Modified Glasgow Prognostic Score (mGPS) Calculator for GI Oncology
The Modified Glasgow Prognostic Score (mGPS) transforms well-validated inflammation-based prognostic scoring into a rapid, point‑of‑care tool for oncologists and GI surgeons.
Quick Navigation
1. Introduction to the Modified Glasgow Prognostic Score (mGPS) in Surgical Oncology
Gastrointestinal (GI) and other solid-organ cancers often follow complex clinical courses in which tumor stage, performance status, and systemic inflammation all influence outcomes. Anatomical staging systems alone rarely capture the host inflammatory and nutritional state that drives postoperative risk, treatment tolerance, and survival. 1, 2
The Modified Glasgow Prognostic Score (mGPS) calculator on our OncoToolkit platform transforms a well-validated inflammation-based prognostic score into a rapid, point‑of‑care tool for oncologists and GI surgeons, helping them quantify systemic inflammation and integrate it into routine decision-making. By combining C‑reactive protein (CRP) and albumin, the calculator provides a simple stratification (scores 0–2) that can be applied across colorectal, gastric, pancreatic, breast, lung, and other cancers. [3], [2], [1]
2. What is the Modified Glasgow Prognostic Score (mGPS)?
The mGPS is an inflammation-based prognostic system that assigns patients to three categories based on serum CRP and albumin:
- mGPS 0: CRP ≤10 mg/L (albumin level does not change the score).
- mGPS 1: CRP >10 mg/L with albumin >=35 g/L.
- mGPS 2: CRP >10 mg/L with albumin <35 g/L. [4], [3], [1]
Higher mGPS reflects a stronger systemic inflammatory response and/or hypoalbuminemia, both of which correlate with poorer overall and cancer-specific survival, increased postoperative complications, and earlier recurrence in multiple tumor types. The system evolved from the original Glasgow Prognostic Score (GPS), which considered low albumin alone abnormal; the “modified” version reclassified isolated hypoalbuminemia to improve the specificity of inflammation-related risk. [5], [6], [2], [3], [1]
Over two decades, mGPS has been applied and validated in diverse cancers—including colorectal, gastric, pancreatic, renal cell, lung, breast, and head and neck malignancies—making it one of the most extensively studied inflammation-based scores. [7], [6], [2], [3], [5], [1]
3. Why the mGPS Matters in Practice
3.1 Prognosis, perioperative risk, and systemic therapy
For GI oncologists and surgeons, the mGPS provides a practical lens on host factors that traditional staging may miss:
- Colorectal and rectal cancer: Elevated mGPS predicts poorer overall and disease‑free survival after elective colorectal resections, including rectal cancer patients undergoing neoadjuvant chemoradiotherapy and surgery. [6], [8], [3]
- Gastric and pancreatic cancer: Literature reviews and meta-analyses demonstrate that higher GPS/mGPS scores are associated with worse survival and can help identify gastric cancer patients less likely to benefit from platinum-based chemotherapy; similar prognostic value has been reported in pancreatic cancer, although some studies show heterogeneity. [5], [1]
- Metastatic and palliative settings: In metastatic breast and other advanced cancers, mGPS independently predicts overall survival and can complement ECOG performance status when counseling patients about realistic outcomes and treatment intensity. [9], [2]
Clinical Pearl: Without digital support, clinicians must manually interpret lab reports and reconcile them with multiple inflammation-based scores. On our platform, the mGPS calculator centralizes this logic into a single interface, reducing “calculator fatigue” and standardizing risk discussion.
4. Clinical Evidence and Validation of the mGPS
4.1 The math behind the tool
The mGPS is a categorical, point-based score, not a continuous nomogram. It uses fixed thresholds for CRP (10 mg/L) and albumin (35 g/L) to allocate 0, 1, or 2 points, which correspond to progressively worse prognosis. This simplicity is one of its strengths: the score is easily applied, yet retains strong associations with survival in multivariable Cox models controlling for stage, performance status, and treatment modality. [2], [3], [1], [4], [5]
A high-sensitivity variant, the HS‑mGPS, lowers the CRP cutoff to 3 mg/L to detect milder systemic inflammation. Studies in head and neck cancer and other tumors suggest that HS‑mGPS may provide better discrimination of overall survival than conventional mGPS, with adjusted hazard ratios for death exceeding 3 when comparing HS‑mGPS 2 versus 0. [10], [11], [7]
4.2 Western vs Asian validation data
The prognostic value of mGPS has been confirmed in both Western and Asian populations, though HS‑mGPS data remain largely Asian at present.
- Western cohorts
- European and North American studies in colorectal and renal cell carcinoma have shown that higher mGPS independently predicts worse overall and cancer-specific survival after surgery or systemic therapy. [8], [3], [6]
- Reviews of gastric cancer literature highlight GPS/mGPS as superior or comparable to performance status and other inflammatory markers for prognostication, supporting routine use in Western oncology practice. [1], [2]
- Asian cohorts
- Multiple Japanese, Korean, and Chinese series in gastric, pancreatic, and head and neck cancers report that elevated mGPS and HS‑mGPS are strongly associated with shorter overall survival and higher postoperative complications. [11], [10], [7], [5]
- A meta-analysis of HS‑mGPS included only Asian studies but showed consistent associations across tumor types, with higher scores predicting significantly worse survival, although the authors called for Western validation and cautioned about publication bias. [10]
Taken together, these data support mGPS as a globally applicable inflammation-based score, with strong evidence in both Western and Asian settings, while HS‑mGPS currently has stronger evidence in Asian cohorts and is emerging in other regions. [3], [7], [5], [10]
4.3 Guideline and consensus statements
While mGPS is not embedded as a formal staging criterion in disease-specific guidelines, expert reviews and consensus documents on systemic inflammation in cancer emphasize GPS/mGPS as the most extensively validated inflammation-based prognostic score and recommend its integration into routine assessment where feasible. [12], [2], [1]
- Reviews in gastric cancer describe GPS/mGPS as “tumor stage- and treatment-independent” predictors that can guide decisions about active vs palliative treatment and selection of patients for intensive chemotherapy. [1]
- Broader inflammation-focused reviews conclude that GPS/mGPS identify high-risk patients across many clinical scenarios and may serve as therapeutic targets in future trials. [12], [2]
As such, mGPS is increasingly recognized in expert commentaries, survivorship discussions, and perioperative optimization protocols, even if not specified line-by-line in individual disease guidelines.
4.4 Expansions and updates of the original system
Recent work has proposed several refinements to the original mGPS framework:
- High-sensitivity mGPS (HS‑mGPS): Uses a CRP threshold of 3 mg/L instead of 10 mg/L, improving sensitivity to low-grade inflammation and showing superior prognostic performance in several cancers, including head and neck malignancies. [7], [11], [10]
- Cancer-specific adaptations: Studies in pancreatic, rectal, and renal cell carcinomas have tested mGPS in combination with other factors (e.g., TNM stage, CA19‑9, PNI), sometimes generating composite prognostic indices where mGPS remains a key component. [6], [3], [5]
- Comparisons with GPS and alternative scores: Work in hepatocellular carcinoma and other tumors suggests that, in certain contexts, the original GPS or other inflammation markers may outperform mGPS, underscoring that choice of index should remain disease- and context-specific. [13]
At OncoToolkit, we have focused our calculator on the conventional mGPS thresholds most widely used across GI oncology, while surfacing HS‑mGPS and composite-score evidence within the background text for users who wish to explore more advanced applications.
5. How the OncoToolkit mGPS Calculator Works
The mGPS calculator on OncoToolkit is built for speed and clarity, with a layout optimized for desktop and mobile use. Clinicians need only:
- C‑reactive protein (CRP), in mg/L.
- Serum albumin, in g/L.
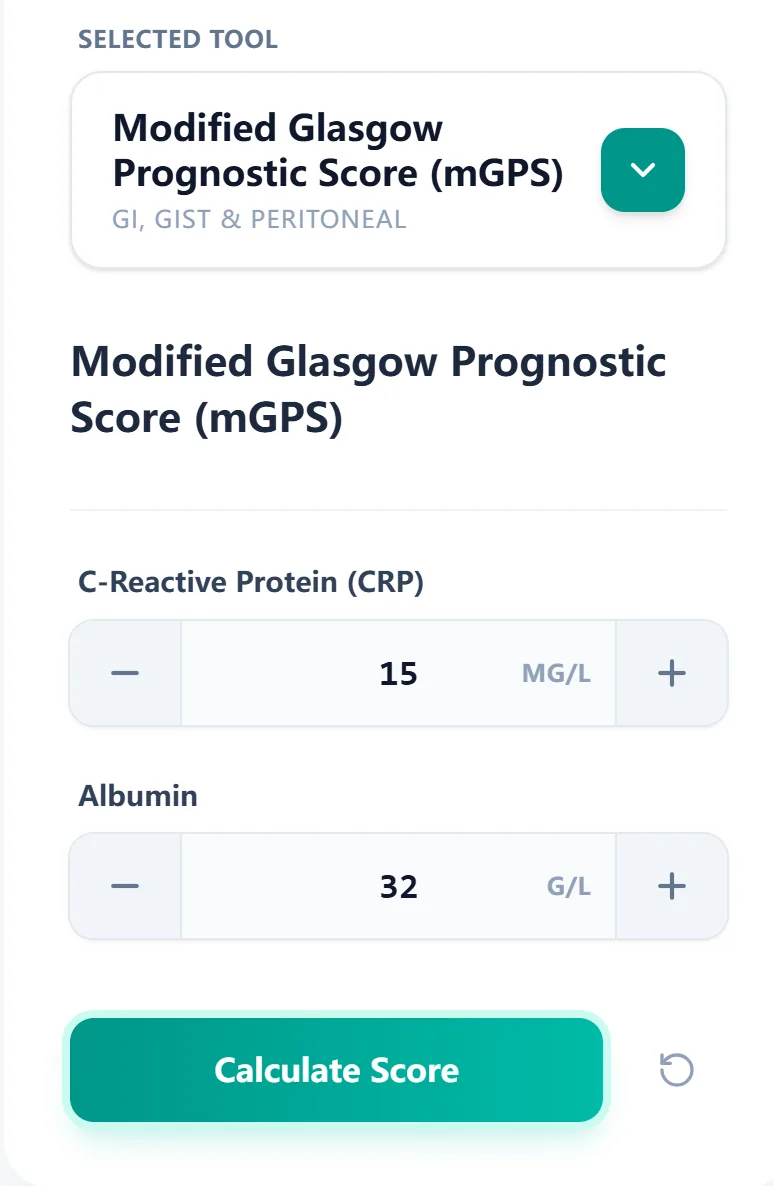
Figure 1. The input form focuses on CRP and albumin with clear units and step controls, allowing oncologists and surgeons to generate an mGPS score from routine labs in seconds.
Once the user clicks Calculate Score, the algorithm maps these inputs to the established mGPS categories:
- Score 0: CRP ≤10 mg/L.
- Score 1: CRP >10 mg/L and albumin >=35 g/L.
- Score 2: CRP >10 mg/L and albumin <35 g/L. [4], [1]

Figure 2. The results view highlights the mGPS category and provides plain-language interpretation, supporting shared decision-making and rapid documentation in clinic letters and MDT notes.
A reference table directly below the result summarizes how each score is defined and what prognosis it typically implies:
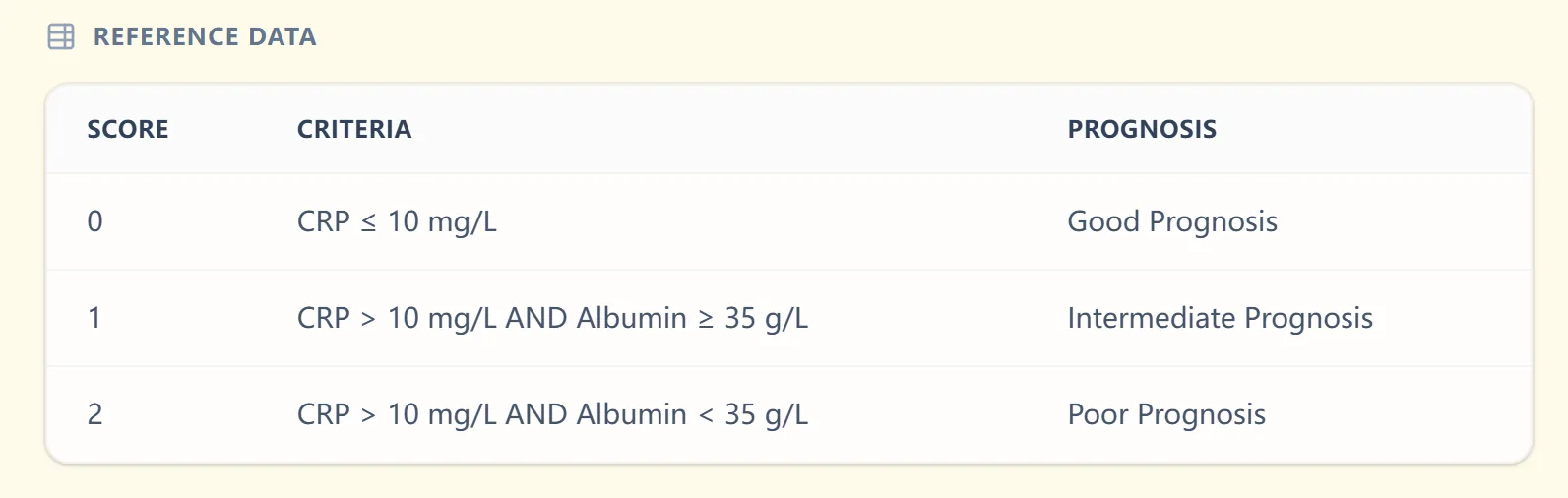
Figure 3. The reference table reinforces the scoring logic (CRP and albumin thresholds) and aligns each mGPS category with good, intermediate, or poor prognosis, helping trainees internalize the system.
The “Clinical Context & Background” pane provides a succinct explanation of the systemic inflammation-based Glasgow scores, citing core reviews and highlighting that mGPS reflects both inflammation (CRP) and nutritional decline (albumin). [2], [1]
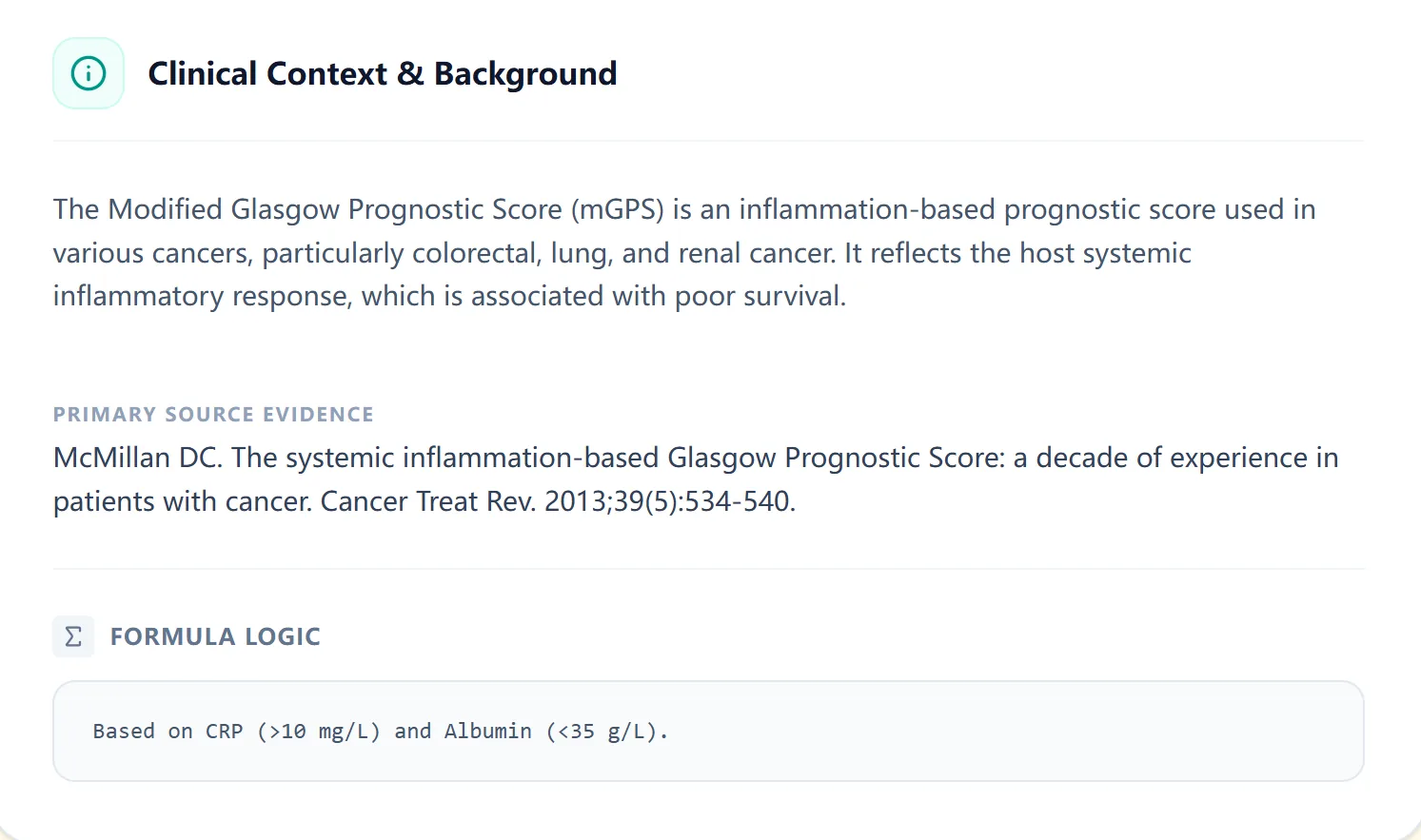
Figure 4. The background view synthesizes the decade-long experience with the Glasgow Prognostic Scores, giving users confidence in how the mGPS has been validated across cancers.
6. Specific Clinical Use Cases for the OncoToolkit Platform
At OncoToolkit, we envisage the mGPS calculator being woven into multiple real‑world workflows:
- Preoperative assessment for colorectal and gastric surgery: Use mGPS to identify patients at higher risk of postoperative complications and mortality, prompting targeted prehabilitation, nutritional optimization, and closer postoperative surveillance. [14], [8], [6]
- Neoadjuvant and adjuvant planning in rectal and pancreatic cancer: Combine mGPS with TNM and performance status when counseling patients about the risks and benefits of chemoradiotherapy or major resections; high mGPS may suggest need for intensive supportive care or closer toxicity monitoring. [5], [6]
- Prognostication in metastatic disease: In metastatic gastric, colorectal, or breast cancer, mGPS supports discussions about expected survival and treatment intensity, alongside ECOG and comorbidity burden, particularly when considering aggressive lines of chemotherapy versus earlier focus on symptom control. [9], [2], [1]
- Serial monitoring of systemic inflammation: Repeat mGPS calculations over time to track response to therapy or supportive interventions; falling scores may reflect improved inflammation or nutritional status, while rising scores can flag deterioration even before imaging changes emerge. [10], [2]
- Research, audit, and QI: Because our calculator standardizes thresholds and categories, clinicians can more easily stratify cohorts by mGPS for retrospective analyses, prospective trials, or quality-improvement projects evaluating perioperative pathways and supportive care models. [3], [12], [2]
Within the platform, we can link from the mGPS calculator to related tools such as a Prognostic Nutritional Index (PNI) calculator, a CRP/albumin ratio calculator, and a TNM staging tool, creating a comprehensive inflammation-and-stage–based prognostic workspace. [15], [16], [17]
7. How the Platform Supports Both Clinical Care and Research
OncoToolkit is designed to minimize cognitive load while enabling sophisticated use of prognostic scores:
- Clinical care:
- Single-screen input and output allow mGPS calculation in busy clinics, theatre briefings, or MDTs without switching systems or manually converting units.
- Visual risk bars and standardized interpretive text reduce variability in how scores are communicated across clinicians and to patients.
- Education and simulation:
- Trainees can adjust CRP and albumin to see how the mGPS changes and discuss how inflammation and nutrition affect surgical outcomes.
- The background panel acts as a micro–journal club summary, pointing trainees to key concepts without requiring them to sift through multiple papers.
- Research integration:
- With appropriate data governance, mGPS outputs recorded via the platform can be linked to local outcomes registries and analytics dashboards, enabling survival curves and complication rates stratified by mGPS or HS‑mGPS research labels. [12], [3], [10]
- This makes it easier to compare local practice with published Western and Asian data and to design trials targeting high mGPS populations with intensified supportive or anti-inflammatory strategies.
8. Clinical FAQ: Frequently Asked Questions about mGPS
How is the OncoToolkit mGPS calculator used in everyday practice?
On our platform, clinicians typically open the mGPS calculator alongside electronic lab results, enter CRP and albumin, and obtain a categorized prognosis within seconds. The result is then pasted directly into notes or tumor board summaries, ensuring that mGPS is documented consistently.
Is the mGPS equally valid in Western and Asian cancer populations?
Yes. Systematic reviews show that elevated mGPS predicts worse survival and higher complication rates in Western colorectal, renal cell, and rectal cancer cohorts, as well as Asian gastric, pancreatic, and head and neck cancer cohorts. Meta-analytic data support similar hazard ratios across regions. [7], [6], [3], [5], [10]
Are there guidelines that recommend using mGPS?
Disease-specific guidelines rarely mandate mGPS, but expert reviews on systemic inflammation in cancer describe GPS/mGPS as the most extensively validated inflammation-based prognostic score and recommend its use as a routine clinical assessment where available. [2], [1], [12]
What recent updates or expansions should clinicians know about?
Clinicians should be aware of HS‑mGPS, which uses a lower CRP threshold (3 mg/L) and may outperform conventional mGPS in predicting survival. Additionally, multiple studies have embedded mGPS into composite scores with nutritional indices or tumor markers. [11], [6], [3], [5], [10], [7]
When should you not rely on mGPS alone to make decisions?
mGPS should not be the sole determinant of surgery or systemic therapy when acute infection, autoimmune flare, liver failure, or nephrotic states drive CRP and albumin changes. It must be interpreted alongside stage, performance status, comorbidities, and patient preferences. [15], [10], [2]
9. Call to Action: Integrating mGPS into Your Practice
To bring inflammation-based prognostication into your GI oncology practice, open the Modified Glasgow Prognostic Score calculator and bookmark it on your workstation and mobile device. Try it with recent colorectal, gastric, pancreatic, or metastatic oncology cases to see how simple lab values translate into meaningful prognostic categories. [18], [19]
As you adopt the calculator in MDTs and clinics, consider pairing it with related OncoToolkit tools—such as PNI, CRP/albumin ratio, and TNM stage calculators—to build a consistent, low-friction prognostic workflow that enhances decision-making without adding to calculator fatigue. 20, 21, 22, 23
Ready to Simplify Your Prognostic Assessments?
Calculate the mGPS instantly and integrate systemic inflammation into your clinical decision-making workflow.
Launch mGPS Calculator
Free to use. No registration required.
10. References
- McMillan DC. The systemic inflammation-based Glasgow Prognostic Score: A decade of experience. Cancer Treat Rev. 2013. Source
- Proctor MJ, et al. A comparison of inflammation-based prognostic scores in patients with cancer. Eur J Cancer. 2011. Source
- Inoue A, et al. Modified Glasgow Prognostic Score is a useful predictor of outcomes. BMC Cancer. 2019. Source
- Pathway Medical. Modified Glasgow Prognostic Score (mGPS). Source
- Fan Z, et al. The prognostic value of the Glasgow Prognostic Score in pancreatic cancer. Sci Rep. 2020. Source
- Geng Y, et al. Predictive value of mGPS in colorectal cancer surgery. Front Oncol. 2023. Source
- Takeno S, et al. High-sensitivity modified Glasgow Prognostic Score (HS-mGPS). J Cancer. 2019. Source
- Clinics in Surgery. Predictive value of mGPS in elective colorectal surgery. 2019. Source
- Breast Cancer Research. mGPS in metastatic breast cancer. 2022. Source
- Meta-analysis of HS-mGPS. Int J Surg. 2023. Source
- Translational Cancer Research. HS-mGPS in Asian cohorts. 2023. Source
- Cancer France Bulletin. Systemic inflammation-based Glasgow Prognostic Score. Source
- Hepatocellular carcinoma markers. BMC Gastroenterol. 2013. Source
- Prognostic importance in Gastric Cancer. Cancer France. Source
- Nutritional Index and Cancer. Nutrients. 2015. Source
- CRP/Albumin Ratio study. OncoTargets Ther. 2019. Source
- TNM Staging and Inflammation. J Cancer. 2019. Source
- OncoToolkit mGPS Tool. Source
- OncoToolkit Home. Source
- Frontiers in Oncology. mGPS Updates 2025. Source
- Systemic Inflammation Markers. PMC. Source
- ScienceDirect. Cancer Prognosis Research. Source
- Semantic Scholar. mGPS Analysis. Source