MSKCC Gastric Cancer Nomogram: Online Calculator for Post-Resection Survival Prediction
Predict 5-year disease-specific survival after R0 resection using the validated MSKCC Gastric Cancer Nomogram. A vital tool for individualized oncology care.

MSKCC Gastric Cancer Nomogram Calculator Interface
Quick Navigation
1. Introduction to the MSKCC Gastric Cancer Nomogram
Gastric cancer remains a leading cause of cancer-related mortality worldwide, and even after an R0 gastrectomy, prognosis varies widely between patients who appear similar by stage alone. For upper GI and pancreatic surgeons, oncologists, and MDTs, providing an individualized survival estimate is central to counselling, adjuvant therapy decisions, and follow-up planning.1, 2, 3
The MSKCC Gastric Cancer Nomogram was developed at Memorial Sloan Kettering Cancer Center to address this challenge by predicting disease-specific survival (DSS) at 5 and 9 years after curative resection for primary gastric adenocarcinoma. Instead of placing patients into broad AJCC stage groups, it uses multiple clinicopathological variables to generate a continuous, patient-specific probability of survival.4, 1
At OncoToolkit, this validated prognostic model has been implemented as a streamlined, mobile-responsive calculator so clinicians can obtain results in seconds during MDT meetings, ward rounds, or outpatient clinics: /calculator/mskcc-gastric.5
2. Understanding the MSKCC Gastric Cancer Nomogram
The MSKCC Gastric Cancer Nomogram is a Cox proportional hazards–based prognostic model derived from 1,039 patients undergoing R0 gastrectomy for primary gastric carcinoma at MSKCC between 1985 and 2000. The primary endpoint is disease-specific survival, defined as death attributable to gastric cancer, which isolates tumor-related risk from competing comorbid mortality.1, 4
2.1 Original Model Variables and Development
The original JCO 2003 model incorporates the following variables:6, 4, 1
- Age at surgery (continuous)
- Sex
- Primary tumor site (proximal, middle, distal stomach, or gastroesophageal junction)
- Lauren histologic type (intestinal, diffuse, mixed)
- Pathologic depth of invasion (pT)
- Number of positive lymph nodes
- Number of negative lymph nodes
- Tumor size
Each covariate is assigned a regression coefficient in the Cox model; together, they form a linear predictor that is mapped to survival probabilities at 5 and 9 years. In the derivation cohort, the bootstrap‑corrected concordance index (C‑index) for the nomogram was approximately 0.80, significantly better than AJCC stage groupings alone (P < 0.001).4, 1
2.2 The OncoToolkit Implementation Strategy
On our platform, the calculator uses a simplified point-based approximation of the original nomogram, focusing on four clinically dominant and easily captured variables:5
- Depth of invasion (pT stage)
- Number of positive lymph nodes / nodal burden
- Tumor location (proximal vs body/middle vs distal stomach)
- Age group
The tool converts each input into points reflecting the original Cox coefficients, sums them into a total score, and maps that score to an estimated 5‑year DSS and a risk category (Favorable, Intermediate, Poor).
Pearls: The clinical context panel summarises the nomogram’s purpose, evidence source (Kattan et al., JCO 2003), and formula logic based on T‑stage, nodal burden, tumor site, and age. This helps attending surgeons and trainees quickly confirm they are using the tool in the right population.
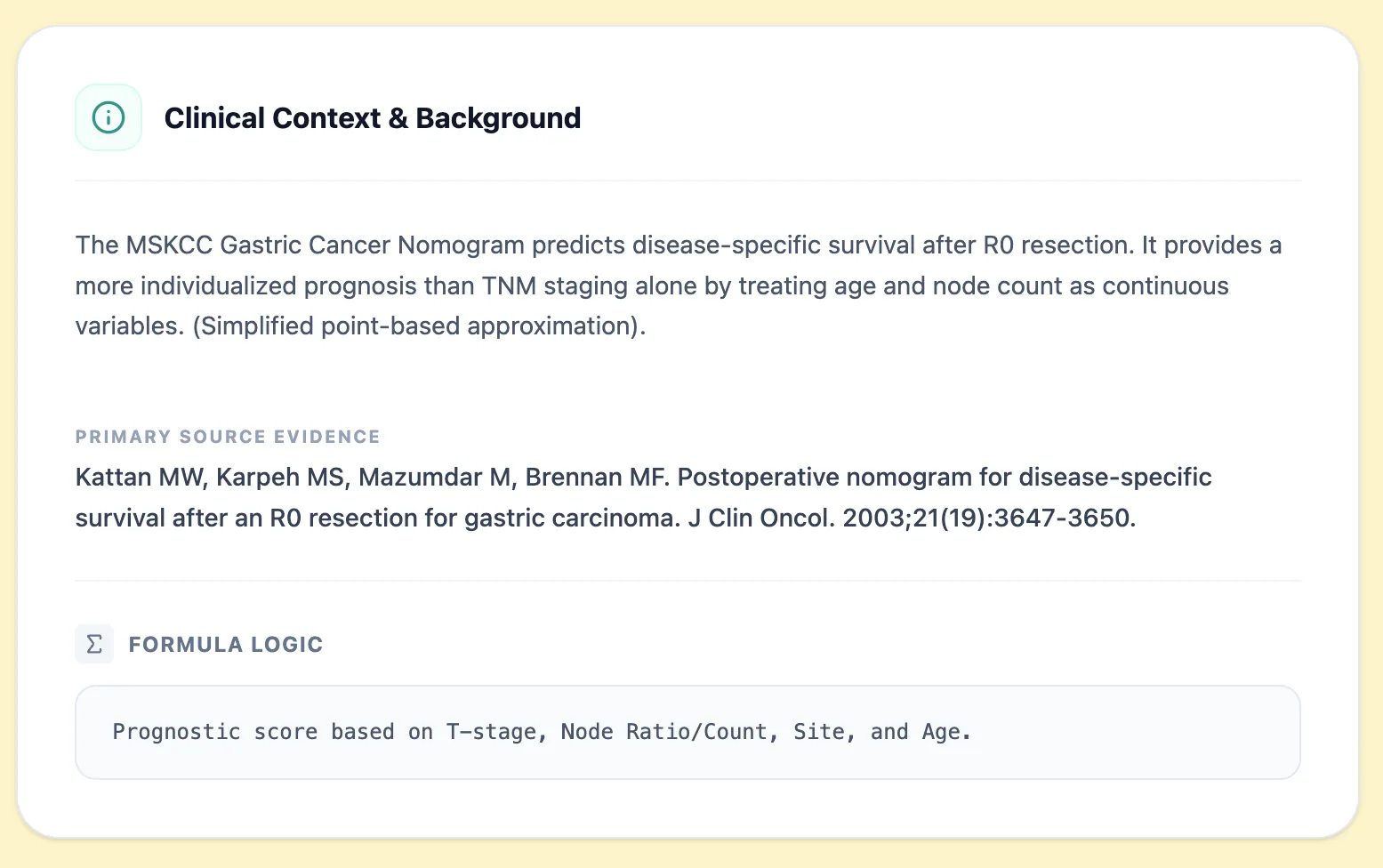
Clinical context panel of the MSKCC Gastric Cancer Nomogram
3. Clinical Significance: Why the MSKCC Nomogram Matters in Practice
3.1 Overcoming the Shortcomings of TNM Staging Alone
AJCC TNM staging remains fundamental for treatment planning and communication, but it has recognized prognostic limitations in gastric cancer:2, 7
- Stage heterogeneity: Patients within the same AJCC stage, particularly stages II and III, can have widely divergent outcomes.7
- Limited covariates: TNM focuses on anatomic extent and does not integrate age, histology, or nuanced nodal metrics such as negative node count.7, 1
- Categorical structure: Stage groupings force continuous variables into discrete categories, losing granularity that is crucial for individualized prognosis.1
Multiple external validations show that the MSKCC nomogram consistently outperforms TNM staging in discriminating DSS, with C‑indices around 0.72–0.80 compared with 0.66–0.72 for AJCC staging.8, 9, 10
3.2 Impact on Real-World Clinical Decision Making
For upper GI and pancreatic surgeons, gastric cancer specialists, and oncologists, the nomogram has practical implications:
- Postoperative counselling: Quantitative, patient-specific survival estimates support transparent conversations about prognosis and long-term expectations.1
- Adjuvant therapy decisions: Where guidelines offer flexibility, baseline DSS can help frame potential absolute benefit from chemotherapy or chemoradiation.3
- Risk-adapted surveillance: Patients in higher-risk categories may warrant more intensive follow-up imaging and endoscopy, while favorable‑risk cases may be observed less intensively within guideline boundaries.3
- Clinical trial design: Nomogram scores can be used to stratify or select patients in trials evaluating perioperative, adjuvant, or systemic strategies.2
3.3 Reducing Calculator Fatigue and Cognitive Load
In high‑volume MDTs, clinicians may review dozens of complex upper GI cancer cases in a single sitting, and observational data indicate that decision quality can deteriorate as teams develop decision‑making fatigue. Manually consulting a paper nomogram—assigning points, summing them, and reading off survival probabilities—adds to cognitive load and increases the chance of arithmetic errors.11, 12, 13
At OncoToolkit, the MSKCC Gastric Cancer Nomogram calculator has been deliberately built to minimise these burdens:
- Four dropdowns plus one click replaces multi-step manual scoring.
- Mobile responsiveness supports quick use during ward rounds and MDTs without returning to a desktop workstation.5
- Immediate visual and textual interpretation eliminates the need to cross‑reference static tables.
This aligns with broader evidence that well‑designed digital decision support can improve adherence to evidence-based care while reducing perceived mental workload.13, 14
4. Clinical Evidence and Global Validation Data
4.1 The Statistical Methodology Behind the Tool
The underlying model is a Cox proportional hazards regression, a standard survival analysis approach in oncology. Each predictor x_i contributes a coefficient β_i; the combined linear predictor η = β_1 x_1 + β_2 x_2 + ... + β_n x_n is linked to the hazard of disease-specific death, and via the baseline hazard function, to survival probabilities at 5 and 9 years.4, 1
On our platform, the continuous linear predictor is represented as a point score:
- Each category of pT, nodal burden, site, and age has a pre-computed point value.
- The total Points is the sum of these values.
- This total is mapped to a 5‑year DSS range and a risk group, reproducing the original model’s predicted probabilities.5
| Study | Population | N | C‑index (Nom) | C‑index (AJCC) | Key findings |
|---|---|---|---|---|---|
| Kattan et al. 2003 | MSKCC (US) | 1,039 | 0.80 | — | Superior to AJCC stage groups |
| Novotny et al. 2006 | Munich (Germany) | 862 | 0.77 | 0.756 | Better discrimination (P < 0.008) |
| Chen/Qin et al. 2013 | Peking Univ (China) | 979 | 0.74 | 0.69 | Superior to AJCC (P < 0.0001) |
4.3 Comparative Research on Nodal Metrics
Subsequent research has explored alternative nomograms based on metastatic lymph node count, lymph node ratio, and log‑odds of metastatic nodes. Many of these models achieve similar or slightly higher C‑indices (≈0.74–0.80) than AJCC TNM, reinforcing the central role of granular nodal information in gastric cancer prognosis. However, the MSKCC nomogram remains one of the most widely recognized and historically validated tools used in both Western and Asian centres.9, 10, 18, 19, 8
4.4 Current Limitations and Scope of Clinical Use
Clinicians should keep several limitations in mind:
- Surgery‑only derivation: The original cohort largely did not receive neoadjuvant or adjuvant therapy.
- GEJ and neoadjuvant era: Re‑validation has shown reduced accuracy for gastroesophageal junction cancers treated with modern neoadjuvant regimens.
- Molecular factors: The nomogram predates widespread use of HER2, MSI, and PD‑L1 testing.
Within its intended scope—primary gastric adenocarcinoma, R0 resection, no neoadjuvant/adjuvant therapy—the MSKCC nomogram remains a high‑performing, guideline‑compatible prognostic tool.10, 1
5. Operational Guide: How the MSKCC Gastric Cancer Nomogram Calculator Works
5.1 Input Requirements: From Pathology Report to Score
On the OncoToolkit calculator, the input process is deliberately minimal:
- Depth of invasion (pT stage) – Select T1, T2, T3, or T4.
- Number of positive nodes – Choose from clinically meaningful ranges (e.g., 0, 1–2, 3–6, 7–15, >15).
- Tumor location – Proximal, body/middle, or distal stomach.
- Age – Select the relevant age band at the time of surgery.
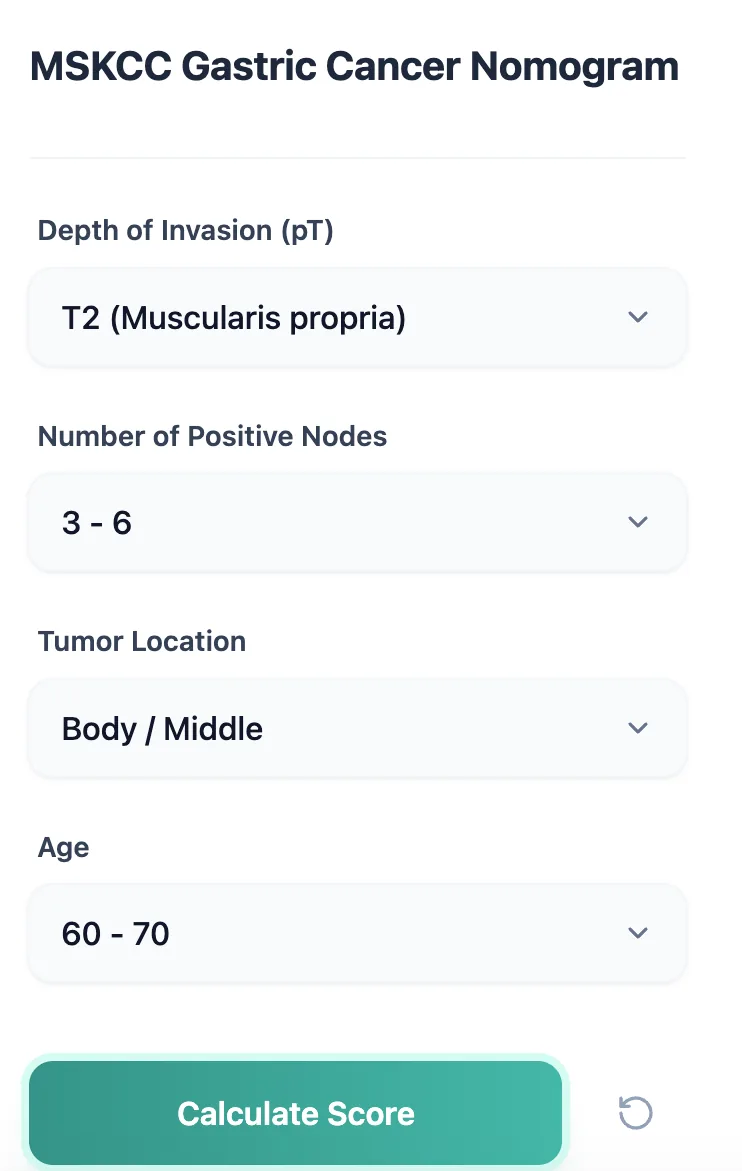
Structured input form for the gastric nomogram calculator
5.2 Understanding the Outputs: Score and Interpretation
The calculator returns:
- Points: The total point score.
- Risk category: Favorable (<100), Intermediate (100–180), or Poor (>180).
- Estimated 5‑year DSS range.
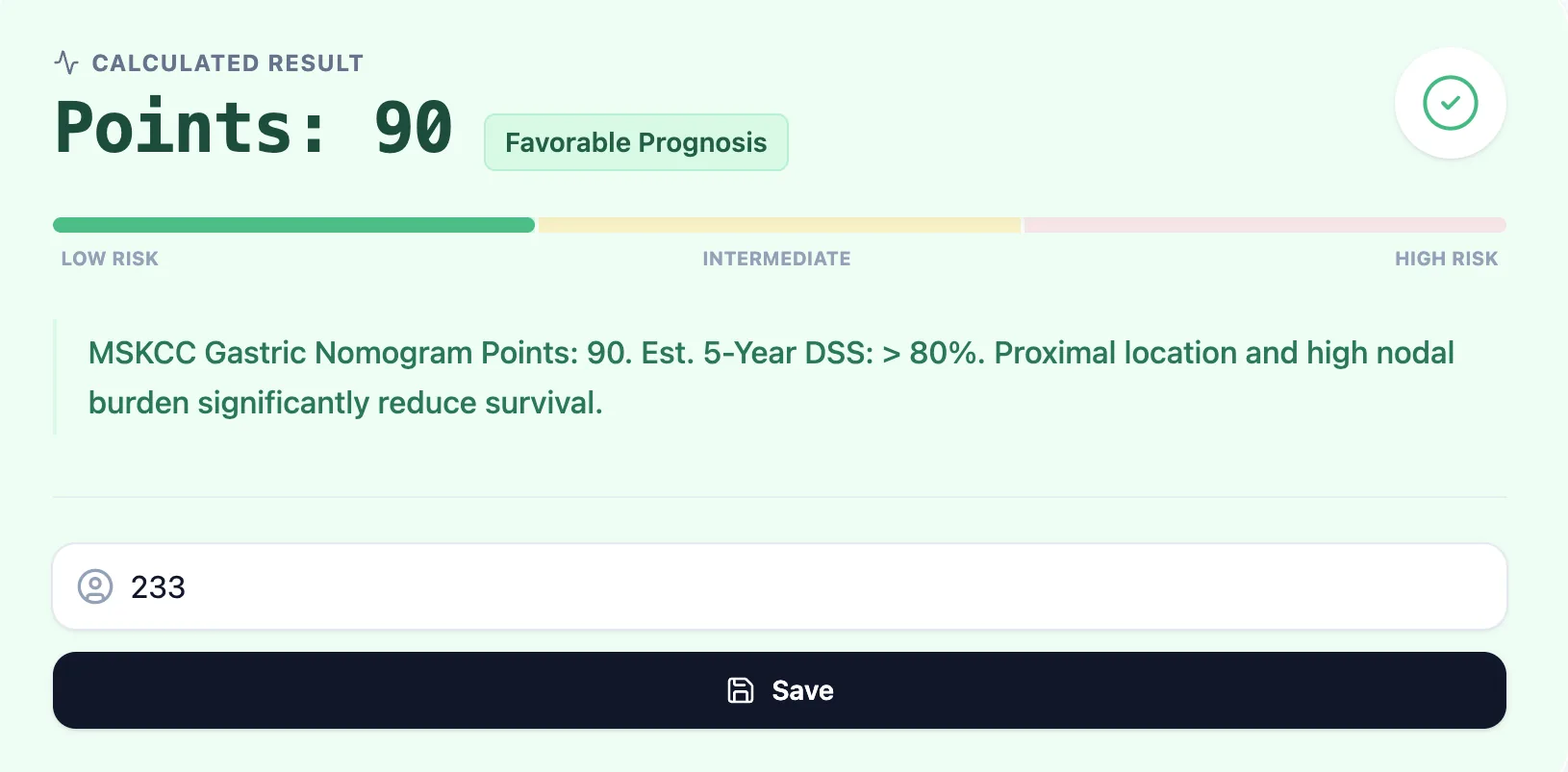
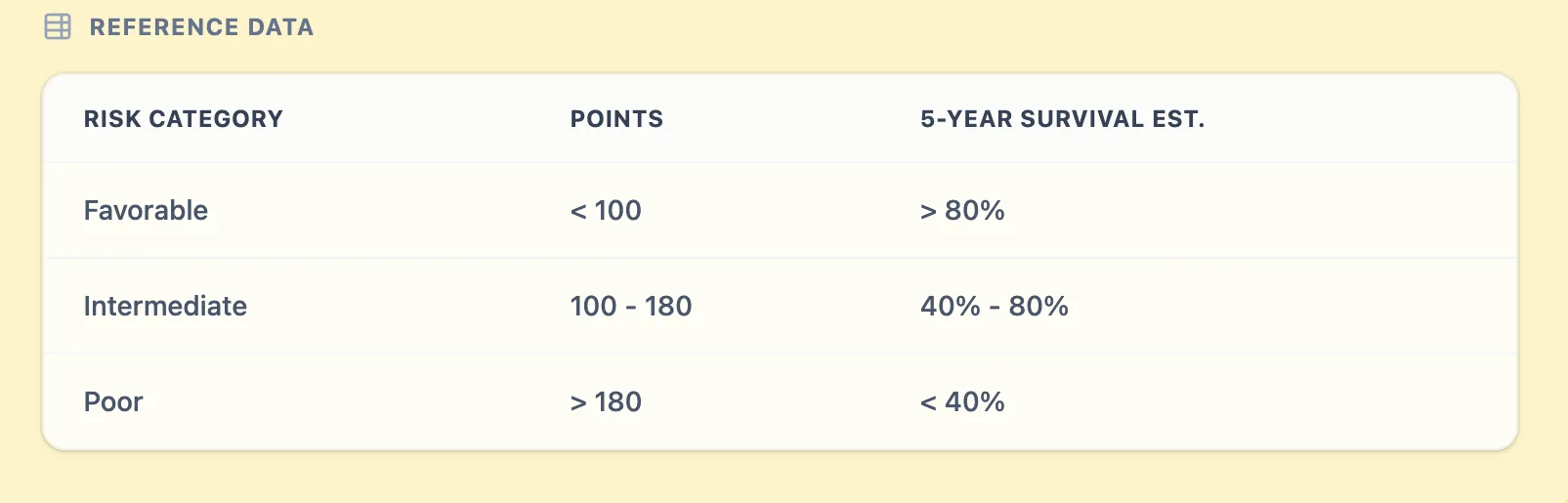
Example output results and point reference table
6. Integrating OncoToolkit into Clinical Care
6.1 Routine Clinical Decision Making
In daily practice, specialists can use the MSKCC nomogram calculator to:
- Add quantified survival estimates to post‑operative consultations.
- Stratify cases before MDT meetings.
- Communicate precisely with referring centres.
6.2 Surgical Education
For fellows and residents, the calculator doubles as a simulation tool: by adjusting T‑stage and nodal ranges, trainees can see how each factor reshapes DSS in real-time.
7. Clinical FAQ: Frequently Asked Questions
Can the MSKCC Gastric Cancer Nomogram be used in patients who received neoadjuvant therapy?
It generally should not be used in this setting. The original nomogram was derived from patients undergoing upfront R0 resection without perioperative chemotherapy.20, 2
8. Conclusion
The MSKCC Gastric Cancer Nomogram is one of the most thoroughly validated tools for estimating post‑resection DSS in primary gastric adenocarcinoma. OncoToolkit translates this evidence into a fast, intuitive calculator designed to reduce cognitive load and “calculator fatigue” for upper GI and pancreatic teams.8, 10, 1
Ready to Simplify Your Post-Resection Prognostic Workflow?
Calculate 5-year disease-specific survival in seconds with our validated MSKCC Gastric Nomogram tool.
Use the MSKCC Nomogram Calculator
Free to use. No registration required.
References
- Kattan MW, et al. Postoperative Nomogram for Disease-Specific Survival After R0 Resection for Gastric Carcinoma. J Clin Oncol. 2003. Source
- Ji X, et al. Individualized prognosis prediction for patients with gastric cancer. Ther Adv Med Oncol. 2020. Source
- NCCN Guidelines. Gastric Cancer. 2021. Source
- Kattan MW, et al. Nomogram for disease-specific survival after resection for gastric carcinoma. J Clin Oncol. 2003. Source
- MSKCC. Gastric Cancer Nomogram. Source
- MSKCC. Prediction Tools for Stomach Cancer. Source
- He J, et al. Limitations of AJCC TNM staging in gastric cancer. Front Oncol. 2018. Source
- Chen J, et al. Validation of the MSKCC gastric cancer nomogram in a Chinese population. Br J Cancer. 2013. Source
- Novotny A, et al. Prediction of individual survival after surgery for adenocarcinoma of the stomach. Ann Surg. 2006. Source
- Nakauchi M, et al. Revalidation of the MSKCC Nomogram for Gastric Cancer in the Modern Era. Ann Surg Oncol. 2022. Source
- Lamb BW, et al. Decision making fatigue in multidisciplinary cancer teams. BMC Health Serv Res. 2019. Source
- Soukup T, et al. Cognitive load and team performance in cancer MDTs. BMC Health Serv Res. 2022. Source
- Shortliffe EH, et al. Clinical Decision Support: The Road Ahead. Health Aff. 2020. Source
- Sutton RT, et al. An overview of clinical decision support systems. npj Digit Med. 2020. Source