RMI I (Risk of Malignancy Index) Calculator for Ovarian and Adnexal Masses
A clinical guide to using the RMI I score for ovarian mass triage, including CA125 interpretation and ultrasound morphology scoring.
RMI I: Integrating morphology, status, and biomarkers for clinical triage.
Quick Navigation
1. Introduction to RMI I (Risk of Malignancy Index)
Ovarian and adnexal masses sit at the intersection of benign gynecology and gynecologic oncology, and misclassifying them has major consequences for patients and services. RMI I (Risk of Malignancy Index) remains a cornerstone of pre‑operative triage in many guidelines, providing a structured way to estimate malignancy risk using information that is already standard of care: CA125, menopausal status, and ultrasound morphology.1, 2, 3, 4
At OncoToolkit, we have implemented a strict, guideline‑aligned RMI I calculator designed for gynecologists, gynecologic oncologists, and trainees who need rapid, reproducible risk estimates at the point of care. Our tool enforces the correct RMI I definitions, maps scores to commonly used cut‑offs (including urgent MDT referral thresholds), and foregrounds the message that the index must always be interpreted within national and institutional clinical guidelines rather than replacing them.3, 4, 5, 6
2. What exactly is RMI I?
2.1 Historical development
The Risk of Malignancy Index was first described by Jacobs et al. in 1990 in a cohort of women undergoing laparotomy for adnexal masses. The authors showed that combining three variables—ultrasound features, menopausal status, and serum CA125—into a single score significantly improved discrimination between benign and malignant disease compared with CA125 alone or subjective assessment.2, 7
Subsequent work produced RMI II, III, and IV by adjusting the weighting of ultrasound and menopausal factors, but systematic reviews and guidelines have consistently found that RMI I is the best‑validated and most widely used iteration. NICE and RCOG therefore describe, and recommend, RMI I specifically in their ovarian cancer and adnexal mass guidance.4, 6, 7, 8, 3
2.2 Formal definition
RMI I is defined as:
RMI I = U * M * CA125
- U: ultrasound score based on defined suspicious features.
- M: menopausal status factor.
- CA125: serum cancer antigen 125 concentration in U/mL.
This simple multiplicative structure makes RMI I easy to implement in digital tools and transparent to explain to colleagues and trainees. OncoToolkit’s calculator displays the formula and each component explicitly so clinicians can see precisely how the final number was generated.2, 3
3. Dissecting each component in detail
3.1 Ultrasound score U – bringing morphology into a single number
Canonical suspicious features
RMI I focuses on five ultrasound features identified in the original and subsequent work as being associated with malignancy:7, 3, 4, 2
- Multilocular cyst.
- Solid areas.
- Bilateral lesions.
- Ascites.
- Intra‑abdominal metastases (e.g., omental cake, peritoneal nodules).
These features overlap substantially with descriptors used in IOTA, O‑RADS, and other structured ultrasound systems.9, 10, 11
Converting features to U
Instead of scoring each feature separately, RMI I compresses them into three levels:8, 3, 4
- No suspicious features → U = 0.
- One suspicious feature → U = 1.
- Two to five suspicious features → U = 3.
This non‑linear scoring reflects the steep escalation in malignancy risk when multiple high‑risk features co‑exist. In validation studies, U=3 is strongly associated with malignant histology, particularly in postmenopausal women.12, 7
Clinical Pearl: On our platform, you don’t have to recall these rules; you simply select “0 features”, “1 feature”, or “2–5 features” and the calculator assigns U = 0, 1, or 3 accordingly. Tooltip text can remind you of the underlying five features, which helps standardise interpretation of external ultrasound reports and supports teaching.13
Practical imaging examples
- Low‑risk morphology: unilocular anechoic cyst, smooth wall, no septations or solid nodules, no ascites → 0 features → U=0.14, 1
- Borderline morphology: multilocular cyst with thin septae but no solid parts or ascites → 1 feature (multilocularity) → U=1.3, 4
- High‑risk morphology: solid and cystic mass with papillary projections, bilateral involvement, and ascites → 2 or more features → U=3.10, 7
Guidelines emphasise that ultrasound should ideally be performed by experienced operators using structured reporting systems (IOTA, O‑RADS), and RMI I should be interpreted alongside this more detailed information.11, 9, 1, 10
3.2 Menopausal status M – encoding baseline risk
Menopause is a strong modifier of ovarian cancer risk; incidence rises significantly after menopause, and the pretest probability of malignancy in an adnexal mass is therefore much higher. RMI I incorporates this using a simple binary factor:1, 4, 8, 3
- Premenopausal → M = 1.
- Postmenopausal → M = 3.
RCOG provides detailed operational definitions: postmenopausal is 1 year or more of amenorrhoea, or age 50 or over after hysterectomy; all others are considered premenopausal unless there is evidence of ovarian failure.4, 8
OncoToolkit’s calculator mirrors these definitions and labels the options “Premenopausal (M=1)” and “Post‑menopausal (M=3)”, reinforcing how the factor is applied and helping trainees internalise guideline definitions.13, 4
3.3 CA125 – useful, but far from perfect
Role in RMI I
CA125 is a well‑known tumor marker used in ovarian cancer detection and monitoring, but as numerous reviews stress, it is neither specific nor sensitive enough to stand alone. RMI I uses the actual numeric CA125 value (U/mL) as a proportional contributor to the overall risk score, magnified by U and M.15, 16, 17, 2, 3, 4
On OncoToolkit, you enter the lab result directly; there is no need to normalise or cap values. Extremely high CA125 levels in a postmenopausal patient with U=3 will drive the RMI I into very high territory, aligning with guideline emphasis on urgent referral in such cases.5, 6, 3, 4
Causes of false positives and negatives
Elevated CA125 is common in a range of benign and non‑gynecologic conditions:16, 17, 18, 15, 4
- Endometriosis and adenomyosis.
- Uterine fibroids.
- Pelvic inflammatory disease.
- Liver disease, heart failure, and non‑malignant ascites.
- Physologic states such as menstruation and pregnancy.
Conversely, CA125 can be normal in:17, 1, 4
- Early‑stage epithelial ovarian cancers.
- Borderline and mucinous tumors.
- Germ‑cell and sex‑cord stromal tumors.
Reviews of CA125 emphasise that clinicians must never interpret CA125 (and therefore RMI) out of context; both NICE and RCOG state this explicitly. OncoToolkit’s clinical background section repeats this caveat and prompts users to follow their guidelines when deciding how heavily to weigh CA125, especially in premenopausal women and patients with known benign conditions.6, 17, 4
4. Risk cut‑offs and how guidelines use RMI I
4.1 NICE, RCOG, and local pathways
The NICE ovarian cancer guideline (CG122) includes an appendix that specifies RMI I and originally used 250 or more as the threshold for referral to a specialist MDT at a cancer centre. Subsequent surveillance work for NICE suggested that a lower cut‑off of 200 maintained acceptable specificity while improving sensitivity, and many UK cancer network pathways now treat RMI I 200 or more as high risk.19, 5, 6, 3
RCOG’s Green‑top Guideline 62 similarly concludes that RMI I (and II) are the only indices sufficiently validated for routine use and states that RMI I should be calculated to guide management decisions, particularly referring women with suspected ovarian cancer to a specialist centre.8, 4
4.2 OncoToolkit’s risk bands
To stay compatible with both the published data and your UI, our RMI I calculator uses three primary bands:
| RMI I score | Approximate malignancy risk (literature) | Guideline‑style interpretation |
|---|---|---|
| < 25 | Very low; malignancy often < 3–5% at this level.7, 18, 12 | Usually suitable for conservative management or follow‑up if consistent with guideline.14, 4, 1 |
| 25–199 | Intermediate; PPV around 15–25%, NPV high.20, 7, 12 | Requires further evaluation: repeat/expert ultrasound or cross‑sectional imaging.4, 8, 1 |
| ≥200 | High risk; PPV often > 70–75%, specificity > 80–90%.20, 7, 18, 12 | Urgent referral to a gynecologic oncology MDT.3, 5, 4, 6 |
OncoToolkit’s result text makes this linkage explicit: high scores are described as “highly suggestive of malignancy” and associated with urgent referral to gynecologic oncology, but always anchored with language such as “as per your national and institutional guideline”.6, 13
5. Evidence base – looking beyond simple sensitivity and specificity
5.1 Prospective and retrospective validations
Multiple prospective and retrospective studies across diverse settings have evaluated RMI I and related variants:18, 20, 7, 12
- In a study of 126 women, RMI I at a cut‑off of 200 achieved sensitivity around 77%, specificity around 89%, and an AUC around 0.86 for distinguishing benign from malignant adnexal masses.20
- A larger series reported that at a cut‑off of 200, RMI achieved sensitivity 70.5% and specificity 87.8%; lowering the cut‑off to 25 increased sensitivity but reduced specificity markedly, while raising it to 1000 produced excellent specificity but unacceptable sensitivity.7
- Prospective cohorts from various regions (including the Middle East and South Asia) have demonstrated similar performance, though some have proposed local cut‑offs (e.g., 150 or 265) to better match regional case‑mix and resource considerations.21, 12, 18
Overall, RMI I tends to perform best as a triage tool for deciding who should be managed in a cancer centre versus a general unit, rather than as a definitive diagnostic test. This is exactly how guidelines recommend using it and how we frame it in the OncoToolkit interface.1, 4
5.2 Head‑to‑head comparisons with modern models
Recent research has compared RMI with ultrasound‑based and multivariate models:
- External validation of the IOTA ADNEX model showed ADNEX outperforming RMI on AUC and sensitivity, particularly when applied by experienced sonographers.22, 23, 24
- A prospective comparative trial found that IOTA Simple Rules had sensitivity 90.9% and specificity 95.1% (AUC 0.93) in conclusive cases, whereas RMI2 at cut‑off 200 had sensitivity 72.7% and specificity 97.8% (AUC 0.85), highlighting RMI’s higher specificity but lower sensitivity.25
- Another study comparing IOTA Simple Rules, ADNEX, and RMI reported that IOTA‑based approaches significantly reduced false negatives, supporting recommendations that they be first‑line in centres where they can be implemented.26, 27, 28, 29
Consensus statements from ESGO/ISUOG/IOTA/ESGE and O‑RADS working groups similarly recommend structured ultrasound models as preferred tools where feasible, with RMI remaining an acceptable adjunct or fallback. OncoToolkit aligns with this by providing RMI I, O‑RADS‑aligned calculators, and (where relevant) ROMA in one place rather than positioning RMI as the only option.30, 31, 32, 9, 10, 11
6. How OncoToolkit’s RMI I calculator supports real‑world workflows
6.1 Interface and calculation
The RMI calculator page is structured to match how clinicians think through the case: menopausal status, CA125, ultrasound features, then overall risk.

Figure 1. The in‑app reference table maps RMI I bands to approximate malignancy probabilities and guideline‑style management actions.
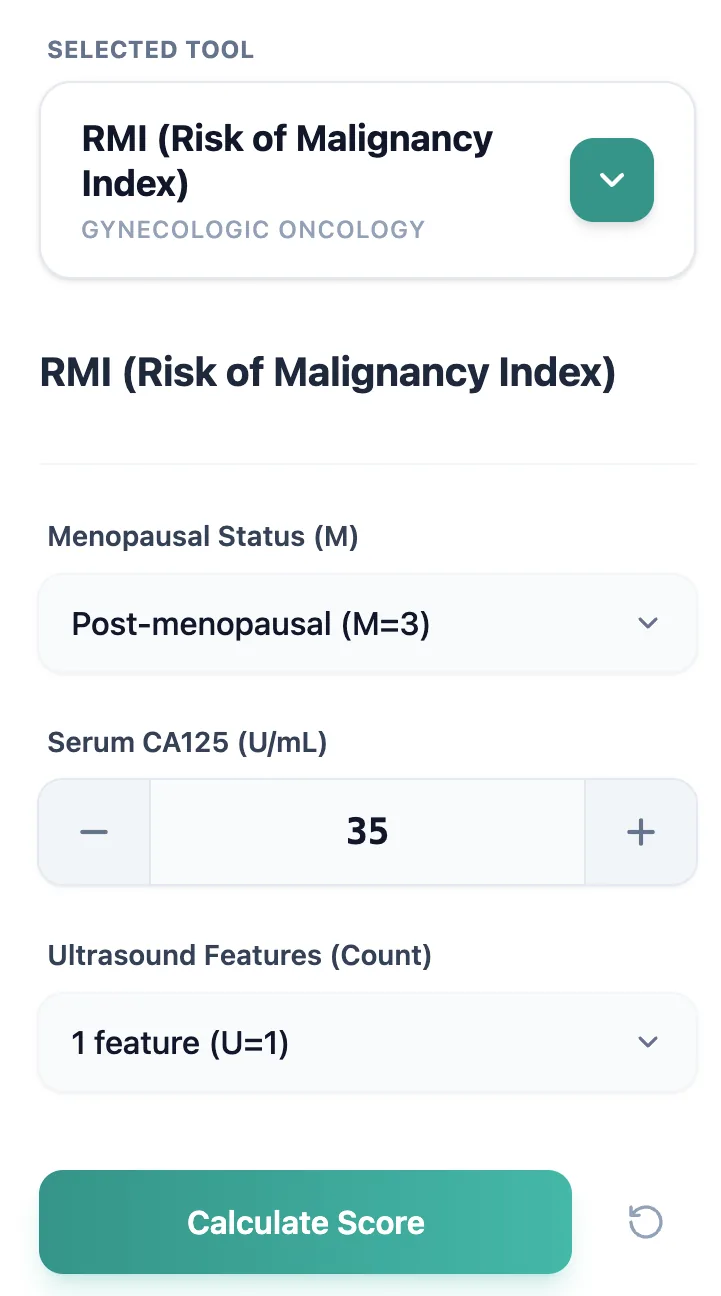
Figure 2. The input form reflects the RMI I definition—M, CA125, U—displaying multipliers explicitly.
- Choose menopausal status (M).
- Enter CA125 (U/mL).
- Select ultrasound feature count (U).
- Click “Calculate score”.
- The calculator computes U x M x CA125 and assigns the result to low, intermediate, or high risk according to the bands above.
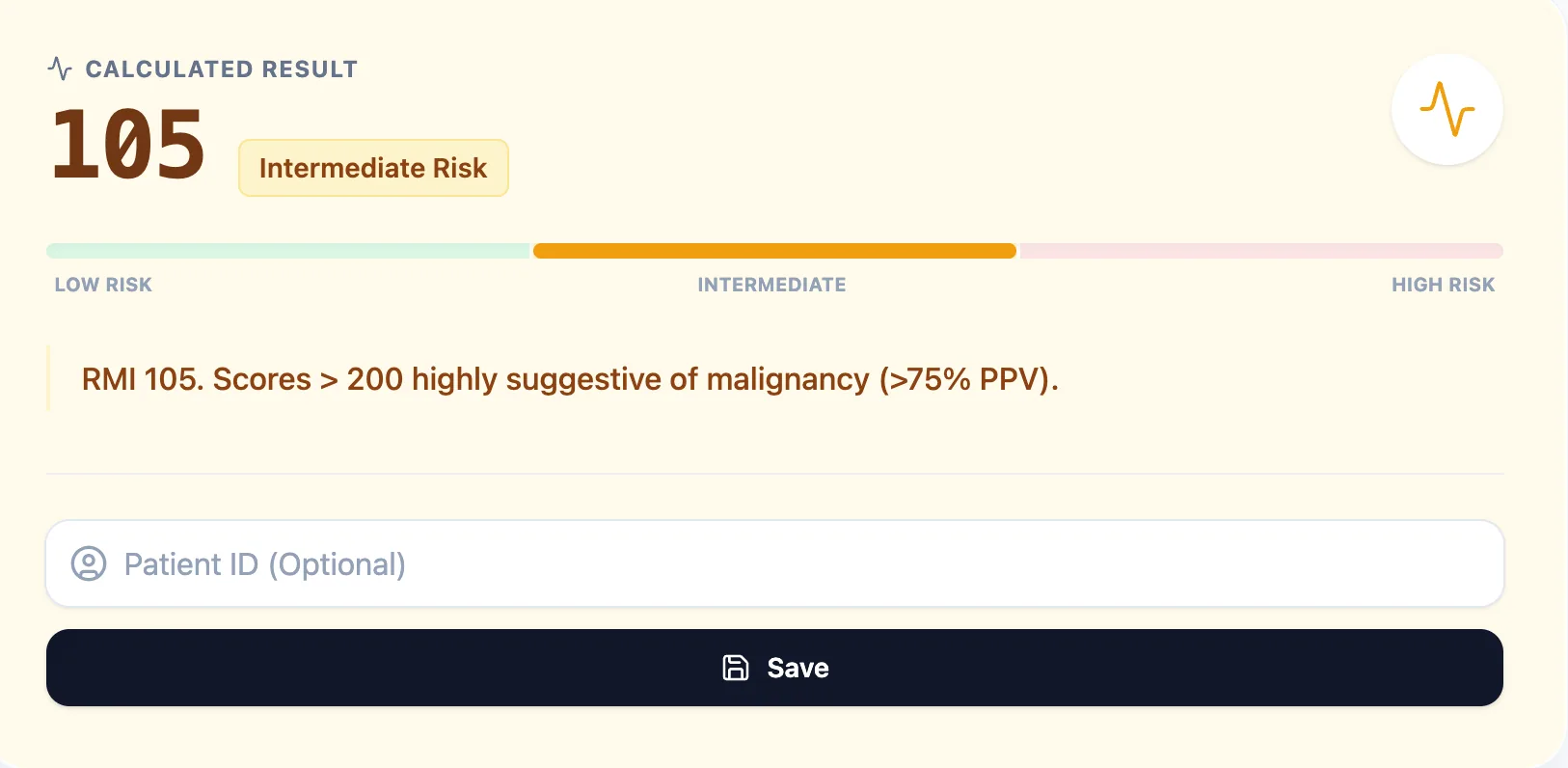
Figure 3. The result panel displays the numeric RMI I, a colour‑coded risk band, and interpretive text.
- Interpret and document.
- The output provides:
Figure 4. The “Clinical Context & Background” section cites the Jacobs derivation work and outlines RMI I’s role in pre‑operative triage.
6.2 Example documentation lines
- “RMI I = 540 (U=3, M=3, CA125=60 U/mL) – high‑risk; meets criteria for urgent referral to gynecologic oncology MDT as per [institution] ovarian cancer pathway.”5, 4
- “RMI I = 120 (U=1, M=1, CA125=120 U/mL) – intermediate risk; case to be discussed at gynae MDT; further imaging requested in line with guideline.”1, 4
The ability to export or copy these details supports clinical governance, audit, and quality‑improvement work, such as tracking how often patients meeting RMI‑based referral criteria actually reach a gynecologic oncology MDT within the recommended timeframe.35, 33, 5
7. Education, audit, and research
Education and simulation
For residents and fellows, RMI I is a natural bridge between basic teaching (“CA125 and ultrasound”) and more advanced topics (IOTA/ADNEX, O‑RADS, ROMA). OncoToolkit’s calculator supports several educational use‑cases:
- Case‑based learning: supervisors can present anonymised scenarios and ask trainees to predict RMI I and consequent guideline steps before revealing the calculated score.
- Understanding weighting: by manipulating U, M, and CA125 in the tool, trainees can see how postmenopausal status and multiple suspicious features dramatically amplify risk, reinforcing what guidelines say about high‑risk morphology in older women.4, 1
- Comparative reasoning: switching between RMI, ROMA, and O‑RADS calculators for the same case illustrates why guidelines increasingly favour structured ultrasound models and biomarker panels in some settings.29, 31, 11, 10, 9
Quality improvement and research
Because each RMI I calculation can be saved with a patient identifier and exported, the calculator also facilitates:
- Audit of referral adherence: comparing the proportion of patients with RMI I 200 or more who were actually referred to gynecologic oncology within the guideline‑recommended time.35, 5
- Local performance evaluation: linking exported RMI I data with histopathology and outcomes can show whether current cut‑offs are appropriate for your population.12, 18, 21
- Research into combined models: investigators can study how RMI performs when used sequentially with O‑RADS or contrast‑enhanced ultrasound.36, 26, 9
These projects still require formal ethics and governance approvals, but having structured, exportable RMI data from the outset reduces manual data‑entry burden.
8. Safety, limitations, and the primacy of clinical guidelines
All major guidelines emphasise that tools like RMI I are adjuncts, not replacements, for clinical assessment and multidisciplinary decision‑making. OncoToolkit reflects this in its language and design:37, 9, 6, 1, 4
- We state clearly that the calculator is intended for healthcare professionals as a decision‑support and educational resource, not for patient self‑use.30, 13
- We remind users that management decisions—including imaging, referral, surgery, and follow‑up—must always follow current national and institutional guidelines such as NICE, RCOG, ESGO/ESMO/ESGE, and local cancer network protocols.9, 37, 3, 6, 4
- We highlight the limitations of RMI I in premenopausal women, in non‑epithelial tumors, and where ultrasound quality is suboptimal, echoing the cautionary notes in RCOG, consensus statements, and contemporary reviews.9, 10, 1, 4
In other words, OncoToolkit’s RMI I calculator is designed to fit inside guideline pathways, not to redefine them.
Ready to Simplify Your Ovarian Risk Triage?
Calculate RMI I instantly and align your management with NICE and RCOG guidelines.
Open RMI I Calculator
Free to use. No registration required.
References
- Manganaro L, et al. Adnexal masses: RMI or O-RADS? Source
- Jacobs I, et al. A risk of malignancy index to assess ovarian masses. Source
- NICE. Risk of malignancy index (RMI I) Appendix. Source
- RCOG. Management of Suspected Ovarian Masses in Premenopausal Women (GTG 62). Source
- NIHR. Surveillance of NICE Ovarian Cancer Guidelines. Source
- NICE. Ovarian cancer: recognition and initial management (CG122). Source
- Ashrafganshouei T, et al. Validation of Risk of Malignancy Index. Source
- BSGE. Green-top Guideline 62 Appendix. Source
- IOTA/ESGO Consensus. Source
- O-RADS Ultrasound Committee Report. Source
- Timmerman D, et al. O-RADS v2022. Source
- Yavuzcan A, et al. Validation of RMI I in a Turkish population. Source
- OncoToolkit. RMI Ovarian Calculator. Source
- RACGP. Investigation of an ovarian mass. Source
- Biomarkers in Ovarian Cancer. Source
- CA125 False Positives Review. Source
- Bottoni P, et al. Cancer Antigen 125. Source
- Geomini P, et al. RMI performance study. Source
- NCBI Bookshelf. Ovarian Cancer Guidelines. Source
- Obeidat B, et al. RMI I Jordan Study. Source
- IJRCG. Regional RMI Analysis. Source
- ADNEX vs RMI Validation. Source
- Van Calster B, et al. ADNEX validation. Source
- External Validation of ADNEX. Source
- IOTA Simple Rules vs RMI. Source
- Eltawab, et al. Comparative Trial. Source
- Prospective comparison study. Source
- Diagnostic value study. Source
- IOTA vs RMI reduction of false negatives. Source
- OncoToolkit Portal. Source
- ROMA vs RMI Diagnostic Value. Source
- Multimodal triage evaluation. Source
- Care360 Data Export. Source
- Clinical Documentation Protocols. Source
- NHS England. Multi-Team Working. Source
- CEUS vs RMI Study. Source
- ESMO Practice Guidelines. Source
- MDT Audit Format. Source
- MDT Meetings Guide. Source
- S10 AI MDT Templates. Source
- HSE MDT Outcome Template. Source
- RMI Assessment Procedure. Source
- Adnexal Mass Management Review. Source
- RMI Database Facility Links. Source
- Mental Health Hybrid Workflow. Source