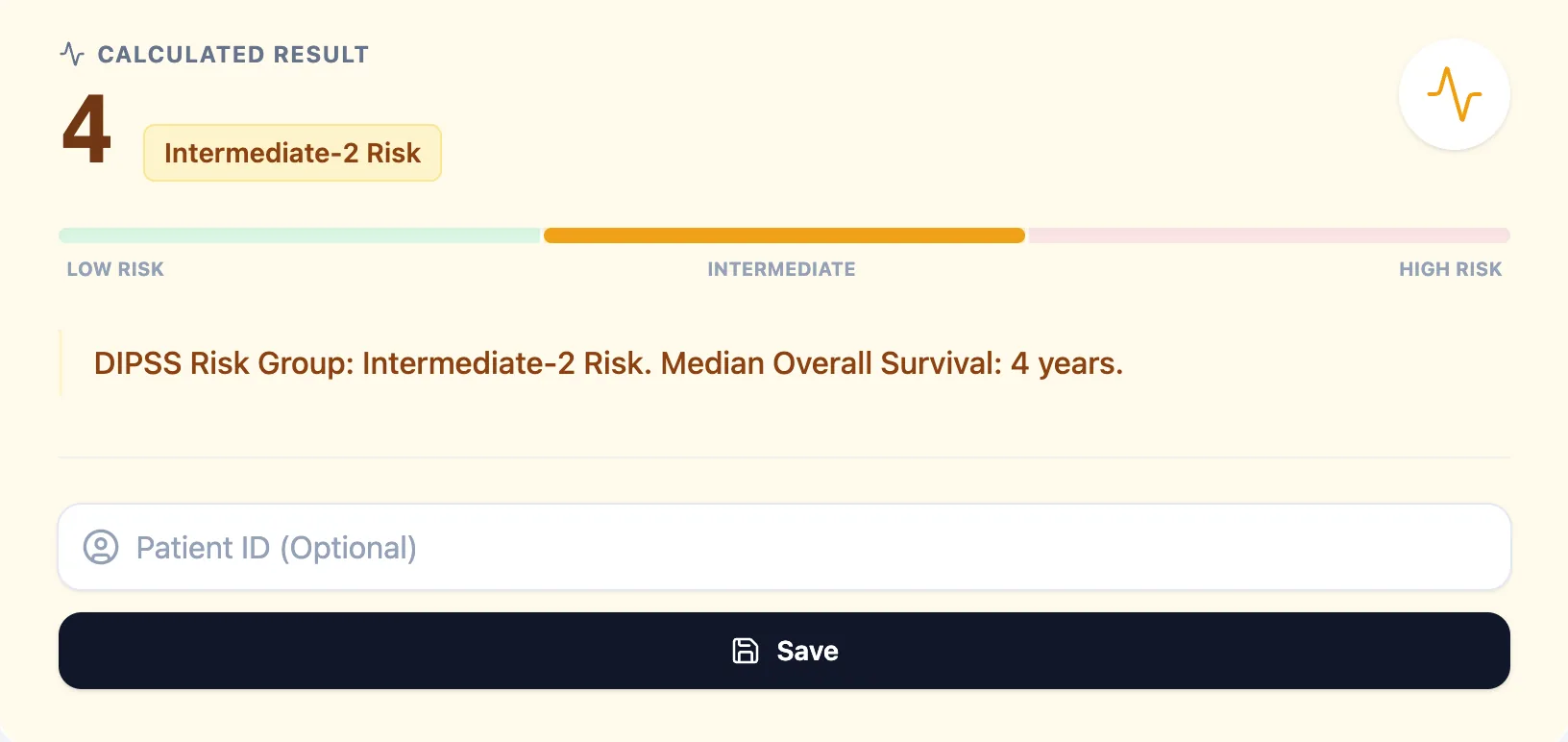
Figure 3. The results screen summarizes total DIPSS points, risk group, and median survival, with a color‑coded risk bar to support rapid interpretation during MDT discussions or transplant clinics.
The output includes:
- Total DIPSS score (0–6).
- Risk group (Low, Intermediate‑1, Intermediate‑2, High).
- Estimated median overall survival, paraphrasing published data.
- A visual bar from low to high risk.
- Optional patient ID field and buttons to save/export the result into the clinical record.
5.3 Embedded Reference Table and Formula Logic
Below the result, the calculator contains a quick‑reference section summarizing how score ranges map to risk groups.
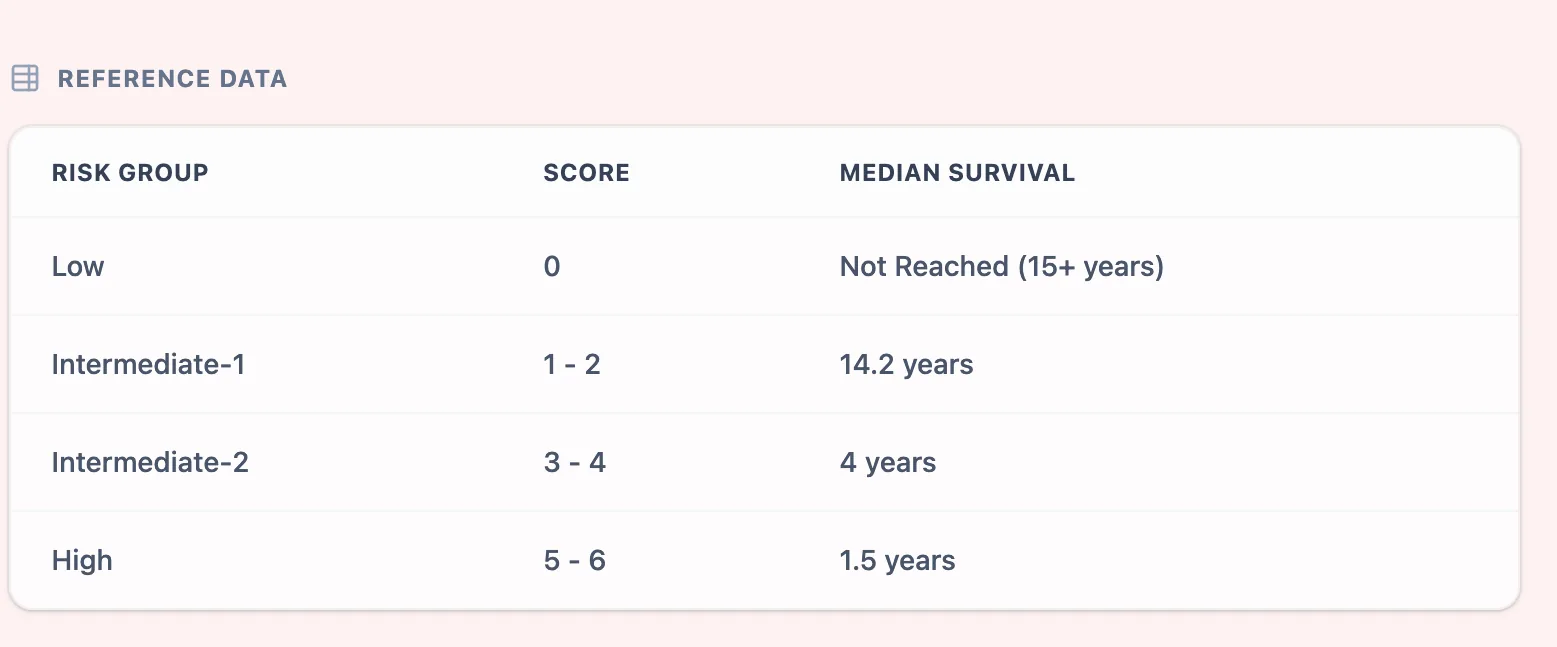
Figure 4. The reference table provides an at‑a‑glance mapping from DIPSS score to risk group and median survival, functioning as a teaching aid and a double‑check for clinicians who still calculate manually.
This section also reiterates the additive scoring logic, emphasizing that hemoglobin carries 2 points while other variables carry 1.
6. Supporting Clinical Care, Education, and Research
6.1 Routine Clinical Decision Support
In daily practice, the DIPSS calculator can be used to:
- Stage patients at diagnosis (together with IPSS) and at key inflection points such as new transfusion dependence, rising blasts, or transition to accelerated/blast phase. 1, 3
- Frame transplant discussions by documenting the patient’s DIPSS category at the time of referral and re‑evaluation, and by pairing it with transplant comorbidity indices like HCT‑CI using our internal calculators. 13, 14, 1
- Guide systemic therapy choices, using DIPSS risk group to contextualize starting JAK inhibitors, escalating therapy, or prioritizing clinical trial enrollment. 10, 13, 1
- Support shared decision‑making with patients by providing a transparent, numerical risk estimate that can be discussed alongside symptom burden, quality of life, and personal goals.
6.2 Education and Simulation for Hematology Trainees
For hematology fellows, residents, and advanced practice providers, OncoToolkit’s DIPSS calculator serves as an educational scaffold:
- Supervisors can construct simulated MF cases, altering hemoglobin or blast count to show how risk class shifts dynamically.
- Trainees can compare outputs from the IPSS calculator, DIPSS calculator, and DIPSS‑Plus myelofibrosis risk calculator, reinforcing understanding of how additional variables refine risk. 23, 7, 16, 17
- The visual risk continuum and survival benchmarks facilitate teaching about prognostic heterogeneity within MF and why risk‑adapted therapy matters.
6.3 Research Applications and Quality Improvement
Although batch entry is not currently supported, exporting or documenting DIPSS results enables:
- Prospective registries where DIPSS and DIPSS‑Plus can be tracked over time alongside treatments and molecular data. 3, 1
- Transplant outcomes studies correlating pre‑HCT DIPSS risk, splenomegaly, and JAK inhibitor exposure with post‑HCT survival and graft outcomes. 20, 14
- Quality improvement projects evaluating whether intermediate‑2/high‑risk patients receive timely transplant referral or access to JAK inhibitor therapy in line with guidelines. 15, 10
Our goal is to let clinicians move seamlessly from individual point‑of‑care calculations to aggregated analyses driven by the same reproducible scoring logic.
7. Clinical Application by Risk Group: Management Framework
DIPSS is best used as a decision framework rather than a rigid algorithm. Below we outline typical management considerations referenced in expert reviews and guidelines; these are not prescriptive treatment recommendations. 13, 15, 1, 3
7.1 Low‑risk (score 0)
- Often observed with periodic monitoring if asymptomatic, as survival may approach that of age‑matched controls in some series. 2, 1
- Symptom‑directed therapy (e.g., low‑dose aspirin, cytoreduction for concomitant PV/ET features) may be used based on local protocols.
- Transplant is generally deferred, but may be discussed in younger patients with high‑risk mutations or adverse cytogenetics, where genomic scores (e.g., MIPSS70) indicate higher biological risk than DIPSS alone. 19, 1, 3
7.2 Intermediate‑1 (score 1–2)
- Highly heterogeneous; younger patients with additional adverse features (ASXL1 mutation, high molecular risk, unfavorable karyotype) may be considered for early transplant. 19, 1, 3
- Symptomatic splenomegaly or constitutional symptoms often prompt JAK inhibitor therapy, with close monitoring of anemia and thrombocytopenia. 10, 1, 13
- For older or comorbid patients, strategies may emphasize symptom control, prevention of thrombosis, and careful surveillance for progression.
7.3 Intermediate‑2 (score 3–4)
- Generally viewed as “transplant‑eligible” disease in appropriate candidates given shorter median survival and higher transformation risk. 14, 1, 13
- Many centers initiate JAK inhibitors to reduce spleen size and symptoms, sometimes as a bridge to HCT; emerging data suggest this may improve performance status and transplant tolerance. 14, 13
- Clinical trial enrollment is often prioritized, particularly for patients ineligible for transplant or after JAK inhibitor failure.
7.4 High‑risk (score 5–6)
- Associated with very limited survival and high leukemic transformation rates. 24, 1, 6
- Fit patients may still be offered urgent transplant evaluation, recognizing that non‑relapse mortality is higher but remains the only potentially curative approach. 1, 13, 14
- For many, emphasis shifts to palliative and supportive measures (transfusions, symptom control, infection prevention), with or without disease‑directed therapy, guided by patient preferences and performance status. 2, 1
Throughout these scenarios, DIPSS should be integrated with molecular risk (MIPSS70/GIPSS), comorbidities (HCT‑CI), and individual goals, rather than used in isolation. 16, 3, 19, 1
8. Frequently Asked Questions (FAQ)
Can the DIPSS score be used in post‑PV and post‑ET myelofibrosis?
Yes, DIPSS is frequently applied to post‑PV and post‑ET MF, although it was originally derived in PMF. Secondary MF‑specific models such as MYSEC‑PM may perform better in some settings, but many clinicians still use DIPSS or DIPSS‑Plus alongside MYSEC‑PM to maintain consistency with guidelines and clinical trial criteria. 12, 7, 3, 1
How does DIPSS differ from IPSS, DIPSS‑Plus, and MIPSS70?
IPSS is a static prognostic tool applied at diagnosis, whereas DIPSS can be recalculated at any time and weighs anemia more heavily. DIPSS‑Plus builds on DIPSS by adding karyotype, platelet count, and transfusion status, offering finer discrimination of intermediate‑ and high‑risk disease. MIPSS70 and related genomic scores explicitly incorporate high‑molecular‑risk mutations, often used in transplantation‑age patients to refine risk beyond DIPSS or DIPSS‑Plus alone. On OncoToolkit, clinicians can move between the IPSS myelofibrosis risk calculator, DIPSS calculator, and DIPSS‑Plus dynamic myelofibrosis score to match the clinical question. 18, 7, 17, 16, 3, 19, 1
When should you be cautious about using the DIPSS score?
Use caution when laboratory values are close to DIPSS thresholds or when local reference ranges differ from those used in original cohorts. For example, borderline hemoglobin around 10 g/dL or blast counts near 1% may be interpreted differently depending on analytic methods and morphology review. Additionally, DIPSS does not account for high‑risk mutations or complex karyotype by itself, so genomic data should be integrated via scores like MIPSS70 or DIPSS‑Plus where available. 21, 7, 11, 19, 1
What is the most common mistake when calculating DIPSS?
Common pitfalls include: 3, 6, 1
- Forgetting that hemoglobin < 10 g/dL contributes 2 adverse points.
- Not formally screening for constitutional symptoms, leading to under‑scoring.
- Misclassifying low‑level blasts due to lack of standardized smear review.
The structured yes/no questions and explicit point labels in our calculator are designed to minimize these errors.
Is DIPSS validated for Asian as well as Western populations?
DIPSS and DIPSS‑Plus were originally derived mainly from European and North American cohorts, but subsequent studies have evaluated MF prognostic factors, including DIPSS variables, in Asian populations with broadly similar risk stratification. Regional differences in splenomegaly prevalence, comorbidities, and access to transplant or JAK inhibitors may influence absolute survival, so clinicians should interpret estimates in light of local experience and practice patterns. 11, 21, 22
9. Integrating DIPSS with Other OncoToolkit Calculators
Risk assessment in MF is inherently multimodal. OncoToolkit offers a growing suite of calculators that can be combined with DIPSS:
- IPSS myelofibrosis risk calculator – for baseline risk at diagnosis.
- DIPSS‑Plus myelofibrosis risk calculator – adds cytogenetic and transfusion variables for refined dynamic risk. 17, 18, 23
- Myelofibrosis transplant risk calculator (HCT‑CI) – to weigh disease‑related risk (DIPSS) against transplant‑related risk. 14, 1
These links help clinicians move from a single DIPSS estimate to a layered, risk‑adapted management strategy across the patient’s journey.
10. Conclusion
The DIPSS (Myelofibrosis) calculator on OncoToolkit is designed to fit seamlessly into real‑world hemato‑oncology workflows—whether you are evaluating a transplant‑age PMF patient, a frail older adult with post‑PV MF, or a complex case in a busy MDT. By turning a validated prognostic model into a clear, exportable digital interface, it helps standardize risk assessment, reduce cognitive load, and support high‑quality shared decision‑making.
OncoToolkit calculators are intended as decision‑support aids for healthcare professionals and must always be interpreted alongside clinical judgment, multidisciplinary input, and patient preferences.
Ready to Simplify Your Myelofibrosis Risk Assessment?
Access our clinical-grade DIPSS calculator and optimize your management decisions in seconds.
Try the DIPSS Calculator Now
Free to use. No registration required.
References
- Geyer H, et al. Management of Myelofibrosis. PMC11416430. Source
- Rare Disease Advisor. Myelofibrosis Prognosis. Source
- Tefferi A. Primary myelofibrosis: 2021 update on diagnosis, risk-stratification and management. PubMed 32886896. Source
- OncoToolkit Home. Source
- About OncoToolkit. Source
- Passamonti F, et al. A dynamic prognostic model to predict survival in primary myelofibrosis. PubMed 20008785. Source
- Guglielmelli P, et al. Prognostic impact of DIPSS and DIPSS-plus. ScienceDirect. Source
- Passamonti F, et al. DIPSS model for MF survival. Blood Journal. Source
- IWG-MRT DIPSS Validation. PMC3311279. Source
- MPN Hub. Applying Prognostic Models in Practice. Source
- Asian Myelofibrosis Outcomes Study. PubMed 39853812. Source
- Passamonti F. DIPSS vs MYSEC-PM in Secondary MF. Blood Journal. Source
- MF Transplant Guidelines 2024. PMC11532677. Source
- Gowin K, et al. DIPSS and Transplant Outcomes. PMC4465357. Source
- Targeted Oncology. Prognostic Stratification in MF. Source
- Targeted Oncology. Prognostic Risk Systems Detailed. Source
- Gangat N, et al. DIPSS-Plus for PMF. JCO. Source
- PubMed 21149668. DIPSS-Plus Validation. Source
- Guglielmelli P, et al. MIPSS70 and clinical-molecular models. JCO. Source
- PubMed 40912470. Pre-transplant Risk Factors. Source
- Blood Journal. Impact of Splenomegaly on MF Survival. Source
- Hematology Advisor. GIPSS in Asian MF Patients. Source
- The Hematologist. DIPSS-Plus Risk Calculator. Source
- PubMed 20947690. High-risk MF Survival. Source