GPA Brain Metastases Calculator: Guidelines & Survival Estimates
Use the Graded Prognostic Assessment (GPA) calculator to estimate survival in brain metastases and align treatment with EANO-ESMO and NCCN guidelines.

1. Introduction to the GPA (Graded Prognostic Assessment) Calculator for Brain Metastases
Neuro-oncologists and radiation oncologists frequently care for patients with brain metastases whose expected survival can range from a few weeks to more than a year, even with similar imaging and primary tumor types. Treatment options—including stereotactic radiosurgery (SRS), whole-brain radiotherapy (WBRT), neurosurgery, systemic therapy, or best supportive care—require a nuanced understanding of prognosis to balance potential benefits with neurocognitive toxicity and resource use. At OncoToolkit, we’ve built a GPA (Graded Prognostic Assessment) calculator to translate a validated prognostic index into a fast, point-of-care tool that reduces cognitive load and “calculator fatigue” in busy neuro-oncology settings[1], [2], [3], [4], [5], [6], [7], [8], [9], [10], [11], [12], [13], [14], [15].
The calculator at /calculator/gpa-brain implements the original, histology-agnostic GPA for brain metastases, providing an overall score from 0 (worst prognosis) to 4 (best prognosis), with directly mapped median survival estimates. While diagnosis-specific GPA variants exist, this original GPA remains a practical starting point for many clinical scenarios, especially when diagnosis-specific or molecular information is incomplete or unavailable[16], [17], [2], [3], [4], [12], [13], [18], [1].
2. Understanding the GPA (Graded Prognostic Assessment) Score Components
The Graded Prognostic Assessment was developed from Radiation Therapy Oncology Group (RTOG) trial data as a more quantitative and user-friendly alternative to the earlier RPA classes for patients with brain metastases. The original GPA assigns points to four widely available clinical factors[2], [5], [1]:
- Age
- Karnofsky Performance Status (KPS)
- Number of intracranial (CNS) metastases
- Presence or absence of extracranial metastases
Each variable receives 0, 0.5, or 1.0 points, and the total GPA score is the sum of these four subscores, yielding a range from 0.0 to 4.0. Higher scores correspond to better prognosis, with clearly separated median survival times across score bands in the original derivation and validation cohorts[19], [20], [3], [4], [13], [18], [1], [2].
On the OncoToolkit calculator, these components are implemented exactly as described in the primary publication, and the interface explicitly notes that this is the original GPA, with separate diagnosis-specific GPA tools available on the platform for more granular applications[21], [12], [13], [16].
3. The Clinical Importance of the GPA Calculator in Neuro-Oncology
3.1 Prognosis-Driven Therapeutic Decisions in Brain Metastases
Clinical practice guidelines and consensus documents emphasize the importance of objective prognostic assessment when individualizing treatment for brain metastases. GPA score categories support decisions such as[6], [7], [8], [22]:
- Aggressive local therapy vs palliative approaches
- High GPA (for example, ≥3) with good performance status often justifies SRS or surgery plus systemic therapy, particularly when extracranial disease is controlled[3], [4], [10], [11], [18].
- Very low GPA (for example, ≤1) supports focusing on symptom control, short-course WBRT, or best supportive care when aligned with patient preferences[9], [18], [22], [6].
- Choice between SRS and WBRT or combined approaches
- Guidelines indicate that in patients with favorable prognosis and limited brain metastases, SRS alone or SRS plus systemic therapy can be preferred over upfront WBRT to reduce neurocognitive toxicity[7], [8], [10], [11], [6].
- Lower GPA scores with widespread intracranial and extracranial disease make WBRT or purely palliative regimens more appropriate[18], [22].
- Radiation dose and fractionation selection
Poor-prognosis GPA groups may receive short-course WBRT schedules, while better-prognosis groups might receive longer-course WBRT, SRS boost, or SRS alone[10], [11], [9].
By providing a numeric score and an associated median survival estimate at the point of care, the OncoToolkit GPA calculator makes it easier to integrate this prognostic framework into guideline-consistent decisions for individual patients[8], [12], [13], [7].
3.2 GPA Utility in MDT Discussions and Patient Triage
Multidisciplinary tumor boards often review multiple brain metastases cases in limited time, and structured prognostic information helps triage discussion depth and treatment planning intensity. Examples include[22], [19], [18]:
- Pre‑meeting triage by calculating GPA scores and flagging borderline or discordant cases.
- Displaying GPA alongside imaging and systemic staging to align perspectives between neurosurgeons, radiation oncologists, and medical oncologists.
- Documenting GPA in MDT summaries to make treatment rationales explicit and auditable.
The OncoToolkit calculator is optimized for rapid use during these sessions, with minimal inputs and a clear color-coded risk bar[12], [13].
3.3 Supporting Communication with Patients and Caregivers
Evidence-based prognostic tools such as GPA help clinicians provide realistic survival ranges while emphasizing individual variability. Numeric GPA categories and associated median survival bands can be translated into approximate ranges (“on the order of months rather than years”), supporting shared decision-making about treatment aggressiveness and goals of care. The calculator’s clear note that higher scores indicate better prognosis reinforces this without overstating precision[4], [13], [23], [19], [3], [12], [18], [22].
4. Clinical Evidence Base and Guideline Alignment for GPA
4.1 Derivation and Validation of the GPA Index
The original GPA index was derived from 1,960 patients enrolled in RTOG brain metastases trials, identifying age, KPS, number of brain metastases, and extracranial metastases as independent predictors of survival. These factors were weighted via Cox proportional hazards modeling and rescaled to the 0–4 GPA range. Subsequent work compared GPA with other indices such as RPA, BSBM, SIR, and GGS, showing that GPA offers at least comparable and often superior prognostic discrimination, especially when extended into diagnosis-specific variants[5], [24], [16], [1], [2], [18].
External validation in neurosurgical and radiotherapy cohorts confirmed that increasing GPA scores are associated with stepwise improvements in survival, though short‑term operative mortality is not perfectly predicted by GPA alone. These findings support using GPA as a backbone prognostic tool, supplemented by clinical judgment and additional risk factors when considering surgery or intensive systemic therapy[20], [25], [26], [19], [6], [22].
4.2 Evolutions: Diagnosis-Specific and Molecular GPA Variants
Diagnosis-Specific GPAs (DS‑GPA) were developed when it became clear that prognosis varies substantially by primary tumor type and tumor biology. DS‑GPA models adjust variable selection and weighting for primaries such as NSCLC, SCLC, breast cancer, melanoma, renal cell carcinoma, and GI cancers, sometimes incorporating tumor subtype or hormone receptor status[27], [28], [19], [3], [4].
Molecular GPA systems such as Lung‑molGPA incorporate EGFR, ALK, and other biomarkers into prognostic scoring for NSCLC brain metastases, improving survival discrimination in the targeted therapy era. Additional work has explored extracranial disease scores (EC‑S and EC‑GPA) that combine extracranial tumor burden, albumin, and LDH with DS‑GPA to better identify patients with very short survival who may not benefit from brain irradiation. Recent analyses continue to show that combining DS‑GPA with extracranial or molecular scores can refine prognostication even further[29], [30], [31], [32], [33], [16].
The 2020 updated DS‑GPA summary report aggregates modern diagnosis-specific indices and introduces the “eligibility quotient,” a concept for using prognosis to broaden clinical trial access without compromising interpretability of results. This reinforces the role of GPA-based tools in both routine care and research design[17], [16].
4.3 Major Guideline Recommendations (EANO, ESMO, NCCN)
Major guidelines and consensus statements confirm that GPA and DS‑GPA are appropriate tools to support clinical decision-making:
- EANO–ESMO 2021 guidelines recommend validated prognostic scores such as GPA/DS‑GPA for stratifying patients and individualizing treatment[7], [8].
- Earlier EANO guidance highlights the importance of performance status, extracranial disease control, and number of brain metastases—core GPA variables—when selecting SRS, WBRT, or best supportive care[6].
- NCCN CNS metastases materials emphasize using performance status and prognosis to guide radiotherapy choices and clinical trial eligibility, with GPA referenced as a practical framework[25], [9].
- Recent reviews confirm that GPA-based systems remain central among prognostic indices, with ongoing refinements rather than replacement[34], [19], [18].
5. Recent Expansions, Modifications, and Updates to the GPA System
Although the OncoToolkit calculator uses the original four-factor GPA, clinicians should be aware of several important extensions:
- Diagnosis-Specific GPA (DS‑GPA)Separate scoring for NSCLC, breast, melanoma, SCLC primaries[28], [3].
- Molecular GPAsIncorporate driver mutations and biomarkers for NSCLC and melanoma[31], [32].
- Extracranial DS‑GPA (EC‑GPA)Combine EC models to define very poor prognosis patients[30], [31].
- Specialized ModelsTailored to poor-prognosis WBRT or postoperative cohorts[35], [36].
OncoToolkit’s GPA page references these evolutions, and related calculators on the platform allow clinicians to transition from the simple original GPA to more complex models when detailed data are available[38], [13], [21], [16], [12].
6. Operationalizing Prognosis: How the OncoToolkit GPA Calculator Works
The OncoToolkit GPA (Graded Prognostic Assessment) calculator is intentionally minimalistic to support rapid, error‑resistant use. Clinicians enter four variables[13], [12]:
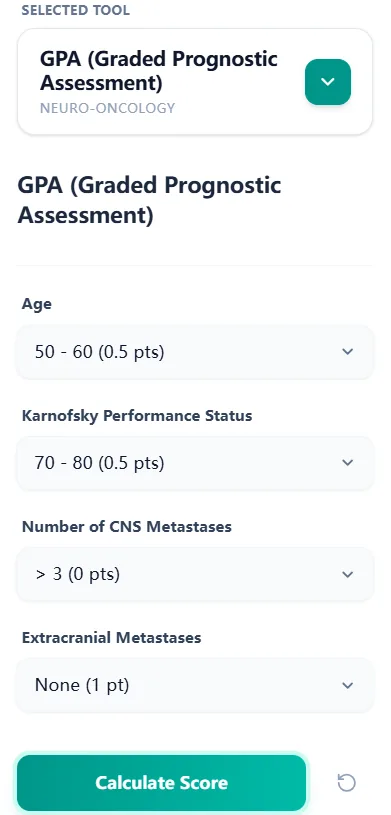
Figure 1. The GPA input form uses four dropdown fields—age, KPS, CNS metastasis count, and extracranial metastases—allowing neuro-oncologists to generate a score rapidly during clinics or MDT meetings.
Each dropdown option maps to a pre-defined subscore (0, 0.5, or 1.0). The calculator sums these subscores to generate the total GPA and then:
- • Displays the GPA score (0–4).
- • Places the score on a color-coded risk bar spanning low, intermediate, and high risk.
- • Shows an estimated median survival derived from RTOG-based survival bands.
- • Provides a brief interpretation stating that higher scores indicate better prognosis.
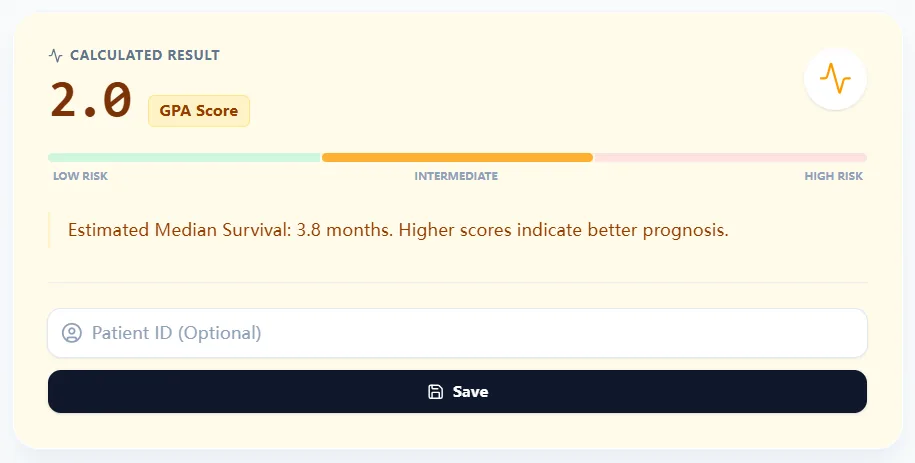
Figure 2. The result panel presents the GPA score, a risk bar, and median survival estimate, reinforcing that higher scores correlate with better prognosis and supporting communication with patients and colleagues.
A reference table below the calculator summarizes survival estimates for score ranges used on the platform:
| GPA Score | Median Survival (OncoToolkit) |
|---|---|
| 0–1.0 | 2.6 months |
| 1.5–2.5 | 3.8 months |
| 3.0 | 6.9 months |
| 3.5–4.0 | 11.0 months |
Figure 3. The GPA reference table maps score ranges to median survival estimates, helping clinicians translate the numeric index into intuitive prognostic categories for shared decision-making.
Clinicians may optionally enter a patient ID and save the result within OncoToolkit, enabling longitudinal tracking and retrospective analyses[12], [13].
7. Clinical Context, Transparency, and E-E-A-T Standards
To support transparency and E‑E‑A‑T standards, the GPA page includes a Clinical Context & Background section that explains:
- That GPA is a prognostic index for patients with brain metastases and is considered more quantitative and user-friendly than RPA[15].
- That the OncoToolkit tool implements the original GPA and notes the existence of diagnosis-specific and molecular GPA variants[14], [15].
- The basic formula logic—that the GPA equals the sum of four subscores (0, 0.5, 1.0) for age, KPS, CNS metastases, and extracranial metastases[11], [15].

Figure 4. The clinical background section outlines the rationale for GPA, cites primary research, and exposes the scoring logic so that technically inclined users can verify the calculation.
This approach is consistent with recommendations that prognostic tools used in oncology be transparent, guideline‑aligned, and accompanied by clear caveats about their role as decision support rather than stand‑alone decision makers[25], [8], [22], [7].
8. Clinical Case Studies and Applications of the GPA Calculator
8.1 Case 1: Newly Diagnosed Brain Metastases with Limited Lesions
A patient with NSCLC, age in the 50s, KPS 80, three brain metastases, and no extracranial disease receives an intermediate‑to‑good GPA score on the OncoToolkit calculator, with median survival in the range of several to many months[12], [14]. In line with EANO–ESMO and NCCN‑aligned guidance, this prognosis supports considering SRS or surgery plus systemic therapy as alternatives to routine WBRT, prioritizing local control and cognitive preservation[11], [8], [10], [6], [7].
8.2 Case 2: Multiple Brain Metastases and Extensive Extracranial Disease
An older patient with KPS below 70, more than three brain metastases, and progressive extracranial disease receives a low GPA, with median survival of only a few months. Guidelines support short-course WBRT or best supportive care for such patients, and GPA provides an evidence-backed reference during discussions about whether to pursue palliative radiotherapy or focus on symptom management alone[1], [2], [9], [18], [22], [6].
8.3 Case 3: Clinical Trial and Registry Enrollment
Trials in SRS, WBRT, systemic therapy, and neuroprotective strategies often require structured prognostic indices for eligibility and stratification. The OncoToolkit GPA score, saved with basic identifiers, allows sites to stratify participants and analyze outcomes by prognostic group in line with the eligibility quotient framework introduced in updated DS‑GPA work[29], [34], [17], [16].
8.4 Case 4: Quality Improvement and Outcomes Benchmarking
Institutions can use GPA data exported from OncoToolkit to benchmark survival, treatment intensity, and radiotherapy utilization across GPA groups, comparing local outcomes with published cohorts. Such analyses support service planning (for example, SRS resource allocation) and may reveal subgroups that could benefit from protocol changes or trial development[24], [26], [19], [18], [29].
9. How OncoToolkit Supports Clinical Care, Education, and Research
OncoToolkit is designed as an integrated ecosystem for clinical decision support, education, and research[14], [15], [13], [12].
- C
- E
- R
10. Frequently Asked Questions (FAQs) About the GPA Calculator
10.1 Limitations: When to Use Caution with Original GPA
The original GPA should not be the sole determinant of therapy in patients whose outcomes are strongly influenced by modern targeted or immunotherapies or by specific molecular features captured by Lung‑molGPA or other molecular indices. It is also less reliable in populations that differ substantially from the RTOG cohorts, reinforcing the need for local validation and integration with clinical judgment[39], [32], [33], [17], [22], [19], [31], [16].
10.2 GPA vs. RPA: Comparative Clinical Utility
RPA divides patients into three classes using age, KPS, and systemic disease status, but yields relatively broad categories and involves more subjective assessment of “controlled” systemic disease. GPA provides a 0–4 scale with simple numeric subscores and has been shown in comparative studies to offer at least equivalent and often superior discrimination, particularly in combination with diagnosis-specific variants[2], [3], [4], [5], [18], [24], [19], [1].
10.3 Using GPA in Comorbid or Frail Populations
Comorbidities and frailty are not explicit GPA components but typically manifest as lower KPS, which indirectly captures some of their prognostic impact. Guidelines recommend integrating GPA with broader geriatric and comorbidity assessments when considering aggressive local therapy in older or highly comorbid patients[8], [22], [25], [1], [2], [6], [7].
10.4 Population Validity: Western vs. Asian Cohorts
GPA and DS‑GPA have been validated in European and Asian cohorts, but several studies show differences in calibration, particularly where systemic therapies and supportive care differ from original datasets. The OncoToolkit calculator uses published coefficients and survival bands; clinicians should interpret results in context and consider contributing to local validation efforts[26], [30], [39], [29], [19], [31].
10.5 Reducing Medical Errors via Digital Calculators
Manual GPA calculation is prone to errors such as mis-estimating KPS, miscounting CNS metastases, or failing to update extracranial disease status when new imaging appears. The calculator mitigates these pitfalls by employing clearly defined dropdowns, explicit variable descriptions, an instant risk bar, and integrated reference tables, supporting safer, more consistent use at the point of care[34], [13], [26], [19], [12].
11. Conclusion: Integrating GPA into Your Neuro-Oncology Workflow
To incorporate GPA into routine practice, visit /calculator/gpa-brain, bookmark it on your MDT workstation and mobile device, and apply it to upcoming brain metastasis cases. Compare predictions with your current decision-making, and then extend use to trial design, audit projects, and training sessions by saving and aggregating scores within the platform[13], [12].
By embedding this evidence-based, guideline-concordant calculator into daily workflows—and pairing it with related diagnosis-specific and molecular tools—neuro-oncology teams can deliver more consistent, transparent, and patient-centered care for individuals facing brain metastases[17], [22], [16], [7], [8].
Ready to Simplify Your Prognostic Assessments?
Access our clinical-grade GPA calculator for immediate survival estimates at the point of care.
Open GPA Calculator
Free to use. No registration required.
References
- Sperduto PW, et al. A new prognostic index and comparison to RPA. Source
- Sperduto PW, et al. The Graded Prognostic Assessment (GPA). Source
- Sperduto PW, et al. Diagnosis-Specific Prognostic Factors and GPA. Source
- Sperduto PW, et al. Summary Report on the Graded Prognostic Assessment. Source
- Sperduto PW, et al. Decision support for brain metastases. Source
- Soffietti R, et al. EANO guidelines on brain metastases. Source
- EANO-ESMO. Clinical Practice Guidelines. Source
- Le Rhun E, et al. EANO-ESMO guidelines summary. Source
- Nabors LB, et al. NCCN Guidelines: CNS Metastases. Source
- Drappatz J, et al. SRS vs WBRT. Source
- Radiotherapy for brain metastases. Source
- OncoToolkit GPA Calculator. Source
- MD Anderson Clinical Calculators. Source
- OncoToolkit Homepage. Source
- About OncoToolkit. Source
- Sperduto PW, et al. The 2020 Updated DS-GPA. Source
- Sperduto PW, et al. Eligibility Quotient. Source
- Prognostic Indices in Brain Metastases Review. Source
- Validation of GPA. Source
- RTOG Validation Cohorts. Source
- BrainMetGPA.com. Source
- Clinical Utility of GPA in Practice. Source
- Communication and Prognosis. Source
- Comparative Studies of RPA and GPA. Source
- ASTRO Guidelines on Brain Metastases. Source
- External Validation Cohorts. Source
- Sperduto PW, et al. NSCLC DS-GPA. Source
- Breast Cancer DS-GPA. Source
- Modern Cohort Validation. Source
- Extracranial Disease Models. Source
- Sperduto PW, et al. Lung-molGPA. Source
- Molecular Markers in Melanoma GPA. Source
- EGFR/ALK and Prognosis. Source
- Review of GPA Evolution. Source
- Comparison of GPA with New Indices. Source
- Postoperative Prognostic Scoring. Source
- WBRT Specific Cohort Models. Source
- CancerCalc GPA Logic. Source
- Asian Population Validation. Source
- Aristot Index Pronostiques. Source
- Modern Radiotherapy Outcomes. Source