RPA (Brain Metastases) Calculator: Fast, Guideline-Aligned Prognostic Tool
Use the RPA (Brain Metastases) calculator to assign RTOG RPA class and estimate median survival in seconds, consistent with major neuro‑oncology guidelines and modern prognostic research.

Quick Navigation
1. RPA (Brain Metastases) Calculator: From Classic RTOG Classes to Modern Neuro-Oncology Practice
Brain metastases are a frequent complication of solid tumors and account for substantial morbidity and mortality in oncology practice. Accurate, rapid estimation of prognosis is central to choosing between stereotactic radiosurgery (SRS), whole-brain radiotherapy (WBRT), neurosurgical resection, systemic therapy, and supportive care. The Radiation Therapy Oncology Group (RTOG) Recursive Partitioning Analysis (RPA) for brain metastases remains one of the most widely recognized prognostic indices, even as more granular scores such as the Graded Prognostic Assessment (GPA) and diagnosis‑specific GPAs have emerged [1], [2], [3], [4], [5].
At OncoToolkit, we’ve built an RPA (Brain Metastases) calculator that turns this three-class system into a fast, mobile-responsive clinical decision support tool. On our platform, clinicians enter only four routinely available variables—Karnofsky Performance Status (KPS), age, primary tumor control, and presence of extracranial metastases—and the calculator immediately returns the RPA class, an estimated median survival, and an embedded reference table, reducing cognitive load and “calculator fatigue” in busy neuro‑oncology workflows [6], [5], [1].
2. What Is the RTOG RPA (Brain Metastases) Classification?
The RTOG RPA system was derived by Gaspar and colleagues using recursive partitioning analysis of approximately 1,200 patients enrolled in three prospective RTOG brain metastases trials evaluating WBRT regimens. Recursive partitioning analysis repeatedly splits the dataset along candidate prognostic factors to identify combinations of variables that best separate survival outcomes, ultimately generating a decision tree that can be simplified into clinically interpretable classes [7], [5], [8].
In the final model, four variables were retained as key prognostic discriminators:
01 Age (< 65 vs ≥ 65 years)
02 Karnofsky Performance Status (KPS ≥ 70 vs < 70)
03 Control status of the primary tumor (controlled vs uncontrolled)
Based on these variables, three mutually exclusive RPA classes were defined:
Age < 65, KPS ≥ 70, controlled primary tumor, and no extracranial metastases.
All patients not meeting criteria for Class I or Class III.
KPS < 70, regardless of other factors [5].
In the original RTOG dataset, median overall survival was approximately 7.1 months for Class I, 4.2 months for Class II, and 2.3 months for Class III. Subsequent validations confirmed that RPA reliably stratifies prognosis across multiple cohorts, treatment modalities, and primary tumor types [9], [8], [1], [7], [5].
3. Why the RPA Score Still Matters in Daily Oncology Practice
Although more sophisticated prognostic tools have been developed, RPA retains several important strengths in contemporary practice.
3.1 Simplicity and broad applicability
RPA is intentionally simple, relying on four variables that are almost always available at the time of initial brain metastases consultation: age, KPS, primary control, and extracranial disease. It is agnostic to tumor histology, making it applicable to patients with brain metastases from lung, breast, melanoma, renal cell, gastrointestinal, and other primaries. Because of this simplicity, RPA has been adopted as the “gold standard” baseline prognostic system in many comparative analyses of brain metastases scores [10], [1], [7], [6].
3.2 Shared language across specialties
RPA classes are widely recognized in radiation oncology, medical oncology, and neurosurgery, which improves communication and facilitates joint decision making in multidisciplinary tumor boards. Historical WBRT and SRS trials stratified outcomes by RPA class, so clinicians often interpret contemporary patients’ prognosis by referencing these classic RPA-based survival curves [11], [1], [9], [7].
3.3 Anchor for guideline and trial interpretation
Major guidelines, including ASTRO and ASCO recommendations on radiation therapy for brain metastases, emphasize performance status, extracranial disease status, and systemic treatment options as key determinants of care intensity. RPA operationalizes these dimensions and remains a useful anchor when interpreting legacy trials or educational materials that still report results by RPA class. Even when diagnosis-specific GPA scores are preferred, RPA provides a historical context and approximate risk gradient that many clinicians intuitively understand [2], [3], [12], [13], [1], [7].
4. Clinical Evidence, Validation, and the Math Behind RPA
4.1 Original derivation and survival separation
Gaspar et al. derived RPA from three RTOG randomized trials by using recursive partitioning analysis to explore how candidate variables partitioned overall survival. KPS emerged as the dominant prognostic factor, with patients having KPS < 70 experiencing markedly poorer outcomes, leading to the creation of RPA Class III. Within patients with KPS ≥ 70, younger age, controlled primary tumor, and absence of extracranial metastases were associated with the best survival, defining Class I, while all other combinations became Class II [8], [5].
4.2 Independent validations and systematic reviews
A comprehensive review of prognostic indices for brain metastases confirmed that RPA consistently stratifies patients into groups with significantly different median survival across various clinical series, though it shares limitations with other indices, such as large intermediate-risk groups and limited sensitivity for very long-term survivors. A systematic review of prognostic scoring systems identified nine different indices and reported that most—including RPA—achieved area-under-the-curve (AUC) values in the range of 0.64–0.90 when predicting poor-prognosis groups, underscoring their utility but also the room for refinement [10], [7], [1].
4.3 Comparisons with GPA and diagnosis-specific models
The Graded Prognostic Assessment (GPA) was developed from multiple RTOG datasets as a more granular scoring system, assigning points based on age, KPS, number of brain metastases, and extracranial disease. Later, diagnosis-specific GPAs further incorporated histology and molecular markers, such as EGFR and ALK status in NSCLC and breast cancer receptor subtypes, resulting in more accurate survival estimates by tumor type [14], [15], [16], [2].
4.4 Known limitations and thoughtful use
Several limitations of RPA are important in modern practice:
- It was derived in the pre–targeted therapy era and may underestimate survival in patients benefiting from contemporary systemic therapies such as EGFR/ALK inhibitors or immune checkpoint inhibitors [2], [16].
- It ignores number and volume of brain metastases, which strongly influence treatment eligibility for SRS and surgery [17], [1], [9].
- It does not capture comorbidities or geriatric vulnerability beyond KPS, which may be critical in older patients [7], [1].
5. How the OncoToolkit RPA Calculator Works
Our RPA (Brain Metastases) calculator faithfully implements the original RTOG classification while emphasizing usability, transparency, and guideline alignment.
Figure 1. The input form collects KPS, age, primary tumor status, and extracranial metastases through concise fields optimized for rapid data entry at the point of care.
5.1 Step 1: Enter core clinical variables
The calculator prompts clinicians to enter four variables:
- Karnofsky Performance Status (KPS) – Chosen from categorical options reflecting the ≥ 70 vs < 70 split central to RPA [5].
- Age – Entered as a numerical value; the algorithm automatically determines whether the patient is < 65 or ≥ 65 years [5].
- Primary tumor status – Selected as “Controlled” or “Uncontrolled,” capturing whether the primary tumor is stable or progressing [1], [5].
- Extracranial metastases – Recorded as “Yes” or “No” to reflect systemic metastatic burden [5].
5.2 Step 2: Apply RTOG RPA decision logic
Once inputs are entered, the calculator applies the canonical RPA logic:
- • If KPS < 70 → assign RPA Class III.
- • Else, if KPS ≥ 70:
- ◦ If age < 65, primary controlled, and no extracranial metastases → RPA Class I.
- ◦ Otherwise → RPA Class II [1], [5].
Figure 2. The background panel summarises the derivation of RPA from RTOG WBRT trials, listing the variables and decision rules used to assign each class and linking to the primary methodological publication.
5.3 Step 3: Interpret the result
The result view highlights the assigned RPA class and median survival.
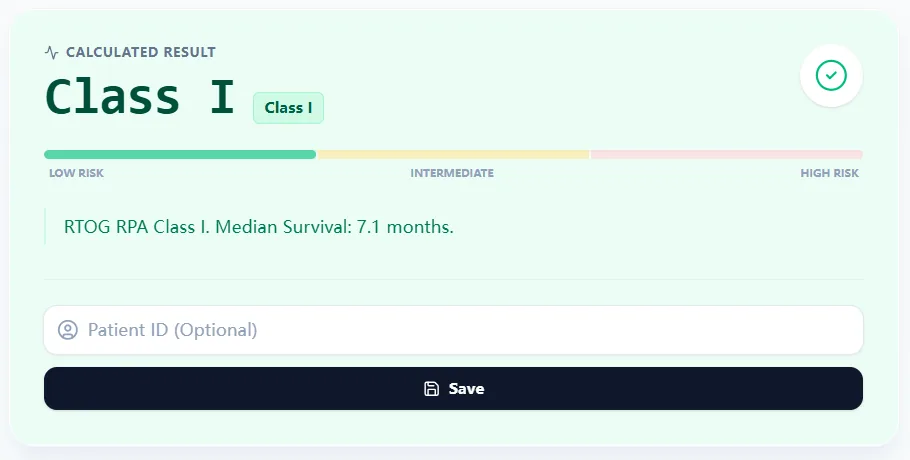
Figure 3. The result card displays the RPA class with a color‑coded risk continuum and a clear survival estimate, supporting rapid interpretation, charting, and communication with patients and colleagues.
Key elements include:
- Prominent display of RPA Class I, II, or III.
- A low–intermediate–high risk bar for intuitive visualization.
- A short text summary of estimated median survival in months based on RTOG data [8], [5].
5.4 Step 4: Review the reference table
To reinforce learning and transparency, the calculator includes a concise reference table summarizing class definitions and survival.
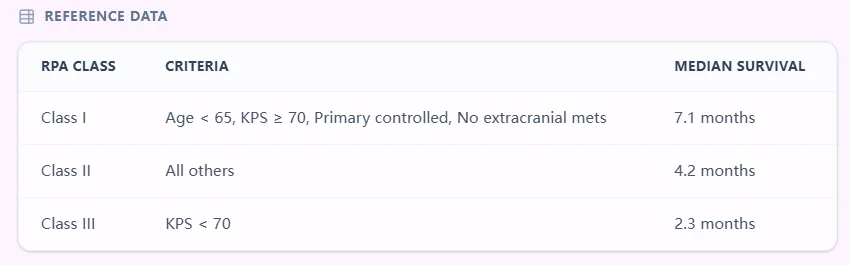
Figure 4. The embedded table presents RPA classes I–III with defining criteria and median survival times, allowing clinicians to cross‑check results and explore how changes in KPS, age, or systemic disease would alter class assignment.
6. Specific Clinical Use Cases and Applications
6.1 Initial consultation for newly diagnosed brain metastases
At the first visit, oncologists must integrate imaging, systemic disease status, and patient functional status to recommend SRS, WBRT, surgery, systemic therapy, or supportive care. RPA classification can help [3], [4]:
- Identify Class I patients with relatively favorable prognosis who are suitable for aggressive local approaches (SRS, surgery, or combined modalities) plus systemic therapy when indicated [4], [3], [1].
- Recognize Class III patients with poor performance status for whom guidelines suggest carefully weighing the limited survival benefit of WBRT against toxicity and logistical burden, often favoring supportive care or short-course palliation [12], [13], [3].
6.2 Multidisciplinary tumor board (MDT) planning
In MDT meetings, standardized prognostic language helps align neurosurgeons, radiation oncologists, medical oncologists, and palliative care specialists. The RPA calculator can be used to [1]:
- Pre-calculate RPA classes for all cases on the agenda and display them alongside MRI findings.
- Stratify cases for SRS vs WBRT vs best supportive care, particularly when numbers of metastases and systemic status differ [6], [3].
- Provide a common reference for comparing institutional outcomes with RTOG and other published series [9], [1].
6.3 Treatment re-evaluation over time
Performance status and systemic disease control can change substantially during the disease course. Clinicians can re-run the calculator when:
- KPS improves after corticosteroids, surgery, or rehabilitation.
- New extracranial metastases emerge on staging scans.
- Systemic therapy induces a deep response, altering prognosis [19], [9], [1].
6.4 Clinical trial eligibility and stratification
Many historical and some contemporary brain metastases trials stratify or report results by RPA class, and RPA remains a commonly used eligibility criterion. The calculator can rapidly screen patients for trials requiring minimum performance status or excluding RPA Class III [14], [11], [1].
6.5 Quality improvement and institutional benchmarking
Using the calculator systematically within a center enables analysis of local survival outcomes by RPA class compared with RTOG and modern cohorts, identifying patterns such as overuse of WBRT in RPA Class III or underuse of SRS in RPA Class I–II [11], [19], [1], [3], [12], [9].
7. Guideline Perspective: Where RPA Fits Today
7.1 ASTRO and ASCO guidelines on brain metastases
The 2022 ASTRO clinical practice guideline on radiation therapy for brain metastases and the corresponding ASCO endorsement emphasize an individualized approach guided by performance status, intracranial disease burden, extracranial disease status, and systemic therapy options. While they do not prescribe a single prognostic index, they acknowledge that scores like RPA and GPA have historically informed treatment decisions and trial design [12], [3].
7.2 EANO–ESMO guidelines
The 2021 EANO–ESMO clinical practice guideline for brain metastases highlights performance status, number of brain metastases, extracranial disease burden, and systemic treatment options as key factors for therapeutic planning. The guideline notes that several prognostic indices—including RPA and GPA—have been validated and can aid in decision making [4], [1].
7.3 NCCN and educational materials
NCCN education on CNS metastases similarly underscores KPS, intracranial disease burden, systemic status, and available systemic therapies when recommending SRS, WBRT, surgery, or supportive care. RPA is still used in many educational discussions and institutional pathways as a simple triage tool [13], [7], [6], [1].
8. Expansions, Modifications, and Updates to the Original RPA System
8.1 Single-institution refinements of RPA
Several groups have proposed extensions or modifications of RPA. Lutterbach et al. suggested subdividing RPA Class III based on age, primary control, and number of brain metastases to yield Classes IIIa–IIIc with more refined prognosis. Rades and colleagues developed an alternative prognostic index that uses age, KPS, extracranial metastases, and interval from primary diagnosis to WBRT [10], [1], [21].
8.2 Transition to GPA and diagnosis-specific GPAs
The most impactful “update” to RPA has been the development and widespread validation of GPA and diagnosis-specific GPAs. The 2020 multi-cohort GPA update further refined these models by incorporating modern systemic therapy outcomes and molecular markers, and introduced the concept of an eligibility quotient to improve clinical trial design [15], [16], [2], [14].
8.3 Contemporary prognostic research and AI-based models
Recent work has explored nomograms and machine‑learning models that integrate detailed imaging features, laboratory parameters, and treatment variables to further improve survival prediction for brain metastases. Although these models show promise, they are often more complex and less widely implemented than RPA [17], [19], [20].
9. Clinical FAQ: Using RPA in Modern Neuro-Oncology
When should you not rely solely on the RPA class? RPA should not be used as the sole prognostic tool when validated diagnosis‑specific GPAs and molecularly informed models are available for the patient’s tumor type, such as NSCLC, breast cancer, melanoma, or renal cell carcinoma [2], [14], [16], [18], [4], [10].
Can the RPA score be applied to patients with numerous brain metastases or leptomeningeal disease?
RPA was developed in cohorts that included patients with variable numbers of brain metastases but did not explicitly model lesion count or leptomeningeal spread. For patients with very high lesion burden or leptomeningeal disease, prognosis may be worse than RPA-based estimates suggest [12], [3], [4], [1], [5].
How does RPA differ from GPA and other modern scores?
RPA is a three-class system built on age, KPS, primary control, and extracranial metastases, whereas GPA is a point-based score that incorporates histology and molecular markers. Comparative studies indicate that GPA and diagnosis-specific GPAs generally have better discriminative ability [21], [14], [11], [2], [10], [1], [9].
What is the most common mistake when calculating RPA manually?
Common errors include mis-estimating KPS, misclassifying the primary tumor as “controlled” despite recent progression, and failing to recognize occult extracranial metastases on imaging [7], [1], [9].
Is the RPA score validated across Western and Asian populations?
RPA has been evaluated in multiple Western and Asian cohorts, and nearly all analyses confirm that it stratifies patients into groups with distinct survival, although absolute survival times vary with treatment era and systemic therapy availability [20], [17], [19], [1], [9].
10. Integrating the RPA Calculator Into Your Workflow
You can access the OncoToolkit RPA (Brain Metastases) calculator at: /calculator/rpa-brain
To integrate it effectively:
- Bookmark the calculator on your primary browser and mobile device.
- Document RPA class for all new brain metastases consults as a standardized prognostic descriptor.
- Use it in teaching by having trainees calculate RPA for case discussions.
- Leverage internal links on OncoToolkit using anchor text such as “brain metastases prognostic calculator,” “diagnosis-specific GPA,” and “performance status assessment” [16], [2], [18].
As with all decision support calculators, the RPA (Brain Metastases) tool is intended to enhance—not replace—expert clinical judgment. By combining a historically validated, guideline‑consistent prognostic framework with a streamlined digital interface, OncoToolkit aims to help neuro‑oncology teams deliver more consistent, data‑informed, and patient‑centered care [1].
Ready to Simplify Your Prognostic Assessments?
Assign RTOG RPA classes and estimate survival in seconds with our guideline-aligned tool.
Use the Calculator Now
Free to use. No registration required.
References
- Gaspar L, et al. Recursive partitioning analysis (RPA) of prognostic factors in three RTOG trials. Source
- Sperduto PW, et al. Summary report on the graded prognostic assessment. Source
- Gondi V, et al. ASCO Guideline Endorsement of ASTRO Guideline. Source
- Le Rhun E, et al. 2021 EANO-ESMO Clinical Practice Guidelines. Source
- Gaspar L, et al. RTOG recursive partitioning analysis. Source
- Vecht CJ, et al. RPA and GPA in patients with brain metastases. Source
- Sperduto PW, et al. A new prognostic index for patients with brain metastases. Source
- Gaspar LE, et al. RPA of prognostic factors. Source
- Lagerwaard FJ, et al. Identification of prognostic factors in 1292 patients. Source
- Zindler JD, et al. External validation of the GPA and RPA. Source
- Rades D, et al. Validation of the RPA and GPA. Source
- Schiff D, et al. Radiation Therapy for Brain Metastases: ASTRO Clinical Practice Guideline. Source
- Nabors LB, et al. NCCN Guidelines: Central Nervous System Cancers. Source
- Sperduto PW, et al. Diagnosis-Specific Graded Prognostic Assessment. Source
- Sperduto PW, et al. GPA for patients with brain metastases. Source
- Sperduto PW, et al. Updated Graded Prognostic Assessment (GPA). Source
- Fariselli L, et al. Machine learning models for brain metastases. Source
- Nieder C, et al. Lung adenocarcinoma GPA update. Source
- Miyatake S, et al. Survival analysis of brain metastases. Source
- Li J, et al. AI-based prognosis prediction in brain metastases. Source
- Rades D, et al. A new prognostic index for brain metastases. Source
- Gaspar L, et al. Validation of RPA. Source