Oncotype DX® Colon Recurrence Score Calculator: A Comprehensive, Guideline‑Aligned Guide for GI Oncologists
Use the Oncotype DX® Colon Recurrence Score calculator to individualize adjuvant therapy in stage II/III colon cancer. See guideline-aligned use cases, latest data, and practical tips.

Quick Navigation
1. Introduction: The Adjuvant Dilemma in Colon Cancer
Adjuvant chemotherapy decisions in stage II and early stage III colon cancer remain among the most debated topics in gastrointestinal oncology. Traditional high‑risk features—T4 lesions, lymphovascular invasion, inadequate nodal sampling—do not fully explain the wide heterogeneity in recurrence risk across patients.[1], [2]
The Oncotype DX® Colon Recurrence Score (CRS) assay adds a quantitative genomic layer by integrating expression of 12 genes into a 0–100 score that refines recurrence risk estimation beyond conventional clinicopathologic factors in resected stage II and anatomic stage III A/B colon cancers. At OncoToolkit, our Oncotype DX® Colon Recurrence Score calculator operationalizes this assay result within seconds, translating the laboratory‑reported CRS, tumor stage, and MMR status into clear risk categories and interpretive narratives that are aligned with contemporary evidence and guideline statements.[3], [4], [5], [2], [6]
This expanded article integrates specific use cases, guideline positions, and recent data updates to support GI, GIST, and peritoneal oncologists in using the calculator safely and effectively.
2. What is the Oncotype DX® Colon Recurrence Score?
The Oncotype DX® Colon Recurrence Score is a 12‑gene RT‑PCR assay performed on formalin‑fixed paraffin‑embedded colon tumor tissue. Seven cancer‑related genes (BGN, INHBA, Ki‑67, C‑MYC, MYBL2, FAP, GADD45B) and five reference genes are combined using prespecified Cox proportional hazards coefficients, then scaled to yield a continuous Recurrence Score from 0 to 100.[5], [2]
The assay has been analytically validated and clinically validated as a prognostic marker for recurrence risk in:
The CRS is prognostic—not directly predictive of chemotherapy benefit—but high scores correlate with higher baseline recurrence risk, and real‑world data suggest chemotherapy benefit is concentrated in high‑risk groups.[8], [6], [7], [5]
OncoToolkit’s calculator consumes the numeric CRS provided by the central lab and combines it with tumor stage and MMR status to output a risk group, approximate 3‑year recurrence risk, and tailored interpretive text for adjuvant decision‑making.[4]
Figure 1. The clinical context section on our platform summarizes the 12‑gene assay, eligible populations (stage II pMMR and stage III A/B colon cancer), and core trials such as QUASAR and CALGB 9581.[6], [7], [5], [2], [3]
3. Why the Colon Recurrence Score matters in modern GI oncology
3.1 Moving beyond “stage plus a few risk factors”
Guidelines such as the ASCO 2022 stage II colon cancer guideline and ESMO localized colon cancer guidance agree that adjuvant chemotherapy can be considered for high‑risk stage II patients but provide only broad risk groupings based on clinicopathologic features. These features—T4 tumors, perforation, positive margins, lymphovascular or perineural invasion, inadequate lymph node sampling, poor differentiation—are necessary but insufficient for individualized risk estimation.[9], [10], [1]
The CRS refines this paradigm by:
- Quantifying recurrence risk within clinicopathologic categories (e.g., among T3N0 pMMR patients).[7], [2], [6]
- Identifying patients with low gene‑expression risk who may derive little absolute benefit from chemotherapy.[8], [6], [7]
- Flagging patients with high gene‑expression risk, whose outcomes resemble those of anatomically more advanced disease and who may justify more intensive adjuvant therapy.[5], [3], [8]
By embedding this logic into a calculator, OncoToolkit helps clinicians apply genomic information consistently rather than relying on mental shortcuts.
3.2 Addressing cognitive burden and “calculator fatigue”
In busy GI oncology clinics, clinicians may handle multiple colon cancer cases per session, each with different pathologic features, MMR status, and now genomic assays. Recalling CRS cutoffs, interpreting them in context, and reconciling them with guideline language can be cognitively demanding.[11], [12], [13]
OncoToolkit’s calculator is mobile‑responsive, harmonized with other GI tools on the platform, and tuned for “few‑click” operation. It presents CRS results with color‑coded risk bars and concise text that highlights key guideline‑relevant points (e.g., limited role of 5‑FU monotherapy in dMMR tumors), helping reduce cognitive load and decision variability across clinicians.[4]
4. Clinical evidence and validation
4.1 Model development and formula logic
The CRS was derived from pooled analyses of large phase III trial cohorts, including QUASAR, CALGB 9581, and NSABP C‑07. Investigators used multivariable Cox proportional hazards modeling to relate gene expression to time‑to‑recurrence, adjusting for clinicopathologic factors such as T stage, nodal status, and MMR status.[14], [2], [7]
The linear predictor from the final model is transformed into the 0–100 CRS via proprietary scaling; higher scores correspond to higher relative hazard of recurrence. OncoToolkit does not recalculate gene expression; it uses the provided score to look up risk strata and calibrated recurrence probabilities based on these validation studies.[2], [4], [5]
4.2 Key prospective and real‑world studies
Representative evidence includes:
- O’Connell et al. (development/validation): Demonstrated that the 12‑gene CRC assay provides an independent, quantitative estimate of recurrence risk in stage II colon cancer, with hazard ratio per interquartile range of ~1.38 for recurrence after adjusting for clinicopathologic factors.[2]
- Prospective multicenter decision‑impact study: In stage II colon cancer, CRS testing changed adjuvant treatment recommendations in ≈29–45% of patients and was associated with decreased chemotherapy use in low‑risk cases.[11], [7]
- Real‑world Clalit cohort and expanded analyses (2023–2024): Among >800 stage II pMMR patients, CRS stratified 5‑year recurrence risk from ~13% in low RS (0–29) to ~30% in high RS (41–100); chemotherapy was used more frequently in high‑risk patients, and outcomes appeared risk‑concordant.[6], [3], [8]
- Large experience report (>20,000 stage II patients): Confirmed the reproducibility of CRS distribution and decision‑impact in broad clinical practice, supporting its stability over time.[15]
Collectively, these data support the CRS as a well‑calibrated, clinically meaningful prognostic tool for stage II and early stage III pMMR colon cancer.[7], [3], [8], [6], [2]
5. How guidelines and expert statements view the CRS
Guideline and policy positions are nuanced and vary by body:
- ASCO 2022 stage II colon cancer guideline: Notes that multigene assays (including the 12‑gene CRS) may provide additional prognostic information beyond standard factors but concludes there is currently insufficient evidence to routinely recommend them to guide adjuvant chemotherapy decisions outside of clinical trials or specific decision‑impact settings.[10], [1]
- NCCN Colon Cancer guidelines (as summarized in independent policy documents): State that “there are insufficient data to recommend the use of multi‑gene assays to determine adjuvant therapy” in stage II colon cancer, though they recognize that assays like Oncotype DX Colon may be used in select cases as prognostic tools.[16], [11]
- ESMO localized colon cancer guideline and consensus statements: Emphasize clinicopathologic risk and MMR status as the primary drivers of adjuvant decisions; multigene assays such as CRS are acknowledged in reviews but are not standard of care.[17], [9], [16]
- International and payer technology assessments (e.g., BlueCross, case‑study reviews): Agree that the CRS is prognostic and influences physician decision making but differ in whether its use is considered standard or investigational, often citing the need for more prospective outcome data.[16], [11]
OncoToolkit’s calculator is therefore positioned as a decision support tool for individualizing prognosis and facilitating shared decision making, not as a standalone determinant of chemotherapy use. This positioning is explicitly echoed in our interpretive text.
5.1 Limitations and caveats
Key limitations to convey when using the calculator include:
- Population boundaries: The CRS is validated primarily in resected, non‑metastatic colon cancer (not rectal), with pMMR stage II and stage III A/B most strongly supported.[18], [3], [6], [2]
- MMR biology: dMMR/MSI‑H tumors show favorable prognosis and may have paradoxical or absent benefit from fluoropyrimidine monotherapy; guidelines recommend prioritizing MMR testing itself over multigene assays in this group.[19], [1]
- Guideline reservations: ASCO, NCCN, and ESMO note that multigene assays should not replace established clinicopathologic risk factors and are not mandated for adjuvant decisions.[9], [1], [11], [16]
Our calculator reflects these nuances by displaying eligibility notes and explicit caveats within the results.
6. How the OncoToolkit Colon Recurrence Score calculator works
6.1 Intuitive inputs
OncoToolkit’s CRS calculator is structured around three key inputs that mirror clinical practice.[4]
Figure 2. The input panel accepts the 0–100 Recurrence Score, tumor stage (e.g., stage II T3N0 or stage III A/B), and MMR status.[4]
Required fields:
- Recurrence Score (0–100) – copied directly from the lab report.
- Tumor stage – focused on stage II and stage III A/B colon cancer.
- MMR status – pMMR vs dMMR/MSI‑H, reflecting its major therapeutic implications.[19], [1]
Optional: a de‑identified Patient ID to facilitate saving and later retrieval of results for MDT or registry use.[4]
7. Internal logic: mapping CRS to risk groups and recurrence estimates
After submission, the calculator maps the CRS to risk groups and approximate 3‑year recurrence risk based on published calibration data.[8], [4]
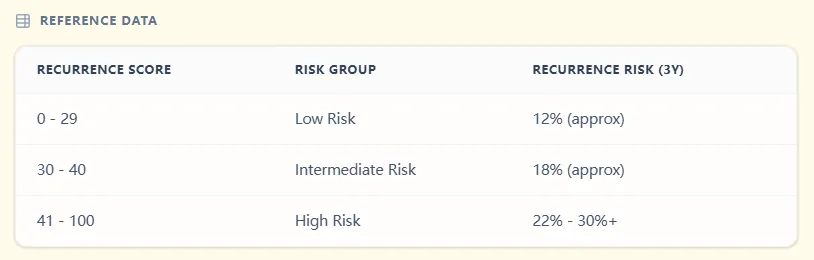
Figure 3. The reference table shows CRS ranges, corresponding risk groups, and approximate 3‑year recurrence risks.[6], [8], [4]
| Recurrence Score | Risk group | Approx. 3‑year recurrence risk |
|---|---|---|
| 0–29 | Low risk | ~12% |
| 30–40 | Intermediate risk | ~18% |
| 41–100 | High risk | ~22–30%+ |
8. Specific clinical use cases for the calculator
8.1 Stage II pMMR T3N0 with borderline high‑risk features
A common scenario is a 65‑year‑old with stage II (T3N0), pMMR colon cancer, 16 nodes examined, and focal lymphovascular invasion but no T4 or obstruction. Traditional guidelines would consider this “intermediate/high‑risk,” leaving room for either observation or fluoropyrimidine ± oxaliplatin.[9], [1]
Using the calculator:
- If CRS=18 (low risk), OncoToolkit categorizes the patient as low risk (~12% 3‑year recurrence) and highlights that the absolute benefit of chemotherapy is likely modest, supporting observation.[8], [6], [4]
- If CRS=45 (high risk), the tool reports a ~22–30%+ recurrence risk and notes that outcomes resemble those of higher anatomic stages, supporting consideration of chemotherapy, potentially with oxaliplatin, in line with guideline flexibility.[5], [3], [8]
8.2 Stage II pMMR T4 disease
For T4 lesions, guidelines already lean strongly toward adjuvant chemotherapy due to high baseline risk. In such cases, CRS is not used to withhold chemotherapy but may refine discussions around regimen intensity and duration.[9], [1]
- A T4N0 pMMR patient with CRS in the low or intermediate range may still receive FOLFOX or CAPOX, but the calculator can be used to contextualize risk and explain why a shorter duration or selection of regimen is reasonable.[3], [1]
- A very high CRS could further reinforce the need for aggressive adjuvant therapy and closer surveillance.[5], [8]
8.3 Stage III A/B pMMR disease with favorable clinicopathologic profile
For anatomically low‑burden stage III (e.g., T1–2N1, “low‑risk stage III”), guidelines now allow shorter oxaliplatin duration and sometimes de‑escalation strategies. In such patients, CRS can help:[1], [9]
8.4 Multidisciplinary tumor board standardization
MDTs often discuss dozens of colon cancer cases per session. The CRS calculator enables:
8.5 Patient counseling and shared decision making
Patients frequently ask, “What is my personal risk of recurrence?” and “How much will chemotherapy help?” The CRS and the calculator provide a basis for:
- Explaining approximate recurrence percentages aligned with published real‑world data (e.g., ~13% 5‑year recurrence for low RS vs ~30% for high RS).[6], [8]
- Clarifying that chemotherapy benefit is more likely meaningful in high‑risk patients, while in low‑risk patients the absolute benefit may be small.[7], [1], [8]
9. How the platform supports care, teaching, and research
9.1 Routine decision support in GI oncology
OncoToolkit’s CRS calculator fits naturally into GI oncology workflows:
- Post‑operative clinics: Quickly interpret new CRS reports, document the risk category, and agree on adjuvant plans.[4]
- Inpatient consults: Review CRS for patients recovering from surgery while planning for adjuvant therapy or clinical trial referrals.[3], [6]
- Regional tumor boards: Provide a shared, web‑based interface for CRS interpretation across institutions.[7], [6]
9.2 Education and simulation for trainees
For GI fellows and residents, the calculator offers an interactive way to understand prognostic modeling:
9.3 Research, registries, and quality improvement
Because the calculator standardizes CRS categorization and interpretation, it is well suited for:
10. Recent advances, expansions, and updates
10.1 Evolving real‑world evidence (2023–2024)
Recent work has enriched our understanding of how CRS performs outside clinical trials:
- A large real‑world dataset reported in 2023–2024 evaluated stage II pMMR patients whose treatment decisions incorporated CRS results, showing that chemotherapy was used in ~13% of low RS vs ~60% of high RS patients, with 5‑year recurrence risks aligning with test predictions (≈13% vs >30%).[3], [8], [6]
10.2 Expanded messaging around MMR and molecular markers
Guidelines increasingly emphasize MMR status, BRAF V600E, and other molecular markers as essential for adjuvant decision making. educational content within OncoToolkit’s CRS calculator has been updated to:[19], [9]
10.3 Position of CRS among other multigene assays
Other prognostic assays such as ColoPrint and ColDx have also been developed for stage II colon cancer. Comparative policy reviews note that all three assays provide prognostic information, but the 12‑gene CRS has some of the most extensive validation and real‑world decision‑impact data.[11], [16]
11. Frequently asked questions (FAQ) for clinicians
When should you not rely solely on the Colon Recurrence Score?
CRS should not be used as the sole determinant in T4 tumors, dMMR/MSI‑H tumors, or rectal cancers, stage I, or stage III C disease, where the assay is not validated.[18], [2], [6]
How does the CRS differ from clinicopathologic “high‑risk” criteria?
Clinicopathologic criteria group patients based on coarse features. CRS provides a continuous measure of tumor biology that is independent of, and additive to, these features.[2], [7]
What is the most common mistake when using the CRS and the calculator?
Applying CRS results to rectal primaries or mislabeling MMR status. OncoToolkit mitigates these by embedding eligibility reminders.[19], [1]
12. Conclusion: Integrate the CRS calculator into your GI practice
The Oncotype DX® Colon Recurrence Score adds granular prognostic information to stage II and early stage III pMMR colon cancer, with robust validation and increasing real‑world data. Bookmark the calculator, trial it on a series of recent stage II/III cases, and incorporate it into your next MDT discussion. Over time, this integrated, guideline‑aware decision support ecosystem can help your team deliver more consistent, evidence‑based, and patient‑centered care in GI oncology.[2], [7], [8], [6], [3], [4]
Ready to Optimize Your Adjuvant Decisions?
Access our guideline-aware calculator to individualize prognosis with precision.
Use the Calculator Now
Free to use. No registration required.
References
- ASCO 2022 Stage II Colon Cancer Guideline. Source
- O’Connell et al. (2010) Development and Validation of 12-gene CRS. Source
- ScienceDirect Analysis (2024) Real-world Evidence. Source
- OncoToolkit Oncotype Colon Calculator Tool. Source
- Exact Sciences Precision Oncology Portal. Source
- PMC Israel Clalit Real-world Evidence (2024). Source
- PMC QUASAR Trial Clinical Validation Analysis. Source
- ASCO Publications CALGB 9581 Data Analysis. Source
- ESMO Localized Colon Cancer Guidance Statement. Source
- Angen23 ASCO Policy Document (2024). Source
- BlueCross Laboratory Policy for Multigene Assays. Source
- PMC Review on Cognitive Burden. Source
- PMC Decision Fatigue in Cancer Care Analysis. Source
- NSABP C-07 and Oncotype DX Methodology Policy. Source
- ASCO 2018 Experience Report. Source
- PMC NCCN Guideline Summary. Source
- Oxford RDM Publication. Source
- Exact Sciences Colon Cancer Validation. Source
- PMC Analysis on MMR Status (2024). Source
- Oncotype DX Recommended Guidelines. Source
- ASCRS Toolkit: Management 2022. Source
- Exact Sciences Comparison Report. Source
- Hemonc.org Wiki Colon Cancer. Source
- Henry Ford Pathology NCCN Genetics. Source