Milan Criteria Calculator for HCC Liver Transplant: A Practical Guide for Oncologists
Hepatocellular carcinoma (HCC) in the setting of cirrhosis presents one of the most challenging decision-making problems in hepatology and oncology. The Milan Criteria remain the global benchmark for selecting patients for liver transplantation.

Quick Navigation
1. Introduction
Hepatocellular carcinoma (HCC) in the setting of cirrhosis presents one of the most challenging decision-making problems in hepatology and oncology: when is liver transplantation justified, and for which patients will scarce grafts deliver the greatest survival benefit? Clinicians must balance tumor burden, underlying liver function, waiting-list dynamics, and evolving selection criteria—often under time pressure during multidisciplinary tumor boards and bedside consults.1, 2
The Milan Criteria remain the global benchmark for selecting patients with HCC and cirrhosis for liver transplantation, defining a subset with post-transplant survival comparable to non-malignant indications. On our platform at OncoToolkit, we have built a streamlined Milan Criteria calculator (/calculator/milan-liver-transplant) that translates the classic selection rules into a fast, mobile-responsive decision support tool designed for busy HCC oncologists and hepatologists.2, 3
2. What are the Milan Criteria?
Originally proposed by Mazzaferro and colleagues in 1996, the Milan Criteria specify that transplantation is appropriate for patients with a single HCC lesion no larger than 5 cm, or up to three lesions, each no larger than 3 cm, in the absence of macrovascular invasion or extrahepatic metastasis. These simple radiologic criteria were derived from a prospective cohort of cirrhotic patients undergoing liver transplantation and showed a 4‑year overall survival around 75% and recurrence-free survival above 80%, essentially matching outcomes for non-tumor indications.4, 3, 5, 2
Today, the Milan Criteria are embedded in most major liver transplant guidelines and allocation systems, including AASLD and EASL recommendations that endorse liver transplantation for early-stage HCC within these limits when curative resection or ablation is not feasible. Many organ-allocation schemes also grant standard exception points for patients with HCC meeting Milan Criteria, acknowledging their favorable post-transplant prognosis relative to non-Milan HCC.5, 6, 1, 2
3. Why the Milan Criteria Matter in Daily Practice
For HCC oncologists and hepatologists, the Milan Criteria do far more than categorize tumor size and number. They:
- Guide referral timing for transplant evaluation and listing. Patients just within criteria may need expedited workup, while those approaching the limits may require bridging therapy to prevent progression beyond eligibility.1, 2
- Inform choice between resection, ablation, transplant, or palliative systemic therapy by anchoring discussions in expected post-transplant survival and recurrence risk.3, 1
- Shape waiting-list prioritization and exception MELD scoring, ensuring that patients with early HCC but preserved liver function still receive equitable access to grafts.6, 2
Without digital support, applying the Milan Criteria may sound trivial but can become error-prone in practice: clinicians juggle multiple imaging reports, evolving lesion measurements, and notes about vascular invasion or extrahepatic disease, often across different systems. Our Milan Criteria calculator is optimized for quick, reliable assessments during MDT meetings, telehealth case reviews, or ward rounds, reducing “calculator fatigue” and cognitive load when decisions need to be made in minutes rather than hours.
4. Clinical Evidence and Global Validation
4.1 Core evidence behind the criteria
The original Mazzaferro study demonstrated that strict adherence to the criteria produced 4‑year overall survival of about 75% and recurrence-free survival above 80%, a dramatic improvement over earlier series where broader inclusion yielded roughly 30% 5‑year survival. Subsequent analyses over more than 15 years confirmed that patients within Milan Criteria consistently achieve 5‑year post-transplant survival rates around 65–75%, with low recurrence rates, across multiple centers.7, 2, 3, 5
The logic is binary rather than probabilistic: if tumor burden is within size-number limits and there is no macrovascular invasion or metastasis, the patient is considered “within criteria”; otherwise, they fall “outside criteria” with a markedly higher recurrence risk and worse long-term survival after transplantation.4, 2, 3
4.2 Western vs Asian validation data
The Milan Criteria have been validated across both Western and Asian transplant programs, though case mix and expansion efforts differ by region.
Western cohorts (Europe, North America) Generally confirm 5‑year survival of 65–75% for patients within Milan Criteria, with survival approximating non-HCC transplant indications.2, 7, 5
Asian registries (China, Korea) Show that patients within Milan Criteria similarly achieve favorable long-term survival, while also exploring expanded criteria such as the Hangzhou or Asan criteria to accommodate local epidemiology and donor availability.8, 9, 10
For example, analyses of the China Liver Transplant Registry demonstrated that expanded criteria (Valencia, UCSF, Navarra, Hangzhou) could safely extend transplant eligibility beyond Milan in selected patients, with survival comparable to the Milan group when biological markers such as alpha‑fetoprotein (AFP) and tumor burden stratification were applied. Meanwhile, Western centers have focused on UCSF and Up‑to‑Seven expansions, confirming acceptable 5‑year outcomes in patients slightly beyond classical limits.11, 12, 13, 10, 5
5. Expansions and Updates to the Original Milan Criteria
Over the last two decades, multiple groups have proposed modifications to address concerns that Milan is overly restrictive. Key expansions include:
UCSF criteria: solitary tumor ≤6.5 cm, or up to three nodules each ≤4.5 cm with total tumor diameter ≤8 cm, associated with approximately 75% 5‑year survival—similar to Milan.11, 5
6. Guideline Context and Latest Advances
Modern liver transplant guidelines from professional societies such as AASLD, EASL, and APASL continue to:
- Endorse liver transplantation for patients within Milan Criteria when resection or ablation is not appropriate.5, 6, 1
- Recommend careful candidate selection for any “beyond Milan” protocols, often within clinical trials.3, 6, 1
- Emphasize dynamic assessment—serial imaging, treatment response, and waiting-list behavior.6, 1
7. How the OncoToolkit Milan Criteria Calculator Works
On our platform, the Milan Criteria calculator is designed for intuitive, rapid use on desktop, tablet, or smartphone. Clinicians enter only the clinically essential variables:
Figure 1. The input form captures only the key Milan variables—lesion count, maximum diameter, vascular invasion, and extrahepatic spread—allowing oncologists to obtain a result in seconds during MDT discussions or bedside consults.
After clicking “Calculate Score,” the tool internally applies the classic rule set—single lesion ≤5 cm or up to three lesions all ≤3 cm, no macrovascular invasion, and no metastasis—to categorize the patient as “Within Criteria” or “Outside Criteria.”
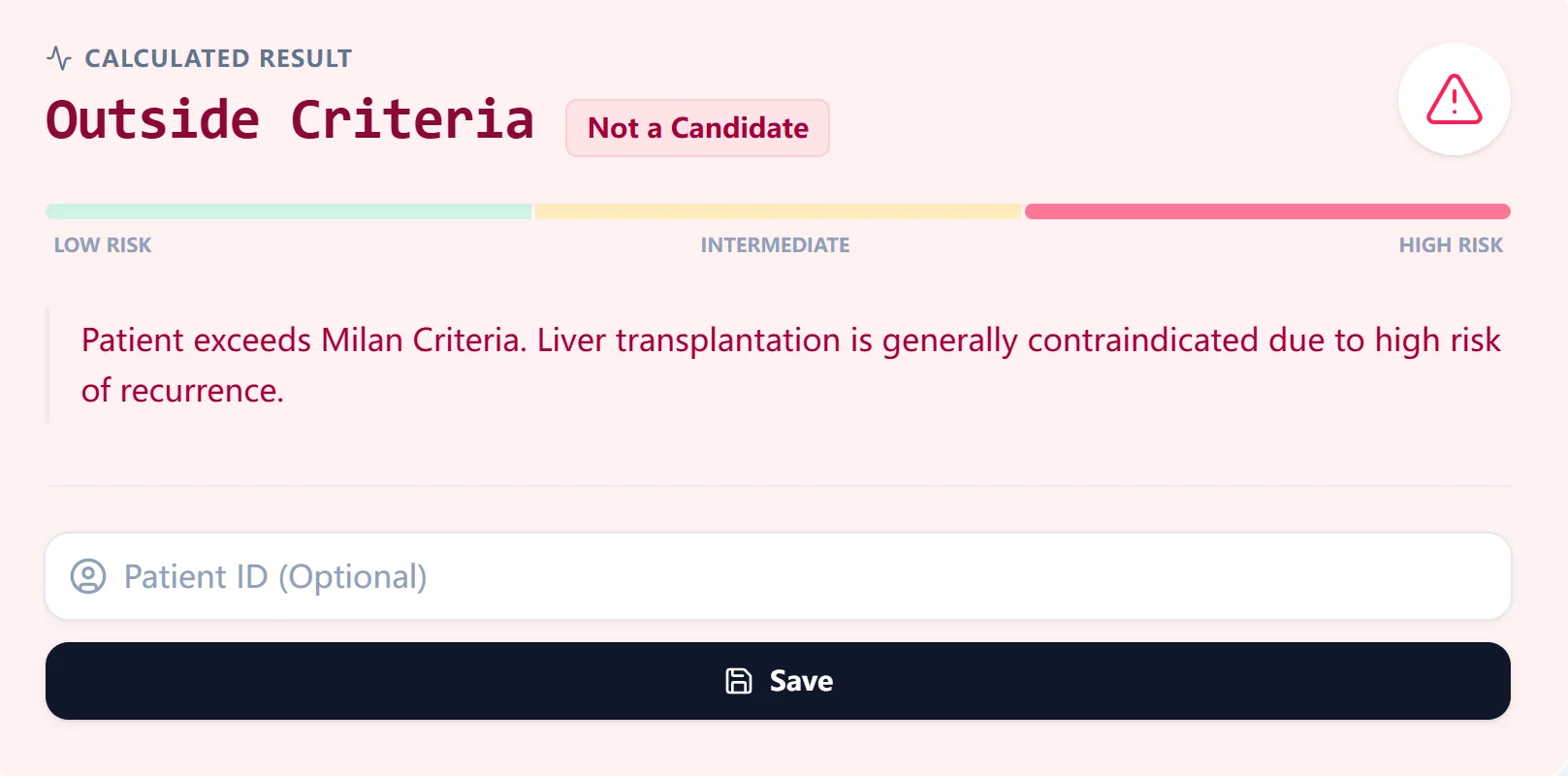
Figure 2. The result view labels the patient as within or outside Milan Criteria and provides plain-language interpretation to support shared decision-making and documentation.
7.1 Reference data and survival expectations
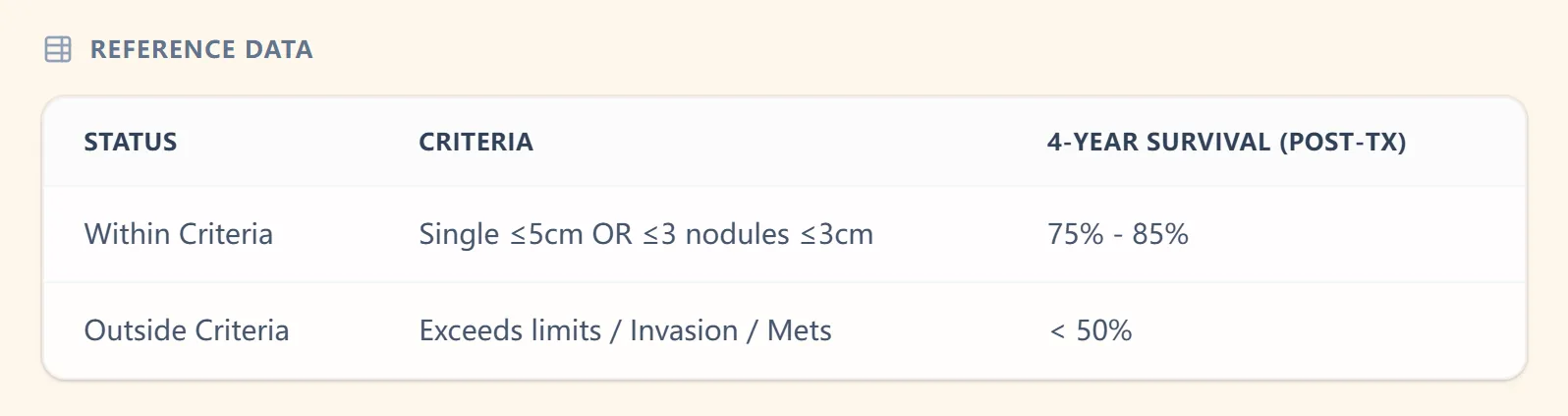
Figure 3. The reference table aligns Milan status with approximate 4‑year post-transplant survival, reinforcing why staying within criteria is critical when counseling patients and planning downstaging strategies.
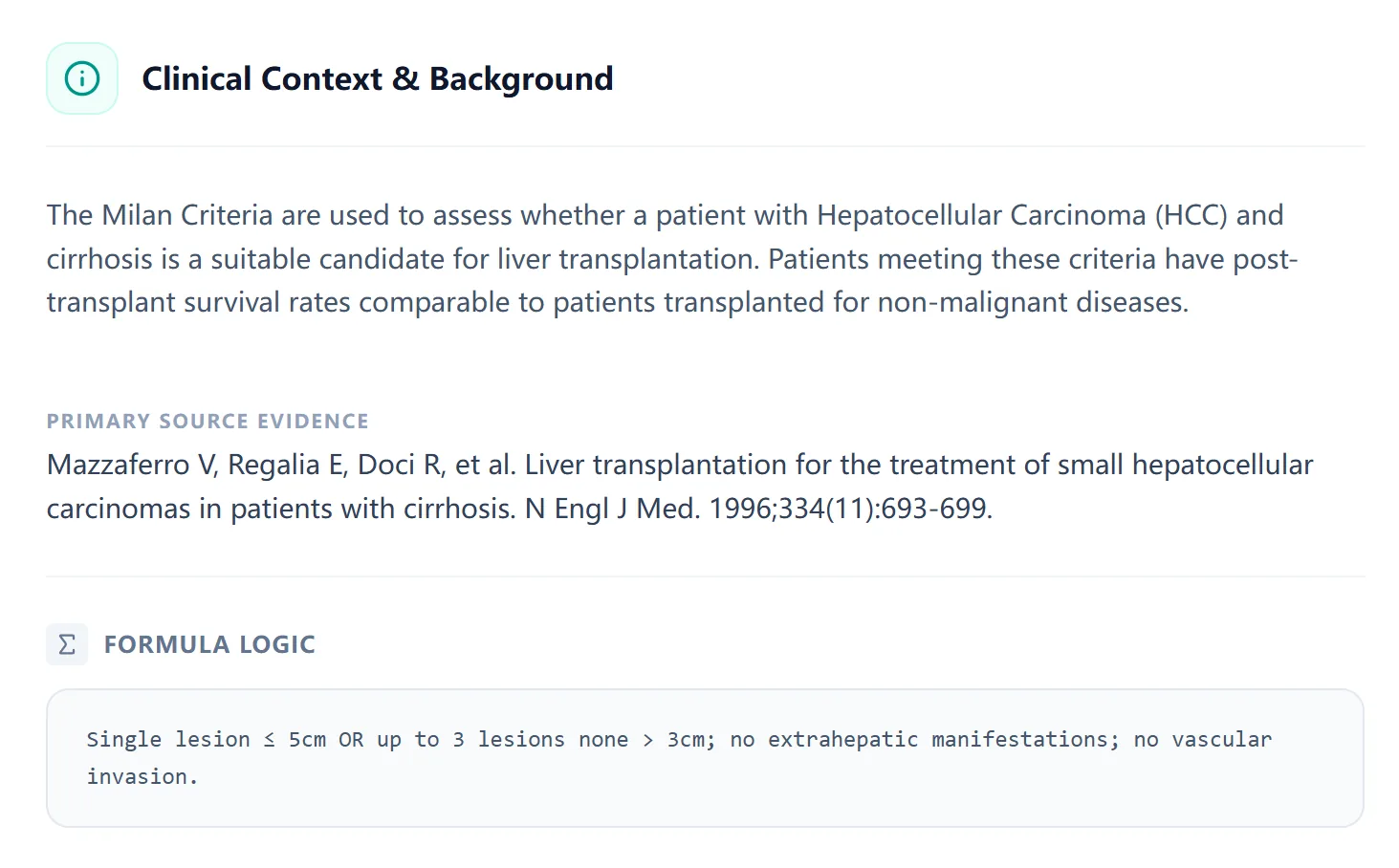
Figure 4. The background view summarizes evidence supporting the Milan Criteria and explicitly states the formula logic, helping users understand the assumptions behind each result.
8. Specific Use Cases for HCC Oncologists
At OncoToolkit, we have seen the Milan Criteria calculator used in several high-impact workflows:
- Initial staging and transplant referral: At first diagnosis, clinicians trigger early transplant referral when appropriate.
- Downstaging assessment: Repeated calculations after TACE or radioembolization help track successful downstaging.
- MDT preparation: Optimized for use on large screens so radiology, oncology, and surgery view the same structured assessment.
- Education: Fellows and residents use the calculator to practice classifying example cases.
- Clinical research: Standardizes Milan classification across services for retrospective QI audits.
9. How the Platform Supports Clinical Care and Research
OncoToolkit is built to support clinicians across the full decision cycle: from first consult to outcomes analysis. With the Milan Criteria calculator, this means:
- Routine decision support to complement staging systems such as BCLC.
- Education and simulation to show trainees how close some patients sit to thresholds.
- Research integration to benchmark performance against validated Western and Asian series.
10. Clinical FAQ
Can the Milan Criteria be used in patients with portal vein thrombosis or extrahepatic disease?
By definition, classic Milan Criteria exclude patients with macroscopic vascular invasion or extrahepatic metastases, as these features correlate with poor post-transplant survival.1, 2, 3
How does the Milan Criteria compare with UCSF and Up‑to‑Seven criteria?
Milan Criteria are more restrictive but well validated, whereas UCSF and Up‑to‑Seven allow larger tumors while maintaining acceptable survival.12, 10
What is the most common mistake when applying the Milan Criteria?
A frequent error is relying on outdated imaging or approximate measurements, which can misclassify patients who are borderline.7, 2
Ready to Simplify Your Transplant Selection?
Access our clinical-grade Milan Criteria calculator for fast, reliable decision support at the point of care.
Access the Milan Criteria Calculator
Free to use • No registration required