RMH Score (Phase I Trials) Calculator: A Practical Guide for Oncologists
Selecting the right patient for an early‑phase oncology trial is clinically and ethically demanding. Estimate survival and risk categories for Phase I oncology trial candidates in seconds with the RMH Score calculator.

1. RMH Score (Phase I Trials): A Practical Guide for Oncologists
Selecting the right patient for an early‑phase oncology trial is clinically and ethically demanding. Prognosis is often poor, prior therapies are extensive, and trial slots are limited, so investigators must balance scientific opportunity with realistic expectations for benefit. The RMH Score (Phase I Trials) provides an objective, easy‑to‑apply prognostic framework that helps stratify patients by expected overall survival using three routinely available laboratory and disease burden markers. [1], [2], [3]
At OncoToolkit, we’ve built an RMH Score (Phase I Trials) calculator that turns this evidence‑based score into a rapid, mobile‑friendly decision support tool for everyday use in clinics, trial units, and multidisciplinary team (MDT) meetings. The tool reduces calculator fatigue by surfacing just the essential inputs and mapping them directly to risk groups and median survival estimates that you can interpret at a glance. [1]
2. Understanding the RMH Score (Phase I Trials) Criteria
The Royal Marsden Hospital (RMH) Score is a prognostic model originally developed to predict overall survival in adults with advanced solid tumors being considered for oncology Phase I trials. It incorporates three binary variables: [2]
- Serum albumin < 35 g/L
- Lactate dehydrogenase (LDH) above the upper limit of normal (ULN)
- More than two sites of metastatic disease [4], [2]
Each adverse factor scores 1 point, yielding a total RMH Score between 0 and 3. Patients with 0–1 factors are categorized as having a good prognosis, while those with 2–3 factors are considered to have a poorer prognosis with significantly shorter median overall survival. [2], [1]
Historically, the RMH score was derived from retrospective analyses of patients treated on Phase I studies at a single institution and then prospectively validated in an independent cohort. It has since been externally validated in multiple centers and tumor types, including large Phase I programs and disease‑specific cohorts, and has become one of the most widely referenced prognostic tools for early‑phase oncology trials. [5], [6], [7], [2]
3. Clinical Importance of the RMH Score in Oncology Practice
In modern oncology, Phase I trials increasingly enroll patients who may derive meaningful benefit from targeted therapies and immunotherapies rather than purely undergoing dose‑finding for cytotoxics. Still, typical patients in these trials have heavily pretreated, metastatic disease with limited life expectancy, making accurate risk stratification crucial. The RMH Score offers several practical advantages: [8], [3], [6]
- •Objective patient selection – It uses routine labs and metastasis count, reducing subjective bias in estimating prognosis and “fitness” for Phase I enrollment. [9], [2]
- •Risk‑aligned trial discussions – Low‑risk patients (score 0–1) have longer median survival, allowing more time for trial participation and potential benefit, whereas high‑risk patients (score 2–3) may be better served by symptom‑directed care or very carefully selected protocols. [6], [2]
- •Harmonized eligibility across centers – Using a common prognostic framework facilitates consistent screening criteria and comparability across trials and institutions. [10], [5]
Without digital support, applying the RMH Score still requires checking laboratory ranges, counting metastatic sites across multiple imaging reports, and cross‑referencing the original validation studies to interpret risk groups. On our platform, this entire workflow is condensed into a single mobile‑responsive interface optimised for use at the bedside, in outpatient clinics, or during MDT discussions. [1]
4. Clinical Evidence and Validation of the RMH Score
4.1. The logic behind the score
The RMH Score was developed using multivariate Cox proportional hazards models in patients enrolled on oncology Phase I trials. Albumin < 35 g/L, LDH above ULN, and more than two metastatic sites were each independent predictors of worse survival, and the investigators assigned 1 point to each factor to create a simple additive score from 0 to 3. In other words, the underlying statistics are sophisticated, but the operational form is a straightforward point‑based index. [2]
On our calculator, this logic is implemented transparently: each “Yes” response to an adverse factor adds one point, and the sum determines the prognostic group and associated median overall survival. This mirrors the original publications while making the math fully reproducible and auditable for clinical and research use. [1], [2]
4.2. Validation across Western and Asian populations
In Western Phase I populations, the original prospective validation at the Royal Marsden Hospital showed that patients with an RMH Score of 0–1 had a median overall survival of about 33 weeks, whereas those with scores of 2–3 had a median survival around 16 weeks. Large external series from North American centers, including the MD Anderson Phase I program and disease‑specific cohorts such as metastatic renal cell carcinoma and bone sarcoma, confirmed that higher RMH scores independently predict shorter survival and help select patients more likely to benefit from trial participation. [11], [5], [6], [2], [1]
More recently, Asian Phase I programs have evaluated whether the RMH Score retains prognostic value in their settings. A large Asian study from the National University Cancer Institute, Singapore (NCIS) reported that increasing RMH scores were associated with progressively worse overall survival, with hazard ratios rising from approximately 1.3 for a score of 1 to over 4 for a score of 3 compared with score 0. Another analysis of Asian patients treated in Phase I trials found that those with RMH scores of 2–3 had significantly shorter overall survival than those with scores 0–1 (4.9 vs 13.9 months; HR ≈ 2.7). [12], [13]
Taken together, these data suggest:
- In Western cohorts, the RMH Score provides clear discrimination between good‑ and poor‑risk groups and has been validated across multiple tumor types and institutions. [5], [11], [6], [2]
- In Asian cohorts, the same three variables—albumin, LDH, and number of metastatic sites—remain prognostic, and the direction and magnitude of risk gradients are broadly comparable, supporting the score’s international applicability. [14], [13], [12]
However, Asian investigators have also developed region‑specific extensions, such as the NCIS score that builds on RMH variables with additional markers, reflecting potential differences in case mix and treatment patterns. [14], [12]
4.3. Role in guidelines and contemporary practice recommendations
While most major oncology guidelines stop short of mandating a specific prognostic score, several expert reviews and consensus documents on Phase I trial design and patient selection explicitly highlight the RMH Score as a validated tool to support risk‑benefit assessment. Reviews of Phase I methodology and early‑phase immuno‑oncology trials discuss RMH and MDACC scores as standard benchmarks for estimating life expectancy and recommend their use alongside performance status and disease kinetics when considering enrollment. [15], [3], [9], [8], [10]
5. Expansions, Modifications, and Related Scores
Since the original RMH publication, several important expansions and related models have emerged:
- RMH + NLR50 – Investigators at the Marsden incorporated neutrophil–lymphocyte ratio (NLR) into the RMH framework, creating an RMH + NLR50 score that showed improved discrimination for survival in Western Phase I populations. [12]
- MDACC prognostic score (MDAS) – This model adds ECOG performance status and gastrointestinal tumor type to the three RMH variables, generating a 0–4 point scale; in some analyses, MDACC offers limited incremental benefit over RMH alone, especially in sarcoma cohorts. [6], [5]
- Immunotherapy‑focused scores (GRIm, LIPI) – As immuno‑oncology expanded, scores such as the Gustave Roussy Immune Score (GRIm) and the Lung Immune Prognostic Index (LIPI) were developed, integrating LDH, albumin, and inflammatory markers (e.g., NLR) to predict outcomes in IO Phase I trials. [16], [17], [15]
- Region‑specific adaptations – Asian centers have proposed models that combine RMH variables with factors like NLR to better fit local populations, such as the NCIS score mentioned earlier. [14], [12]
6. How the OncoToolkit RMH Score calculator works
On our platform, the RMH Score (Phase I Trials) calculator is designed for rapid, intuitive use with minimal clicks. The main input page presents the three score components with clear binary options. [1]
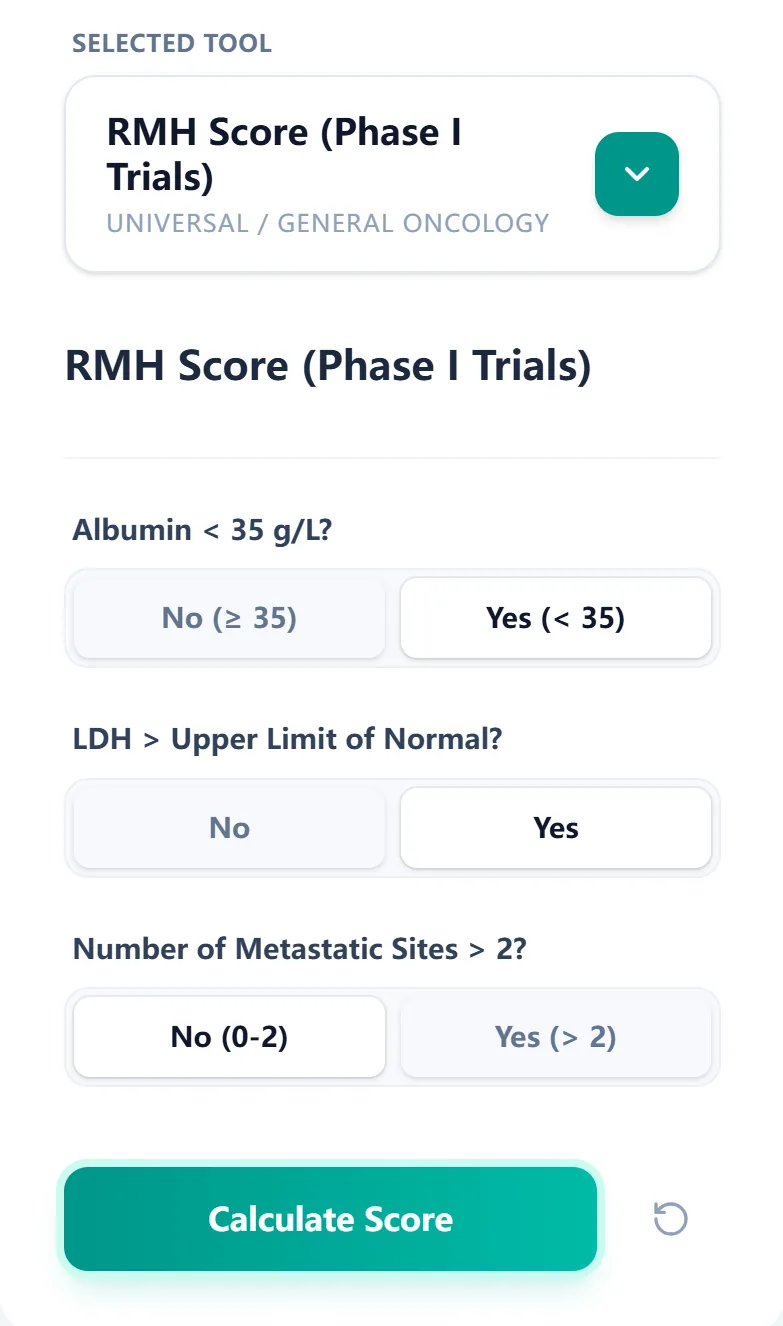
Figure 1. The input screen guides the clinician through the three RMH variables—albumin, LDH, and number of metastatic sites—using simple yes/no buttons.
To calculate the score, you simply:
- Indicate whether serum albumin is < 35 g/L.
- Indicate whether LDH is above the laboratory upper limit of normal.
- Indicate whether the patient has more than two metastatic sites.
Each adverse answer adds 1 point; non‑adverse answers add 0 points. When you click Calculate Score, the tool instantly displays: [2], [1]
- The total RMH Score (0–3).
- The corresponding risk group (good vs poor prognosis).
- An interpreted median overall survival estimate based on reference data. [1]

Figure 2. The reference table links RMH Score ranges to median overall survival.
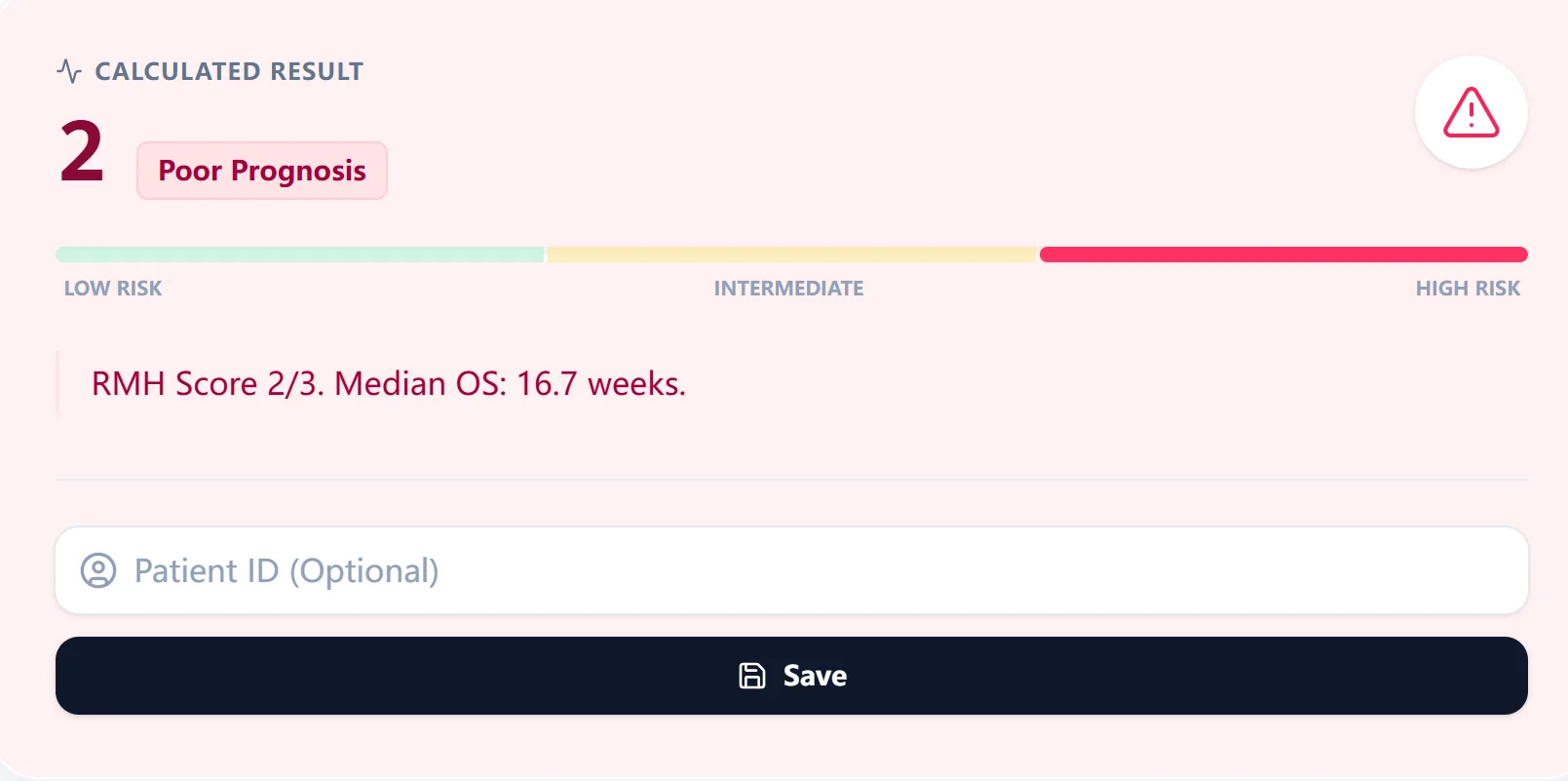
Figure 3. The result view summarizes the total RMH Score, risk label, and median overall survival in a color‑coded bar.
7. Clinical Context, Background, and Transparency
The calculator includes a concise clinical context panel summarizing the origin and intended use of the RMH Score, along with the key source study and the logic behind the 0–3 point system. [2], [1]
Figure 4. The embedded clinical background panel recaps how the RMH Score was derived.
8. Using the calculator in clinical care, education, and research
8.1. Specific use cases for the OncoToolkit RMH Score calculator
OncoToolkit’s implementation of the RMH Score supports several concrete clinical applications:
- Referral triage for Phase I clinics – General oncologists can quickly screen advanced cancer patients in outpatient settings and identify those who meet local prognostic thresholds for Phase I referral. [10], [2]
- MDT and early‑phase board discussions – During tumor boards or early‑phase review meetings, the calculator provides a shared, standardized prognostic estimate that complements performance status and organ function assessments.
- Trial selection within a Phase I unit – For centers running multiple studies, the score can be used to match lower‑risk patients to more intensive or longer‑cycle protocols, while reserving shorter or less toxic regimens for those with higher scores. [11], [6]
- Documentation and patient counseling – The structured output and survival estimate can be summarized in clinic letters or electronic records to support transparent risk communication with patients, families, and referring physicians. [9]
8.2. Education and simulation for trainees
For fellows and residents, the RMH Score calculator functions as a teaching aid in several ways:
- Reinforces the prognostic importance of albumin, LDH, and metastatic burden in advanced cancer. [6], [2]
- Allows rapid what‑if simulations during case conferences.
- Encourages a structured, measurable approach to Phase I referrals. [3]
8.3. Research, quality improvement, and outcomes analysis
OncoToolkit is designed to move clinicians from single‑patient calculations to aggregated insights. By saving RMH Scores over time and across patients, centers can:
- Audit consistency in Phase I referral patterns. [9], [6]
- Pair baseline RMH Scores with survival outcomes to benchmark institutional performance. [13], [12], [6]
- Compare RMH‑based stratification with newer models such as MDACC, GRIm, LIPI, or RMH + NLR50. [15], [12]
9. Clinical FAQ
When should you not use the RMH Score (Phase I Trials)? The RMH Score should not be the sole determinant of trial eligibility in patients whose clinical situation is dominated by acute issues that are not captured in albumin, LDH, or metastasis counts—for example, rapidly progressive organ failure, uncontrolled infections, or severe performance status impairment. [18], [6]
How does the RMH Score compare with other prognostic models like MDACC, GRIm, or LIPI?
The RMH Score is one of the earliest and simplest Phase I prognostic tools. Later models incorporate additional factors and may offer better discrimination in some immunotherapy cohorts, although they are more complex. Many centers still use the RMH Score alongside these newer indices because of its simplicity and deep validation history. [17], [5], [11], [12], [16], [15], [6], [2]
Can the RMH Score be used in patients with significant comorbidities or poor performance status?
In patients with substantial comorbid disease or ECOG PS ≥2, the RMH Score may underestimate true risk. Clinicians should interpret the result cautiously and integrate broader clinical information. [11], [6], [2]
10. Call to Action: Integrate the RMH Score into Your Workflow
To explore the RMH Score (Phase I Trials) calculator, visit: https://oncotoolkit.com/calculator/rmh-clinical-trial. We recommend bookmarking the page on your clinic workstation and mobile device.
At OncoToolkit, our goal is to reduce cognitive load for oncologists by turning validated prognostic scores into fast, transparent calculators that fit naturally into MDTs, academic discussions, and patient consultations. [17], [10], [16], [15]
Ready to Simplify Your Phase I Stratification?
Calculate the RMH Score in seconds and provide evidence-based prognostic estimates for your patients.
Use the RMH Calculator Now
Free to use. No registration required.