D’Amico Risk Classification Calculator | OncoToolkit
Accurately stratify localized prostate cancer risk using PSA, Gleason score, and T stage with our guideline-aligned D’Amico Risk Classification calculator.

Quick Navigation
- 01Introduction to D’Amico Risk Classification
- 02What is the D’Amico Risk Classification System?
- 03Clinical Significance of the D’Amico Score in Practice
- 04Specific Clinical Use Cases for the D’Amico Calculator
- 05Clinical Evidence and International Guidelines
- 06Recent Expansions and Updates Beyond Classic D’Amico
- 07Operating the D’Amico Calculator on OncoToolkit
- 08Supporting Clinical Care, Education, and Research
- 09Clinical FAQ
- 10References
1. Introduction to D’Amico Risk Classification
In contemporary prostate cancer practice, clinicians must integrate PSA, Gleason grade, clinical T stage, imaging, comorbidities, and patient preferences to personalize treatment. The D’Amico Risk Classification calculator on OncoToolkit provides a fast, standardized way to assign low, intermediate, or high‑risk groups, forming a backbone for treatment discussions, MDT decisions, and research workflows. This enriched overview focuses on concrete clinical use cases, how major international guidelines reference and build upon D’Amico, and recent expansions beyond the original three‑tier model.1, 2, 3, 4, 5
At OncoToolkit, we have designed our D’Amico calculator not just as a digital version of a paper table, but as a clinically intelligent interface that aligns with EAU, NCCN, AUA/ASTRO and NICE‑related practice while remaining easy to use in high‑pressure environments. By combining clear inputs, transparent logic, and evidence‑anchored interpretation, the tool helps reduce cognitive load and supports consistent, guideline‑concordant care.5, 6, 7, 8, 9, 1
2. What is the D’Amico Risk Classification System?
The D’Amico system stratifies men with clinically localized prostate cancer into three risk groups—low, intermediate, and high—using pretreatment PSA, biopsy Gleason score, and clinical T stage. The aim is to estimate the risk of biochemical recurrence following radical prostatectomy or radiotherapy and to guide treatment intensity.3, 4, 10, 11, 5
Classic definitions are:
- Low risk: PSA ≤ 10 ng/mL, Gleason score ≤ 6, and clinical stage T1–T2a.12, 3
- Intermediate risk: PSA 10–20 ng/mL or Gleason score 7 or clinical stage T2b.3, 12
- High risk: PSA > 20 ng/mL or Gleason score 8–10 or clinical stage ≥ T2c.12, 3
Early JAMA cohorts and follow‑up analyses demonstrated clear separation in biochemical recurrence‑free survival between these groups after surgery, external‑beam radiation, and interstitial brachytherapy. Contemporary reviews confirm that D’Amico remains an important reference framework even as risk stratification has evolved to include more granular systems and genomic markers.2, 4, 11, 5
Figure 1. Clinical background panel summarizing that the D’Amico system uses PSA, biopsy Gleason score, and clinical T stage to predict biochemical recurrence after localized therapy, with risk group determined by the worst feature present, reflecting the original publications.4, 11
3. Clinical Significance of the D’Amico Score in Practice
For prostate cancer oncologists, D’Amico risk grouping informs several pivotal decisions: eligibility for active surveillance, intensity of local treatment, the role and duration of androgen deprivation therapy, and whether advanced imaging or nodal staging is warranted. Low‑risk patients are often candidates for active surveillance under EAU, NCCN, and AUA/ASTRO frameworks, whereas intermediate‑ and high‑risk groups more commonly receive definitive surgery or radiation with consideration of systemic therapy.6, 8, 13, 1, 2
Without digital support, applying D’Amico criteria may seem straightforward but becomes error‑prone when multiple variables are in flux—especially as PSA, biopsy reports, and imaging results evolve over time. Paper tables and mental rules such as “highest‑risk feature wins” are easy to misapply under time pressure, leading to inconsistent classification across clinicians and institutions.9, 2, 5
Clinical Excellence: The OncoToolkit calculator automates the "worst feature" rule, ensuring that patients are consistently and correctly stratified according to the highest risk factor identified.
On our platform, the D’Amico Risk Classification calculator minimizes these pain points by providing:
- A mobile‑responsive interface to enter PSA, Gleason score, and T stage in a single view.
- Automated implementation of the “worst feature” rule, ensuring consistent assignment across users and time.4, 3
- Instant translation of the risk group into approximate recurrence risk and a narrative explanation suitable for documentation and patient communication.5, 3
Because the tool is optimized for quick use on phones, tablets, and desktops, it fits naturally into MDT meetings, radiotherapy planning sessions, and ward rounds where consistent risk language is essential.14, 15
4. Specific Clinical Use Cases for the D’Amico Calculator
4.1 Initial Staging and Counseling at Diagnosis
At diagnosis, D’Amico risk grouping offers a rapid way to frame prognosis and treatment options alongside more detailed assessments such as MRI, PSMA PET, and geriatric evaluation.1, 2
Common scenarios where our calculator adds value include:
- Determining whether a newly diagnosed patient with PSA ≤ 10, Gleason 3+3, and T1c/T2a disease meets low‑risk criteria, a group for whom EAU, NCCN, and AUA/ASTRO guidelines support active surveillance in appropriately selected men.8, 6, 1
- Clarifying intermediate‑risk status when only a single adverse feature (for example, PSA 11 with Gleason 3+4 and T1c) is present, which may align with “favorable intermediate risk” in NCCN schemes and allow consideration of surveillance or focal therapy in selected cases.16, 2
- Identifying high‑risk localized disease (for example, Gleason 8 with PSA 9 and T2c), a group for whom multimodal treatment with surgery or radiation plus systemic therapy is often recommended.13, 6, 1
4.2 MDT (Tumor Board) Preparation and Case Standardization
Using a shared digital tool ensures that all MDT participants refer to the same risk classification when reviewing cases.15, 14
- Referring clinicians can compute and document D’Amico risk at the time of referral, reducing variability from ad‑hoc classification.
- In tumor boards, displaying the OncoToolkit D’Amico result (“Intermediate Risk due to Gleason 7”) anchors discussions in a standardized language before layering imaging, genomic results, and patient factors.1, 5
4.3 Radiotherapy and Surgical Planning
Many treatment algorithms in urology and radiation oncology are still organized around low, intermediate, and high‑risk categories.
- In radiotherapy, high‑risk disease is associated with recommendations for dose escalation, pelvic nodal fields, and long‑term ADT, whereas low‑risk disease may be suitable for hypofractionated or brachytherapy‑only approaches.6, 13, 1
- In surgery, high‑risk D’Amico classification often triggers discussion of extended pelvic lymph node dissection and closer PSA follow‑up, consistent with AUA/ASTRO and EAU guidance.17, 13, 1
Our calculator provides a quick confirmation of D’Amico class during planning, helping align plans with these guidelines.
4.4 Triage for Advanced Imaging and Staging
D’Amico risk remains strongly associated with metastatic probability and imaging metrics, even in the era of multiparametric MRI and PSMA PET.18, 12
- Guidelines commonly recommend bone scan or advanced imaging for patients with unfavorable intermediate‑ or high‑risk disease; the D’Amico result supports documentation of why PSMA PET or whole‑body MRI is being ordered.2, 8, 1
- Recent work shows that higher D’Amico categories correlate with higher PSMA SUVmax and greater metastatic burden, underscoring that risk grouping still reflects underlying tumor biology.12
4.5 Post-Treatment Follow-Up and Survivorship
Risk group at diagnosis influences follow‑up intensity.
- High‑risk D’Amico patients have a greater likelihood of biochemical recurrence and are typically followed with more frequent PSA testing than low‑risk men, in line with EAU and NCCN schedules.8, 2, 1
- Documenting the D’Amico class via the calculator at baseline helps institutions audit whether follow‑up intensity matches baseline risk and assess long‑term outcomes by risk group.19, 5
4.6 Research, QI, and Registry Work
D’Amico risk groups remain common stratification variables in clinical trials and observational studies.2, 5
- Prospective use of the OncoToolkit calculator ensures standardized risk assignment across centers, improving comparability of biochemical recurrence and survival outcomes.19, 5
- Trials frequently stratify or adjust for D’Amico or NCCN risk categories, and registries often record these groups in addition to PSA and Grade Group.5, 19, 2
5. Clinical Evidence and International Guidelines
5.1 How Major Guidelines Incorporate D’Amico Risk Groups
EAU Guidelines (Europe). EAU classification for localized and locally advanced disease uses PSA, ISUP Grade Group, and clinical T stage to define low, intermediate, and high‑risk groups, with treatment recommendations tailored to these categories. This structure is conceptually similar to D’Amico and draws on the same triad of variables, while integrating contemporary staging and MRI findings.13, 1, 5
NCCN Guidelines (United States). NCCN risk stratification evolved directly from D’Amico, defining very low, low, favorable intermediate, unfavorable intermediate, high, and very high‑risk categories based on PSA, Grade Group, clinical stage, PSA density, and extent of biopsy involvement. The underlying cut‑offs for PSA and Gleason remain aligned with the original D’Amico concepts (≤10, 10–20, >20; Gleason ≤ 6, 7, ≥ 8).16, 8, 2, 5
AUA/ASTRO/SUO Guidelines. The AUA/ASTRO guideline on clinically localized prostate cancer recommends risk stratification using PSA, Grade Group, and clinical stage; in practice, these categories mirror D’Amico and NCCN groupings and are used to anchor recommendations from active surveillance to multimodal therapy.20, 17, 6
NICE and UK practice. NICE evidence reviews describe traditional three‑tier risk models and highlight newer systems such as the Cambridge Prognostic Groups (CPG1–5), which refine D’Amico/NCCN schemes and are used in several UK centers.7, 21, 22
5.2 The Mathematical Logic Behind the Tool
The D’Amico system is rule‑based rather than regression‑based: each variable is mapped to a risk level (low, intermediate, high), and the final class equals the maximum (worst) level across PSA, Gleason, and T stage. This “worst feature present” principle has been validated across radical prostatectomy and radiotherapy cohorts as a strong predictor of biochemical recurrence.11, 3, 4, 5
Our calculator implements this logic exactly by:
- Categorizing PSA as ≤10, 10–20, or >20 ng/mL.
- Grouping Gleason/ISUP into ≤6 (Grade Group 1), 7 (Grade Groups 2–3), or ≥8 (Grade Groups 4–5).1, 12
- Grouping clinical T stage into T1c–T2a, T2b, or ≥T2c, consistent with TNM and guideline tables.2, 1
The output also explicitly lists which variable(s) drive the final risk group, improving transparency and educational value.
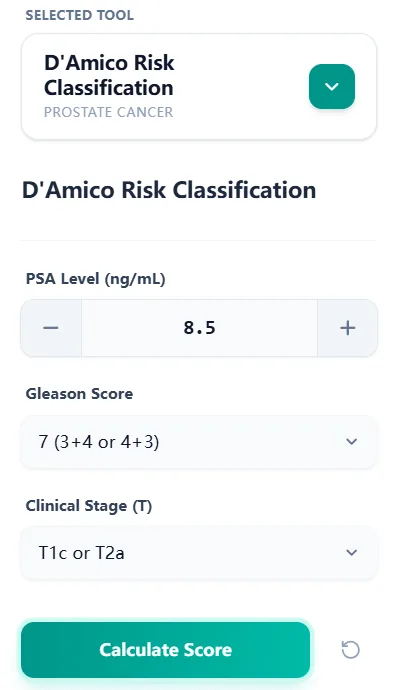
Figure 2. Input panel where PSA, Gleason category, and clinical T stage are entered; each field reflects guideline cut‑offs and TNM groupings, making the calculated class compatible with EAU, NCCN, AUA/ASTRO, and NICE‑influenced practice.7, 6, 1
5.3 Contemporary Validation and Imaging Correlations
Recent studies confirm that D’Amico risk retains prognostic value in the era of MRI and PSMA PET/CT.
- A 2024 PSMA PET/CT study demonstrated that D’Amico and ISUP classifications correlate with prostate SUVmax and metastatic status, with patients in higher D’Amico categories showing significantly higher SUVmax and more frequent metastases.12
- MRI‑based studies show that D’Amico risk correlates with tumor volume and adverse features on multiparametric MRI, although MRI may upstage some patients initially thought to be low risk.18, 2
- Analyses of high‑risk D’Amico patients indicate that outcomes vary by the number and type of high‑risk factors, supporting modern subdivisions such as very high‑risk and unfavorable intermediate risk.23, 24
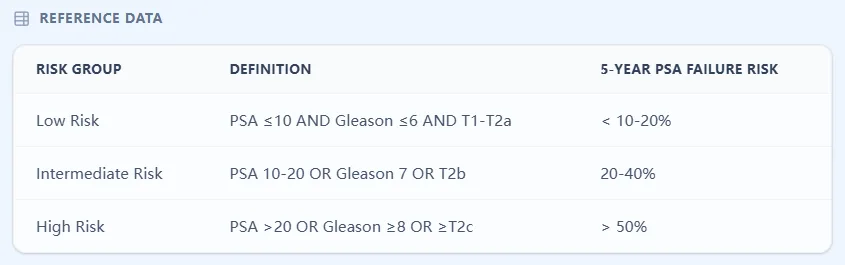
Figure 3. Reference table outlining low, intermediate, and high‑risk definitions with approximate 5‑year PSA failure risks from early cohorts, summarizing the prognostic separation demonstrated in original surgical and radiotherapy series.11, 3, 4
6. Recent Expansions and Updates Beyond Classic D’Amico
6.1 Subdividing Intermediate‑ and High‑Risk Groups
Several groups have refined D’Amico to better reflect heterogeneity:
- NCCN subdivides intermediate risk into favorable and unfavorable based on the number of intermediate‑risk factors, Grade Group, and proportion of positive cores, recognizing that outcomes for favorable intermediate risk may approximate low‑risk disease.25, 8, 16
- High‑risk disease is further partitioned into high and very high risk in NCCN algorithms, with very high‑risk defined by T3b–T4, primary Gleason pattern 5, or multiple high‑risk features.8, 2
- Studies of D’Amico high‑risk cohorts show that men with multiple high‑risk criteria have poorer biochemical control and survival, justifying these subdivisions.23, 5
6.2 Cambridge Prognostic Groups and Other Multi‑tier Systems
The Cambridge Prognostic Group (CPG) classification divides localized prostate cancer into five risk groups and has been shown to improve prediction of prostate cancer‑specific mortality compared with traditional three‑tier systems. CPG uses PSA, Grade Group, and T stage—similar to D’Amico—but with refined combinations and cut‑offs.22, 26
Other models, such as the Candiolo classifier, incorporate additional parameters and statistical modeling to extend D’Amico and improve prediction after external‑beam radiotherapy. Reviews of risk tools consistently use D’Amico as the historical reference against which newer systems are compared.19, 5, 2
OncoToolkit’s D’Amico calculator is therefore a foundational tool that can sit alongside more granular calculators (for example, CPG or CAPRA) for users who need refined prognostic estimates.
6.3 Integration with MRI, PSMA PET, and Genomic Biomarkers
Modern practice integrates clinical risk with advanced imaging and genomics:
- Multiparametric MRI improves detection and localization of clinically significant disease, and its findings refine risk assessment beyond PSA and biopsy alone, though D’Amico groups still correlate with MRI suspicion and tumor burden.18, 2
- PSMA PET identifies nodal and distant metastases at lower PSA levels; higher D’Amico risk groups show greater PSMA uptake and metastasis rates, but imaging can also upstage some intermediate‑risk patients into treatment paradigms more typical of high‑risk disease.18, 12
- Genomic classifiers and AI‑based histopathology models demonstrate superior prognostic discrimination compared with traditional clinical risk groups, yet they rely on D’Amico/NCCN categories as baselines and are typically used as add‑ons rather than replacements.27, 28, 2
6.4 Limitations and Practical Considerations
Recognized limitations include:
- Heterogeneity within each risk group; for example, not all intermediate‑risk patients have the same recurrence risk, especially when only one intermediate feature is present.24, 5
- Calibration differences between historical cohorts and modern populations treated with MRI‑targeted biopsies and advanced radiation techniques.29, 2
- Limited incorporation of comorbidity, life expectancy, and patient preference, which guidelines emphasize when recommending active surveillance or definitive therapy.6, 7, 1
Our calculator addresses these by clearly labeling outputs as decision‑support estimates, highlighting the driving risk factor(s), and reminding users that D’Amico classification should complement—not replace—clinical judgment and multidisciplinary discussion.9, 5
7. Operating the D’Amico Calculator on OncoToolkit
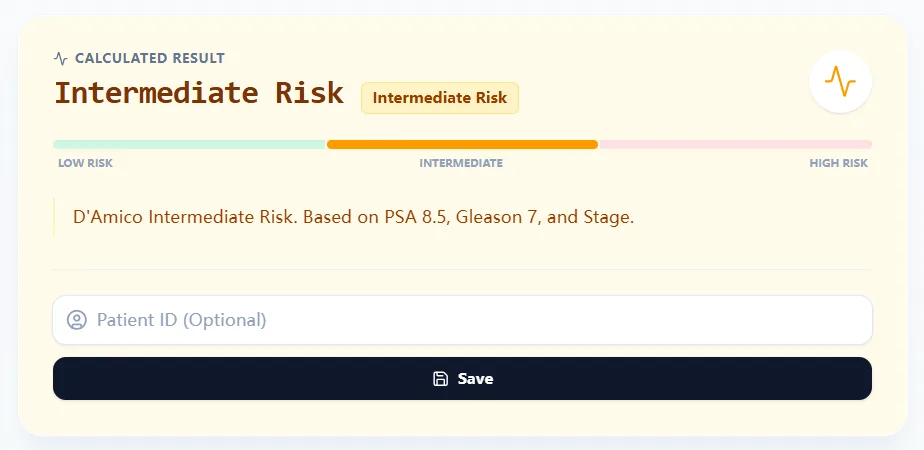
Figure 4. Example output showing an Intermediate Risk result with a narrative explanation tying the classification to PSA, Gleason score, and T stage, displayed on a visual risk scale for rapid MDT communication and documentation.
Step‑by‑step user journey
- Enter PSA in ng/mL; the calculator maps it into ≤10, 10–20, or >20 ng/mL bands in line with D’Amico and major guidelines.3, 5
- Select biopsy Gleason score category (≤6, 7, ≥8), corresponding to ISUP Grade Groups 1–5.1, 12
- Select clinical T stage grouping (T1c/T2a, T2b, ≥T2c) according to TNM categories used in guideline tables.2, 1
- Click “Calculate Score” to obtain the D’Amico risk class, a color‑coded risk band, and a short explanatory sentence that can be pasted into notes.
The calculator’s clarity makes it ideal for trainees, who can run “what‑if” simulations by adjusting PSA, Gleason, or stage and observing risk transitions.
8. Supporting Clinical Care, Education, and Research
Clinical Decision Support
- At diagnosis, the D’Amico result anchors discussions about active surveillance versus definitive therapy within guideline‑defined risk categories.6, 8, 1
- During planning, it supports choices about imaging, surgery, radiotherapy, and systemic therapy consistent with EAU, NCCN, AUA/ASTRO, and NICE recommendations.7, 13, 6, 1
- OncoToolkit can also surface related calculators—for example, CAPRA score, postoperative recurrence nomograms, and ADT toxicity tools—providing a layered decision‑support environment.30, 5, 2
Education and Simulation
- The calculator illustrates how core clinical variables drive risk, reinforcing guideline‑based thinking for residents, fellows, and advanced practice providers.31, 2
- Educators can compare D’Amico outputs with NCCN and CPG risk groups during journal clubs, highlighting the evolution from simple triads to multi‑tier systems.22, 2
Clinical Research and Quality Improvement
- Prospective use standardizes risk labels across institutions, enabling robust comparisons of biochemical recurrence, metastasis, and survival by D’Amico class.5, 19
- When paired with outcomes dashboards, clinicians can evaluate whether local results for low, intermediate, and high‑risk disease align with or exceed benchmarks from published cohorts.5, 2
9. Clinical FAQ
When should D’Amico classification be supplemented with additional tools?
D’Amico classification should be supplemented when MRI, PSMA PET, or genomic testing reveals disease extent or biology that is discordant with clinical risk—for example, extensive MRI‑visible lesions or PSMA‑positive nodes in a formally low‑risk patient. Guidelines encourage integrating imaging and biomarkers with clinical risk rather than relying on D’Amico alone, particularly for decisions about intensifying or de‑escalating therapy.6, 12, 18, 1, 2
How does D’Amico relate to NCCN and Cambridge Prognostic Groups?
NCCN’s multi‑tier scheme extends D’Amico by subdividing intermediate and high‑risk disease and introducing very low and very high categories, but it relies on the same core variables. The Cambridge Prognostic Groups similarly build on D’Amico/NCCN data to create five risk groups that better predict prostate cancer‑specific mortality in UK cohorts.26, 16, 22, 8, 2
Are there validated modifications that incorporate MRI or PSMA PET into D’Amico?
Several studies have evaluated combinations of D’Amico risk with multiparametric MRI scores or PSMA SUVmax, showing improved prediction of extracapsular extension, nodal disease, and metastases when imaging is added. However, major guidelines currently treat imaging and D’Amico as complementary rather than merging them into a formal combined score, so our calculator deliberately preserves the original definition.12, 18, 1, 2, 6
What are the main pitfalls when applying D’Amico in 2026 practice?
Common pitfalls include using post‑treatment rather than pretreatment PSA or stage, ignoring MRI‑based upstaging, and treating all intermediate‑risk patients as homogeneous despite evidence of marked heterogeneity. Another risk is extrapolating D’Amico directly to overall survival without considering age and comorbidities, which guidelines stress when recommending active surveillance in older or frail men.24, 23, 7, 1, 5, 6
Is D’Amico still relevant in the age of genomic classifiers and AI pathology?
Yes—although genomic and AI‑based models often show superior discrimination, they usually use D’Amico or NCCN risk groups as comparators and require the same clinical inputs for calibration. D’Amico remains a globally recognized, low‑cost framework that is especially valuable where advanced testing is unavailable or as a common language across institutions with varying resources.28, 27, 31, 2, 5
Ready to Simplify Prostate Cancer Risk Assessment?
Access the guideline-aligned D’Amico calculator to enhance your clinical decision-making today.
Use the Calculator Now
Free to use. No registration required.
References
- EAU Guidelines: Prostate Cancer - Classification and Staging Systems. Source
- Prognostic Value of D'Amico Risk Groups. Source
- Evidencio - D'Amico Risk Groups. Source
- D'Amico et al., JAMA 1998; 280:969-974. Source
- Contemporary Review of Prostate Risk Stratification. Source
- AUA/ASTRO Guideline: Localized Prostate Cancer. Source
- NICE Evidence Review: Risk Stratification. Source
- NCCN Guidelines for Localized Prostate Cancer. Source
- Reducing Errors in Prostate Risk Calculation. Source
- NCI Dictionary: D'Amico Criteria. Source
- JAMA: Predictive Value of D'Amico Classification. Source
- Correlation of D’Amico and ISUP with PSMA PET/CT SUVmax. Source
- EAU Guidelines: Treatment of Localized Prostate Cancer. Source
- Optimizing MDT Meetings with Digital Tools. Source
- Standardization of Tumor Boards. Source
- Evolving NCCN Risk Groups. Source
- Surgery for High-Risk Prostate Cancer. Source
- MRI Findings and D'Amico Risk. Source
- Long-term Outcomes by Risk Stratification. Source
- Nursing Summary: Prostate Cancer Guidelines. Source
- NICE Evidence Reviews for Localized Disease. Source
- Cambridge Prognostic Groups Analysis. Source
- Outcomes in Multiple High-Risk Factor Men. Source
- Heterogeneity in Intermediate-Risk Disease. Source
- Favorable vs Unfavorable Intermediate-Risk. Source
- Cancer Research UK: CPG Groups. Source
- Nature: genomic Classifiers in Risk Groups. Source
- ASCO: AI Histopathology and Clinical Risk. Source
- Validation of Risk Tools in Modern Cohorts. Source
- UCSF CAPRA Score. Source
- PCF: Understanding Risk Groups. Source