DLBCL Prognosis Panel Calculator — IPI & NCCN-IPI in One Tool
Calculate Classic IPI and NCCN-IPI for DLBCL instantly. Get risk group, 5-year OS, and treatment guidance in seconds. Try the free Lymphoma Prognosis Panel now.

1. Overview of the Lymphoma (DLBCL) Prognosis Panel: IPI & NCCN-IPI Made Simple
Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma worldwide, accounting for approximately 30–40% of all new adult NHL diagnoses. It is an aggressive malignancy with a heterogeneous clinical course: while roughly two-thirds of patients achieve durable remission with frontline rituximab-based chemoimmunotherapy, the remaining one-third will experience primary refractory disease or relapse — a scenario associated with substantially worse long-term outcomes. This clinical heterogeneity makes accurate prognostic stratification at the time of diagnosis one of the most consequential steps in the entire DLBCL care pathway. The score assigned in those first few days shapes the treatment plan, determines clinical trial eligibility, informs the conversation about expected outcomes, and guides decisions about CNS prophylaxis, radiation consolidation, and stem cell transplant planning.1, 2, 3, 4, 5, 6
For three decades, this stratification has rested on the International Prognostic Index (IPI) — a simple, five-variable, bedside-friendly scoring system that has become ubiquitous in lymphoma practice. More recently, the NCCN-IPI (Enhanced International Prognostic Index) has emerged as a refinement designed specifically for the rituximab era, offering wider separation between low-risk and high-risk patients. In modern practice, many clinicians want — or need — both scores simultaneously, whether for MDT presentations, tumor board documentation, or head-to-head comparison during patient counseling.7, 5
At OncoToolkit, we built the Lymphoma (DLBCL) Prognosis Panel to address exactly this need. Rather than switching between two different calculators and manually cross-referencing results, our unified panel computes the Classic IPI and the NCCN-IPI side by side from a single set of inputs — in seconds, on any device. You can try it now at /calculator/panel-lymphoma.
This article provides a comprehensive clinical and technical overview of both scoring systems, the evidence behind them, their limitations, and a detailed walkthrough of how our calculator works in day-to-day practice and research.
2. Understanding the International Prognostic Index (IPI)
2.1 Origin and Development
The International Prognostic Index was published in 1993 by the International Non-Hodgkin's Lymphoma Prognostic Factors Project. The landmark study analyzed more than 2,000 patients with aggressive non-Hodgkin lymphoma treated with anthracycline-containing (CHOP-like) regimens across 16 institutions and cooperative groups in the United States, Europe, and Canada. Using multivariable Cox proportional hazards regression, the investigators identified five clinical features that were independently and significantly associated with overall survival.5, 8, 9, 10
2.2 The Five IPI Factors
Each factor is scored as a simple binary variable (present = 1, absent = 0), for a maximum total of 5 points:
- Age > 60 years — a surrogate for decreased tolerance of intensive chemotherapy and increased competing causes of mortality.
- Ann Arbor Stage III or IV — reflecting advanced, disseminated disease.
- Serum LDH above the upper limit of normal (ULN) — a proxy for tumor burden and proliferative activity.
- ECOG Performance Status ≥ 2 — indicating functional impairment that may limit treatment delivery.
- More than one extranodal site of disease — reflecting widespread organ involvement.11, 8, 9
2.3 IPI Risk Groups and Survival Estimates
The total score (0–5) maps to four risk groups:
| Risk Group | IPI Score | 5-Year Overall Survival |
|---|---|---|
| Low | 0–1 | ~73% |
| Low-Intermediate | 2 | ~51% |
| High-Intermediate | 3 | ~43% |
| High | 4–5 | ~26% |
2.4 The Revised IPI (R-IPI)
In 2007, Sehn and colleagues from the British Columbia Cancer Agency re-evaluated the IPI in 1,063 patients with DLBCL treated with R-CHOP. They found that the same five variables remained predictive, but the distribution of survival across the original four tiers had compressed — particularly in the high-risk group, where 5-year OS had risen to approximately 50–55%. Rather than reworking the variables, the R-IPI simply re-grouped the original 0–5 scores into three tiers: Very Good (0, 94% 4-year OS), Good (1–2, 79%), and Poor (3–5, 55%). This was a pragmatic fix, but many clinicians felt it lost granularity — and it still could not identify a truly poor-prognosis subgroup with long-term survival below 50% in the rituximab era.5, 8
3. What Is the NCCN-IPI?
3.1 Development and Rationale
The NCCN-IPI was developed by Zhou, Sehn, Rademaker, Winter, and a multi-institutional team across seven National Comprehensive Cancer Network cancer centers in the United States, and was published in Blood in 2014. The investigators used raw clinical data from 1,650 adults with de novo DLBCL diagnosed between 2000 and 2010 — squarely within the rituximab era — and set out to build a new prognostic model using traditional clinical features alone, without requiring molecular or immunohistochemical markers.7, 5
The fundamental insight of the NCCN-IPI is that age and LDH are continuous variables with non-linear effects on survival, and that dichotomizing them at a single threshold (as the classic IPI does) throws away clinically important prognostic information. Additionally, the investigators found that the specific anatomic sites of extranodal disease were more prognostically relevant than simply counting the number of extranodal sites.5, 7
3.2 NCCN-IPI Scoring Components
The NCCN-IPI retains the same five clinical domains as the classic IPI but applies a more granular weighting to three of them. A maximum of 8 points can be assigned:
| Factor | Category | Points |
|---|---|---|
| Age | ≤ 40 years | 0 |
| > 40 to ≤ 60 years | 1 | |
| > 60 to ≤ 75 years | 2 | |
| > 75 years | 3 | |
| Serum LDH (normalized to ULN) | ≤ 1 × ULN | 0 |
| > 1 to ≤ 3 × ULN | 1 | |
| > 3 × ULN | 2 | |
| Ann Arbor Stage | I–II | 0 |
| III–IV | 1 | |
| ECOG Performance Status | 0–1 | 0 |
| ≥ 2 | 1 | |
| Extranodal Disease in Specific Sites | None of the specified sites | 0 |
| Bone marrow, CNS, liver/GI, or lung involved | 1 |
This scoring table is derived directly from the original study's multivariate model (Table 2 in Zhou et al.). Notably, the general "number of extranodal sites > 1" variable from the classic IPI was not independently significant in the NCCN-IPI multivariate analysis (HR 1.0, 95% CI 0.8–1.3, P = 0.91). Instead, involvement of specific high-risk organs (bone marrow, CNS, liver/GI tract, or lung) was a far stronger predictor of poor outcome.5
3.3 NCCN-IPI Risk Groups and Survival Estimates
The 0–8 total score maps to four risk groups:
| Risk Group | NCCN-IPI Score | 5-Year OS (NCCN Cohort) | 5-Year OS (BCCA Validation) |
|---|---|---|---|
| Low | 0–1 | 96% | 96% |
| Low-Intermediate | 2–3 | 82% | 77% |
| High-Intermediate | 4–5 | 64% | 56% |
| High | 6–8 | 33% | 38% |
The key clinical takeaway is the dramatic widening of the survival gap between the best and worst groups compared with the classic IPI. In the NCCN cohort, the gap between Low and High is 63 percentage points (96% vs. 33%) with the NCCN-IPI, compared to 36 percentage points (90% vs. 54%) with the IPI applied to the same data.7, 5
4. Why the DLBCL Prognosis Panel Matters in Clinical Practice
4.1 Restoring Prognostic Power in the Rituximab Era
The addition of rituximab to CHOP chemotherapy was one of the most significant advances in lymphoma treatment, improving complete response rates and overall survival across all IPI risk groups. However, this success also eroded the IPI's ability to discriminate between intermediate and high-risk patients. In pooled analyses of European randomized trials (MInT, MegaCHOEP, RICOVER-60), the Kaplan-Meier curves for the IPI-defined high-intermediate and high-risk groups converged, with 5-year OS in the combined high-risk group rising to approximately 50%. For clinical decision-making — especially when considering treatment intensification, novel agents, or stem cell transplant consolidation — a tool that cannot identify a truly poor-prognosis population has limited utility.3, 5
The NCCN-IPI directly addresses this by restoring the spread between risk groups. In its derivation cohort, only 8% of patients fell into the high-risk group, but they had a 5-year OS of just 33% — a genuinely poor-prognosis subgroup that could be targeted for novel therapeutic approaches. At the other extreme, the NCCN-IPI low-risk group achieves 96% 5-year OS, allowing confident identification of patients who can be treated with standard R-CHOP and expected to do very well.7, 5
4.2 Informing Treatment Intensity Decisions
Modern DLBCL management increasingly involves risk-adapted therapy. The POLARIX trial demonstrated that substituting vincristine with polatuzumab vedotin (Pola-R-CHP) improved 2-year progression-free survival in patients with an IPI score of 2–5. The IPI and NCCN-IPI are often used to identify which patients may benefit from such intensified regimens, as well as to guide decisions about:12, 13
- CNS prophylaxis: Patients with high-risk features, including involvement of specific extranodal sites (a component directly captured by the NCCN-IPI), are considered for intrathecal chemotherapy or high-dose methotrexate.
- Radiation consolidation: In limited-stage disease, the risk category can influence whether consolidative radiation is added.
- Clinical trial eligibility: Both indices are used as stratification factors in trial design.
4.3 The Specific Problem for Elderly Patients
Age is the most heavily weighted factor in the NCCN-IPI (up to 3 points). This reflects the well-established association between advancing age and both disease biology and treatment tolerance. In elderly patients (> 75 years), the NCCN-IPI is particularly useful because it appropriately penalizes extreme age without collapsing all patients over 60 into a single category.5
4.4 Reducing Calculator Fatigue at Point of Care
On our platform, the Lymphoma (DLBCL) Prognosis Panel is mobile-responsive and engineered for speed. You enter one set of inputs; the panel returns both scores, both risk groups, and both estimated 5-year OS values simultaneously — making it an ideal companion for MDT prep, bedside rounds, and trainee supervision.
5. Clinical Evidence and Validation
5.1 The Math Behind the Tool
Both the Classic IPI and the NCCN-IPI were derived using Cox proportional hazards regression, the standard statistical framework for modeling time-to-event data in the presence of censoring.5, 9
NCCN-IPI: The investigators applied restricted cubic splines to examine the linearity assumption for age and LDH as continuous variables. Age was found to have a roughly linear relationship with the log-hazard, and the optimal categorization was achieved using 15- to 20-year increments. LDH, by contrast, showed a non-linear relationship that plateaued at a ratio of approximately 3 × ULN; further increases above this threshold did not add proportionally to the hazard. All P-values were < 0.01. The final model was selected through backward elimination and the SCORE method, with global model fit assessed using the Akaike Information Criterion (AIC).5
5.2 Validation Data Across Multiple Populations
The NCCN-IPI has been subjected to rigorous external validation:
- Original NCCN cohort (n = 1,650): Concordance probability estimate (CPE) was 0.80 for the NCCN-IPI versus 0.74 for the IPI.5
- BCCA external validation (n = 1,138): CPE was 0.77 versus 0.74, confirming generalization beyond academic centers.7, 5
- Multi-trial pooled analysis (n = 2,124): The NCCN-IPI achieved the highest concordance index (0.632) versus IPI (0.626) and R-IPI (0.590).21, 22
- Asian cohorts: Validated in Taiwan and Japan, confirming the relative risk stratification holds despite differences in disease epidemiology.23, 24
5.3 What the Indices Do Not Capture
- Cell of origin (COO): Neither incorporates GCB vs. ABC subtypes.
- Double-hit/Triple-hit: MYC, BCL2, or BCL6 rearrangements are invisible to these clinical scores.
- Metabolic tumor volume (MTV): PET-CT MTV is independently prognostic but not part of these tools.
- Frailty: In patients >70, tools like the Geriatric Prognostic Index (GPI) may provide more granularity.
6. How Our Calculator Works
6.1 Step 1 — Enter Clinical Variables
The input form at /calculator/panel-lymphoma collects:
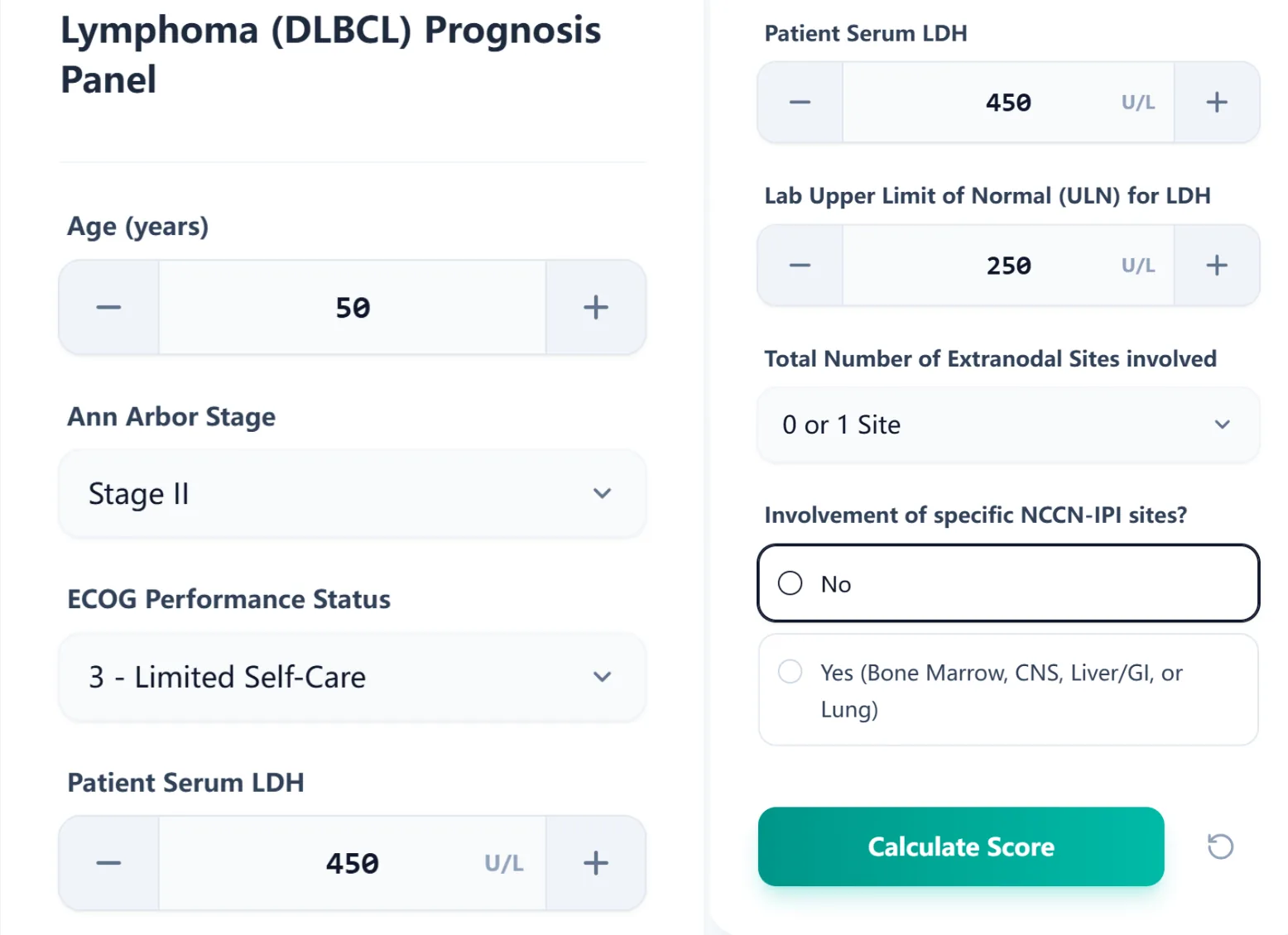
Figure 1. The calculator input form collects all variables needed to compute both the Classic IPI and NCCN-IPI simultaneously.
6.2 Step 2 — Review the Unified Results Panel
After calculation, results appear in a side-by-side display:
- Left card (warm yellow) — Classic IPI (Original)
- Right card (teal) — NCCN-IPI (Enhanced)
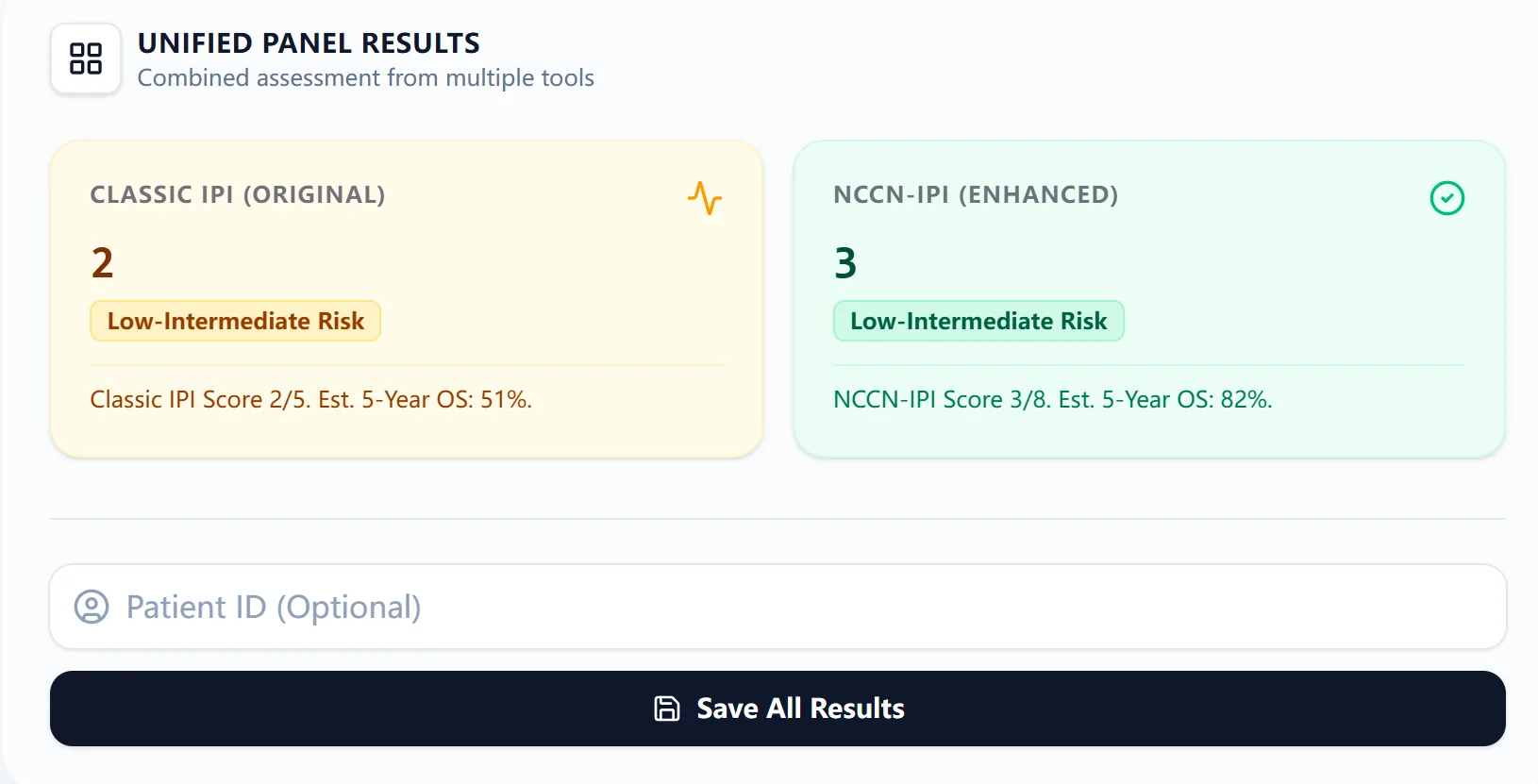
Figure 2. The side-by-side results view highlights discrepancies immediately.
6.3 Step 3 — Consult the Embedded Reference Table
The patient's current risk group is highlighted in a master survival table for context.
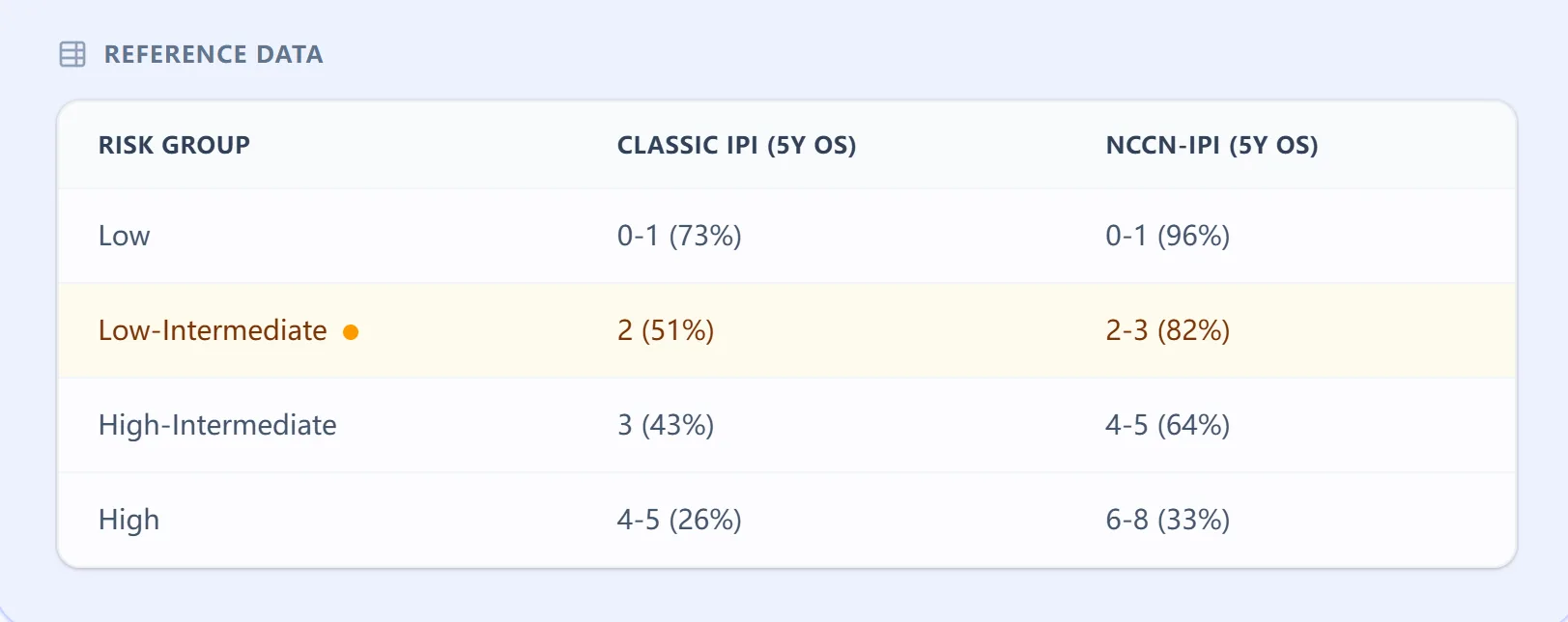
Figure 3. The embedded reference data table.
6.4 Step 4 — Review Clinical Context and Source Evidence
Collapsible panels provide primary source references for transparency.

7. How the Platform Supports Both Clinical Care and Research
7.1 Routine Clinical Decision Support
The panel enables rapid risk stratification directly informing front-line treatment selection, CNS prophylaxis decisions, and MDT preparation.20, 4
7.2 Education and Simulation for Trainees
Functions as a teaching module through variable sensitivity analysis, head-to-head comparison, and LDH normalization practice.
7.3 Clinical Research and Quality Improvement
Consistent, reproducible staging is critical for retrospective cohort studies. Saved results can be linked to patient IDs for institutional registry building.
8. Frequently Asked Questions
8.1 Can the IPI or NCCN-IPI be used in patients with transformed lymphoma?
Both indices were developed for de novo DLBCL. While clinicians sometimes calculate these in transformed cases, the published 5-year OS estimates should not be directly applied.5
8.2 How does the NCCN-IPI differ from the Revised IPI (R-IPI)?
The R-IPI keeps the same scoring but re-groups results. The NCCN-IPI re-engineers the scoring itself with multi-level categorization.21, 5, 22, 8
8.3 What is the most common mistake when calculating the NCCN-IPI manually?
Incorrect LDH normalization. The NCCN-IPI requires the LDH ratio (patient LDH / institution ULN). Our calculator eliminates this source of error.
Ready to Simplify Your DLBCL Risk Stratification?
Calculate Classic IPI and NCCN-IPI side-by-side in seconds.
Use the DLBCL Prognosis Panel
Free to use. No registration required.
References
- Real-world treatment patterns and clinical outcomes ... Source
- Cell of origin is not associated with outcomes of relapsed or refractory diffuse large B cell lymphoma Source
- SEOM–GOTEL clinical guidelines on diffuse large B-cell lymphoma ... Source
- A comprehensive review of the Source
- An enhanced International Prognostic Index (NCCN-IPI) for ... Source
- Diffuse Large B-cell Lymphoma Prognosis Calculator | DLBCL IPI ... Source
- An enhanced International Prognostic Index (NCCN-IPI) for ... Source
- IPI (International Prognostic Index), R-IPI (revised IPI), NCCN-IPI prognostic scores for predicting overall survival in DLBCL Source
- International Prognostic Index - Wikipedia Source
- A predictive model for aggressive non-Hodgkin's lymphoma Source
- Diffuse Large B-cell Lymphoma Prognosis Calculator (IPI) Source
- Pola-R-CHP performed well in first-line Polarix trial-ineligible and IPI ... Source
- Pola-R-CHP performed well in first-line Polarix trial-ineligible ... Source
- CNS prophylaxis is (mostly) futile in DLBCL | BJC Reports Source
- Biological Subtypes... Source
- Clinical Outcomes in Patients With DLBCL Treated With R ... Source
- A Real‐World Data‐Based Analysis of Prognostic Indices as Part of Trial Eligibility Criteria in Diffuse Large B‐Cell Lymphoma Patients Source
- Assessment Predicts Survival In Elderly Patients with DLBCL Source
- The Geriatric Prognostic Index: a clinical prediction model ... Source
- PET-CT staging of DLBCL accurately identifies and provides new insight into the clinical significance of bone marrow involvement Source
- International prognostic indices in diffuse large B-cell lymphoma: a comparison of IPI, R-IPI, and NCCN-IPI Source
- International prognostic indices in diffuse large B-cell lymphoma: A comparison of IPI, R-IPI, and NCCN-IPI Source
- Comparison of Prognostic Indices in Japanese Patients with Diffuse Large B-cell Lymphoma in the Yonago Area Source
- Validation of an enhanced International Prognostic Index ... Source
- Validation of the International Prognostic Index and ... - PMC Source
- Discussion Source
- Simplicity at the cost of predictive accuracy in diffuse large B‐cell lymphoma: a critical assessment of the R‐IPI, IPI, and NCCN‐IPI Source
- Molecular Classification and Treatment of Diffuse Large B-cell ... Source