Stage II Colon Cancer Risk Assessment for High‑Risk Features
A comprehensive guide on evaluating high-risk features in Stage II colon cancer to inform adjuvant chemotherapy decisions using evidence-based clinical criteria.
1. Clinical Context and Need for Structured Risk Assessment
Stage II colon cancer represents a heterogeneous group of patients who are node‑negative but still at variable risk for recurrence after curative resection. While many individuals have an excellent prognosis with surgery alone, a substantial subset has outcomes closer to those of low‑burden stage III disease because of specific adverse clinicopathologic characteristics. Differentiating between truly low‑risk and high‑risk stage II colon cancer is therefore central to modern decision‑making about adjuvant chemotherapy.1, 2, 3, 4
For practicing colorectal surgeons, pathologists, gastroenterologists, and medical oncologists, integrating multiple guideline‑defined high‑risk features into a clear, reproducible recommendation can be cognitively demanding, particularly in time‑pressured multidisciplinary team (MDT) discussions or busy clinics. On our platform at OncoToolkit, the Stage II Colon Cancer Risk Assessment calculator distills these variables into a simple, binary risk classification with concise, evidence‑based interpretation, helping clinicians rapidly determine whether adjuvant chemotherapy should be strongly considered or whether observation or fluoropyrimidine monotherapy is more appropriate.5
2. What is the Stage II Colon Cancer Risk Assessment?
The Stage II Colon Cancer Risk Assessment is a rules‑based clinical decision support tool that categorizes patients with resected stage II colon cancer into low‑risk or high‑risk groups based on the presence of guideline‑recognized adverse features. It does not estimate an exact numerical probability of recurrence; instead, it operationalizes the qualitative risk categories that underpin contemporary NCCN and ASCO recommendations on adjuvant therapy.6, 4, 7, 1, 5
The tool focuses on patients with pathologic stage T3N0 or T4N0 colon cancer. Key factors evaluated include:
- • T4 primary tumor: Including penetration to visceral peritoneum, perforation, or direct invasion.1, 6
- • Lymph node yield: Fewer than 12 nodes examined signaling potential understaging.3, 6
- • Histology: Poorly differentiated or undifferentiated grade 3 or 4 (excluding dMMR).8, 6
- • Invasion: Presence of Lymphovascular (LVI) or Perineural (PNI) invasion.6, 1
- • Clinical Presentation: Bowel obstruction or perforation at diagnosis.5, 1
- • MMR status: Attention to pMMR or microsatellite‑stable disease.8, 1
3. Why this Score Matters in Daily Practice
3.1 Linking Risk Groups to Adjuvant Chemotherapy Decisions
Risk stratification in stage II colon cancer is clinically important because it directly informs the choice to recommend adjuvant chemotherapy, the intensity of surveillance, and discussions with patients about prognosis. High‑risk patients may achieve a meaningful, though modest, absolute reduction in recurrence risk with adjuvant fluoropyrimidine‑based chemotherapy.9, 7, 10
3.2 Reducing Cognitive Load and "Calculator Fatigue"
Without a structured digital tool, clinicians must manually synthesize operative notes, pathology findings, and molecular testing. On our platform, the Stage II Colon Cancer Risk Assessment is designed to be mobile‑responsive and optimized for rapid use—typically requiring less than a minute to enter data. This integrated environment reduces cognitive load and allows clinicians to move seamlessly between anatomical risk stratification and genomic risk tools.11, 12
4. Clinical Evidence and Validation
4.1 Guideline and Consensus Basis
The conceptual foundation of this risk assessment is rooted in authoritative guideline documents and consensus statements. The most recent ASCO guideline updates highlight the importance of shared decision‑making and explicitly recognize that high‑risk stage II disease is often defined by a composite of these factors. Similarly, NCCN Guidelines (Version 3.2024) list these features and recommend that adjuvant chemotherapy be considered rather than automatically prescribed.5, 4
4.2 Typical Cohorts and Outcomes
Most validation data for this risk‑adapted approach come from retrospective cohort studies. High‑risk stage II patients consistently show higher recurrence and lower disease‑free survival than low‑risk stage II patients. Notably, recent large‑scale analyses of high‑risk stage II MSI‑high cohorts have shown that adjuvant chemotherapy may still confer survival benefit when multiple factors accumulate.2, 9
4.3 Western versus Asian Validation Data
While high‑risk criteria were primarily defined in Western datasets, Asian studies support the broad prognostic relevance of similar features. Asian recommendations often advocate more extensive D3 lymphadenectomy for T3/T4 tumors, potentially leading to higher lymph node yields. Despite surgical differences, both cohorts identify T4 stage and poor differentiation as consistently adverse factors.15, 16
4.4 Limitations and Caveats
The calculator treats each high‑risk factor as qualitatively similar and does not attempt to weight them differently. It should be viewed as a binary decision support tool rather than a personalized survival predictor. The tool supplements but does not replace clinician judgment and MDT discussion.2, 6
5. How the Stage II Colon Cancer Risk Assessment Calculator Works
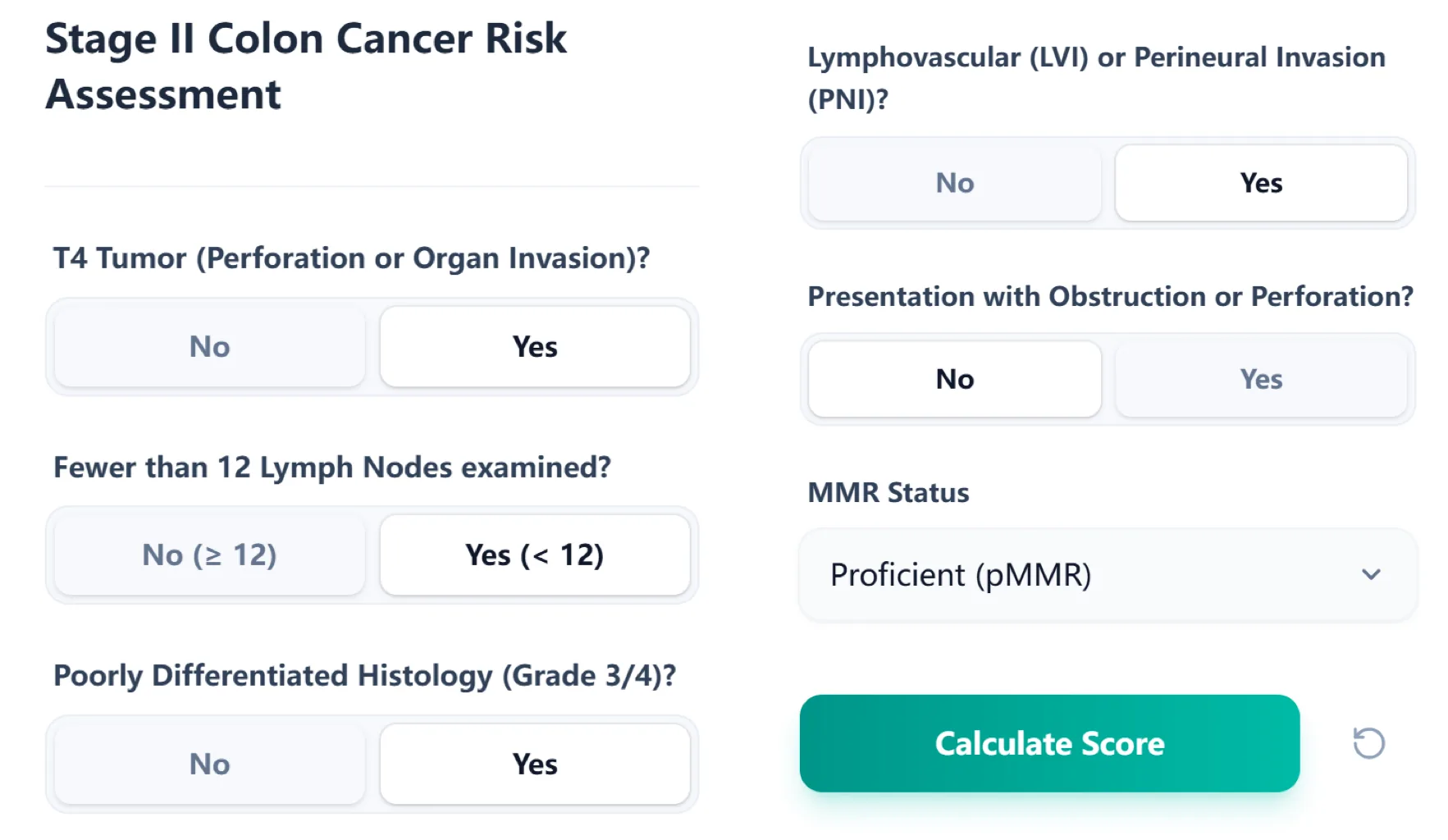
Figure 1. The input form collects key high‑risk features. This structured layout enables rapid, error‑resistant data entry at the point of care.
5.1 Step-by-Step User Workflow
- Confirm clinical context (T3N0 or T4N0)
- Enter tumor T stage
- Record lymph node yield (12-node threshold)
- Specify histologic grade (Grade 3/4)
- Document LVI/PNI
- Capture clinical presentation (obstruction/perforation)
- Enter MMR status
- Calculate risk summary
5.2 How Inputs Map to Outputs
The algorithm tallies the number of high‑risk features. Any tally of at least one high‑risk factor defines the high‑risk group. The total count is displayed prominently with a color‑coded risk bar.6
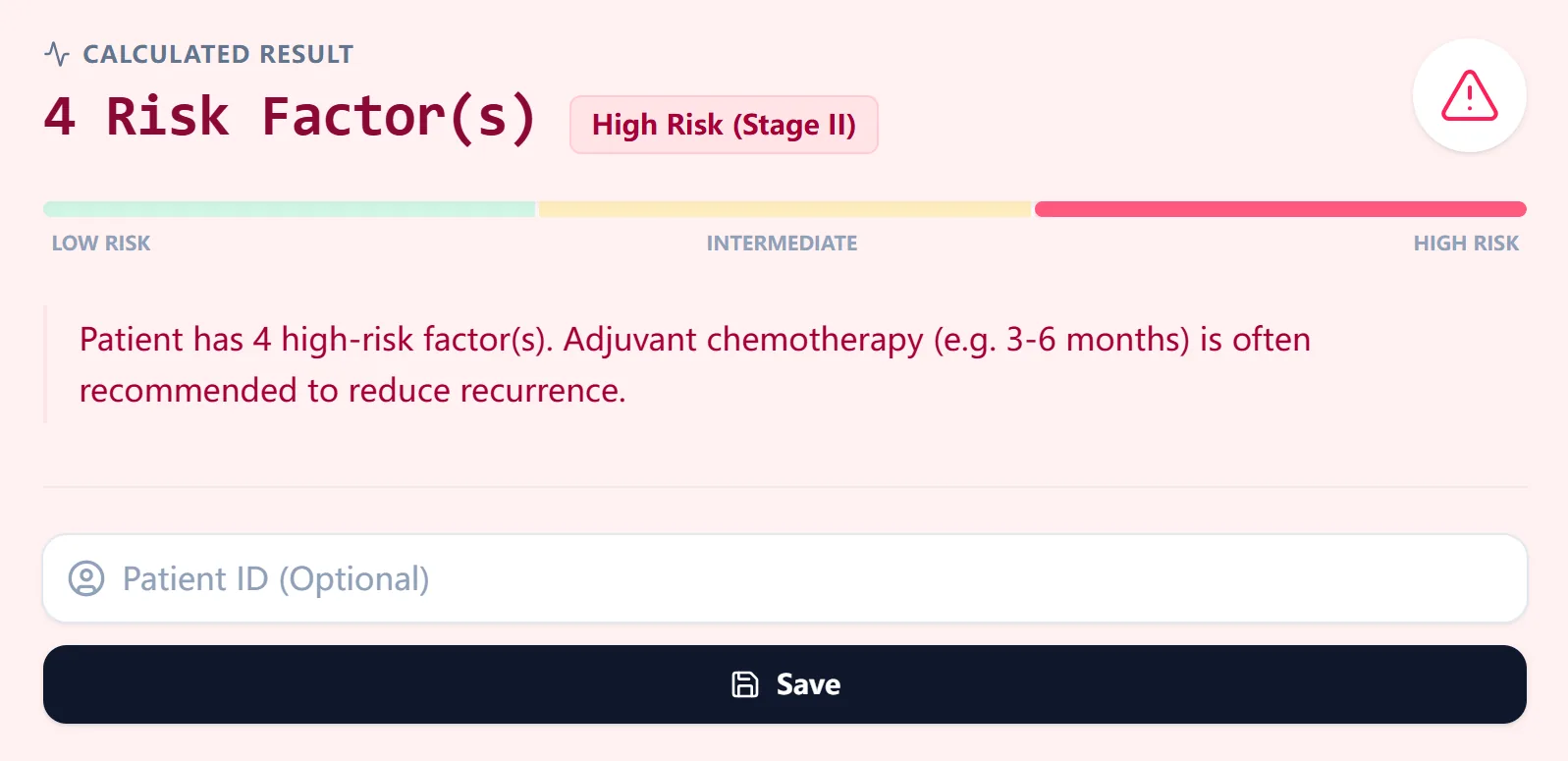
Figure 2. The calculated result displays the total number of factors and a guideline-aligned interpretation.
6. Reference Risk Groups and Suggested Management

Figure 3. The reference table links risk groups to broad management strategies.
7. How OncoToolkit is Used in Clinical Practice
- MDT preparation: Pre-populating fields for tumor board discussions.
- Point‑of‑care consultation: Reviewing high‑risk features with patients.
- Teaching sessions: Illustrating anatomical and molecular risk integration.
- Data standardization: Structuring institutional databases for quality audits.
8. Recent Advances and Refinements
Recent work shows that T4 tumors and very low node counts carry the greatest adverse impact. At OncoToolkit, this evidence base supports future enhancements, such as incorporating weighted high‑risk scores or integrating external nomograms that provide individualized recurrence probabilities.9, 19
9. How the Platform Supports Clinical Care, Education, and Research
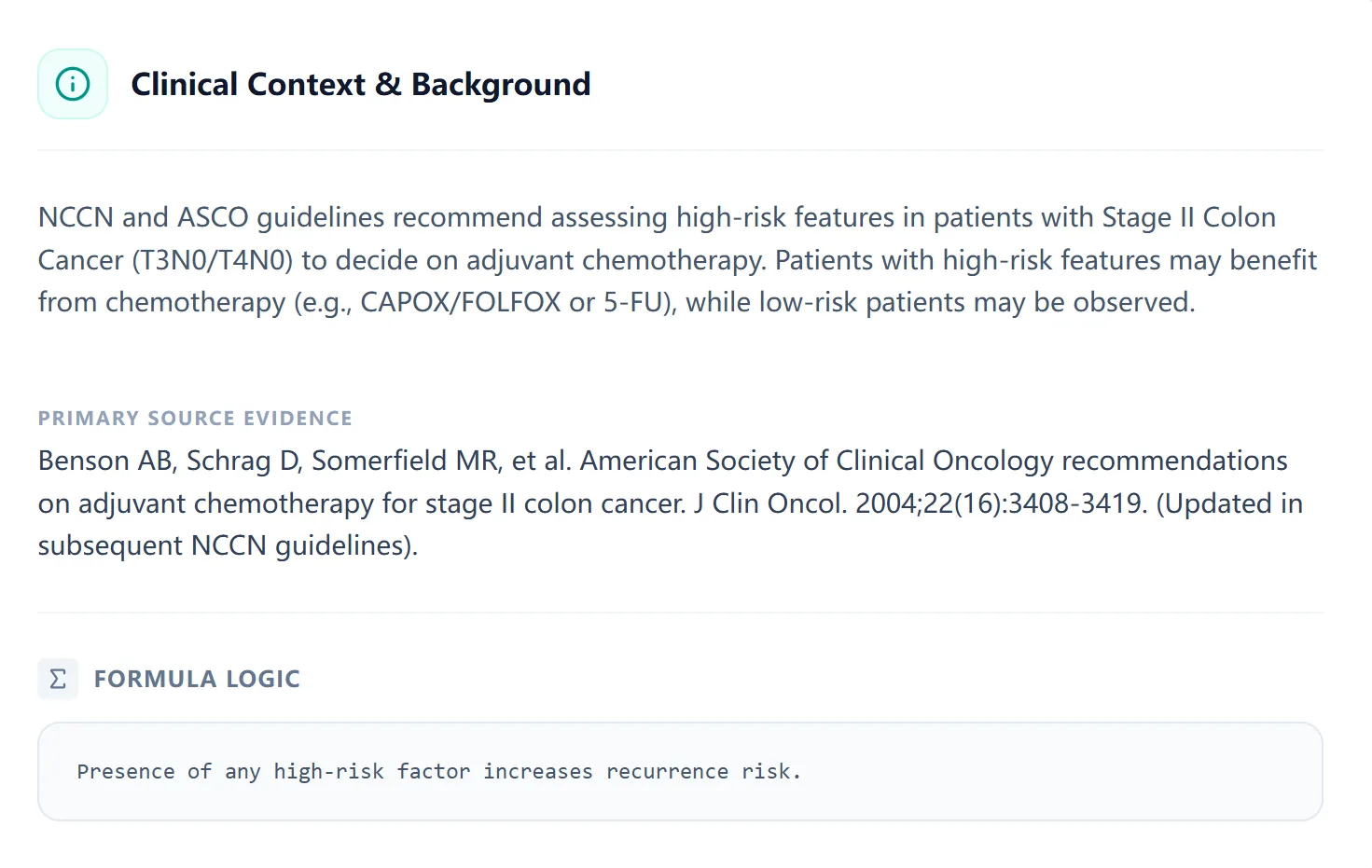
Figure 4. The clinical background panel summarizes guideline recommendations for trainees.
9.1 Routine Clinical Decision Support
Facilitating clear communication post-resection and in MDT meetings.
9.2 Education and Simulation for Trainees
Functioning as an interactive teaching aid for residents and fellows.
9.3 Research and Quality Improvement
Supporting uniform risk classification across subjects in cohort studies.
10. Clinical FAQs
When should you not use the calculator?
Not for stage I, III, or IV colon cancer, nor rectal cancer. Do not use before definitive staging or curative resection.1
How does this differ from TNM staging?
It focuses specifically on within‑stage risk heterogeneity among node‑negative tumors, complementing standard staging.20
What is the most common mistake?
Incomplete review of pathology reports (LVI/PNI) or finalizing plans before MMR status is confirmed.4
Is it valid in both Western and Asian populations?
Broadly applicable associations, but clinicians should remain attentive to regional practice patterns (e.g., lymphadenectomy extent).15
11. Call to Action and Integration into Your Workflow
To explore the tool in detail and integrate it into your practice, visit the Stage II Colon Cancer Risk Assessment calculator at:
https://oncotoolkit.com/calculator/colorectal-cancer-recurrence-risk
OncoToolkit hosts a growing suite of oncology decision‑support tools. Together, these resources are designed to reduce cognitive load and support evidence‑aligned care.12
Ready to Simplify Your Stage II Colon Risk Assessment?
Access our clinical tool to rapidly identify high-risk features and align with NCCN/ASCO guidelines.
Use the Calculator
Free to use. No registration required.
References
- 1. Pathologic Staging and Clinical Outcomes. Source
- 2. Risk Factors in Stage II Colon Cancer. Source
- 3. Management Strategies for Node-Negative Disease. Source
- 4. Updated Clinical Guidelines in Oncology (2024). Source
- 5. ASCO Guideline Update on Adjuvant Therapy. Source
- 6. Comprehensive Review of High-Risk Features. Source
- 7. Benson et al. Adjuvant Chemotherapy for Stage II. Source
- 8. Mismatch Repair Status in Colon Cancer. Source
- 9. Meta-analysis of MSI-High Cohorts. Source
- 10. IDEA Trial Takeaways for Stage II. Source
- 11. Oncotype DX Colon Recurrence Tool. Source
- 12. OncoToolkit Platform Overview. Source
- 13. Historical Consensus on Stage II Risks. Source
- 14. Surgical Outcomes and Recurrence. Source
- 15. Comparison of D2 and D3 Lymphadenectomy. Source
- 16. Taiwan Oncology Journal - CRC Guidelines. Source
- 17. CRS Guidelines Supplement. Source
- 18. Nutritional Modifiers in Asian Cohorts. Source
- 19. Nomogram Integration in Decision Support. Source
- 20. Deep Learning for CRC Recurrence. Source
- 21. NCI Colorectal Cancer Risk Tool. Source
- 22. MD Anderson CRC Decision Aid. Source
- 23. Cancer.gov Risk Calculator. Source