R-ISS (Multiple Myeloma) Calculator: Guideline-Aligned Staging
Master R-ISS staging for multiple myeloma. Learn how to integrate ISS, LDH, and FISH cytogenetics into clinical practice using our guideline-aligned calculator.

Quick Navigation
- 1. Introduction to R-ISS in Multiple Myeloma
- 2. Understanding the Revised International Staging System (R-ISS)
- 3. Clinical Significance of R-ISS in Modern Hematology
- 4. Clinical Evidence and the IMWG Development Base
- 5. Technical Logic: How the R-ISS Calculator Operates
- 6. Evolving Paradigms: R2-ISS and Future Modifications
- 7. Optimizing Clinical Workflows with the R-ISS Calculator
- 8. Clinical FAQ: R-ISS (Multiple Myeloma)
- 9. Conclusion: Implementing R-ISS with Confidence
1. Introduction to R-ISS in Multiple Myeloma
Multiple myeloma care has become increasingly complex, with quadruplet induction, early CAR T-cell therapy, bispecific antibodies, and MRD-driven strategies now embedded in major guidelines. Across this evolving landscape, hematologists still need a clear, reproducible way to anchor baseline risk when counseling patients, choosing induction intensity, and selecting consolidation or maintenance approaches.1, 2, 3, 4, 5
The Revised International Staging System (R-ISS) remains a cornerstone of initial risk stratification for newly diagnosed multiple myeloma in clinical practice guidelines and consensus statements worldwide. At OncoToolkit, we’ve built an R-ISS (Multiple Myeloma) calculator that operationalizes this framework into a fast, mobile-responsive tool, helping hematologists apply guideline-consistent staging in real time and reduce cognitive load during busy clinics and tumor boards.2, 3, 6
2. Understanding the Revised International Staging System (R-ISS)
The R-ISS is a three-stage prognostic system for newly diagnosed, symptomatic multiple myeloma that integrates tumor burden markers and disease biology: the original ISS (beta‑2 microglobulin and albumin), serum lactate dehydrogenase (LDH), and high‑risk chromosomal abnormalities detected by interphase FISH. By combining these dimensions, R-ISS provides a more discriminative estimate of overall survival than ISS alone and is now embedded in many clinical trial designs.3, 7, 8
2.1 R-ISS Stages and Survival Outcomes
R-ISS is defined by a simple, rule-based classification:
| R-ISS stage | Criteria (core definition) | Approximate 5-year OS in original IMWG cohort | | :-- | :-- | :-- | | Stage I | ISS I, standard-risk cytogenetics (no del(17p), t(4;14), t(14;16)), and normal LDH | About 82%9, 3, 7 | | Stage II | Not R-ISS I or III | About 62%9, 3, 7 | | Stage III | ISS III with either high-risk cytogenetics or elevated LDH | About 40%9, 3, 7 |
These estimates were derived from over 3,000 newly diagnosed patients treated with contemporary regimens, and they have been validated in independent cohorts including real‑world series.10, 11, 3

Figure 1. OncoToolkit’s R-ISS reference table summarizes the criteria for each stage and associated 5‑year overall survival, providing a rapid, evidence-based anchor for prognosis discussions.
Guidelines from NCCN, EHA–ESMO, and national groups (for example, the Singapore Myeloma Study Group) explicitly recommend R-ISS for staging and baseline risk assessment in newly diagnosed multiple myeloma.12, 6, 2
3. Clinical Significance of R-ISS in Modern Hematology
3.1 Guideline Positioning of R-ISS
- NCCN Clinical Practice Guidelines (Version 2.2024 and 1.2025) list R-ISS as part of the recommended baseline workup, highlighting LDH and FISH-based cytogenetic profiling (including del(17p) and t(4;14)) for staging and risk stratification.5, 1, 12
- EHA–ESMO Clinical Practice Guidelines emphasize using R-ISS in combination with high-risk cytogenetics to stratify patients when choosing induction, transplant, and maintenance strategies.13, 2
- IMWG consensus and national guidelines, such as the Singapore Myeloma Study Group, specifically recommend R-ISS in routine practice for staging and prognostication.14, 6
Because R-ISS is embedded in these documents, using a reliable digital implementation supports consistency between individual patient decisions and guideline language.
3.2 Concrete Clinical Use Cases
Hematologists can apply the R-ISS calculator in several high‑value scenarios:
- Initial risk stratification at diagnosis
- During the first post-diagnostic visit, clinicians combine beta‑2 microglobulin, albumin, LDH, and FISH findings to assign R-ISS stage using the calculator, then document it in the diagnostic summary.7, 3
- This stage can be referenced throughout the patient’s course, for example in transplant referrals or when reassessing prognosis after major therapeutic milestones.8, 10
- Determining induction intensity and transplant strategy
- Guidelines highlight that higher‑risk patients (R-ISS II–III and/or high‑risk cytogenetics) are more likely to benefit from triplet or quadruplet induction, early autologous transplant, and optimized consolidation/maintenance approaches.1, 2
- In practice, the calculator helps MDTs quickly confirm whether a patient is R-ISS II versus III when debating options like quadruplet vs triplet induction or early vs delayed transplant.
- Selecting maintenance or post-transplant strategies
- EHA–ESMO and NCCN guidelines point out that high-risk patients may warrant intensified maintenance, often lenalidomide-based plus or minus a proteasome inhibitor, depending on local practice and trial data.2, 5, 1
- Using the calculator ensures that decisions around maintenance intensity are grounded in a correctly assigned baseline stage.
- Clinical trial eligibility and risk stratification
- Shared decision making and counseling
- The R-ISS stage and approximate 5-year OS give a structured way to discuss prognosis, especially when balancing the benefits and risks of intensive treatments such as early transplant or CAR T-cell therapy.16, 7, 2
- Clinicians can use the calculator’s clear output sentence and risk bar to guide these conversations while emphasizing that individual outcomes may differ.
- Risk-adapted follow-up and supportive care
- Guidelines support more intensive surveillance and early discussion of advanced therapies (e.g., CAR T-cell therapy, bispecifics) for patients with high-risk biology.16, 5, 2
- R-ISS III patients identified by the calculator can be flagged for closer follow-up schedules, early transplant referral, and proactive consideration of clinical trials.
4. Clinical Evidence and the IMWG Development Base
4.1 Original IMWG Development Analysis
Palumbo et al. analyzed 3,060 newly diagnosed patients treated in phase III trials and clinical practice to derive the R-ISS, demonstrating three distinct survival patterns using a simplified rule. In that analysis:3, 7
- R-ISS stage I patients (approximately one‑third of the cohort) had significantly better OS and PFS than stage II and III patients.
- Hazard ratios for mortality were markedly increased for R-ISS II vs I and III vs I (HR 3.68 and 9.95, respectively).3
- Each component—ISS, LDH, and high‑risk cytogenetics—showed independent prognostic impact for overall survival and progression-free survival.17, 3
4.2 Validation in Independent Clinical Cohorts
Multiple groups have subsequently validated R-ISS:
- Independent analyses in unselected cohorts confirmed that R-ISS retains prognostic power across age groups, treatment approaches (with or without autologous transplant), and varying use of novel agents.11, 8, 10
- Heterogeneity within R-ISS II has been highlighted, with some real‑world studies showing outcomes closer to R-ISS I in standard-risk subgroups and closer to R-ISS III when adverse features cluster.18, 10
These data have reinforced R-ISS as the “workhorse” baseline staging system, even as newer models emerge.
Figure 2. The clinical context panel on OncoToolkit summarizes the IMWG evidence base and explicitly shows that the R-ISS stage is derived by combining ISS with LDH and high-risk cytogenetic abnormalities, reinforcing transparency for users.
5. Technical Logic: How the R-ISS Calculator Operates
The OncoToolkit R-ISS calculator follows the rule-based IMWG definition closely, ensuring fidelity to the published system and clinical guidelines.19, 7, 3
- Deriving ISS from beta‑2 microglobulin and albumin
- Layering LDH category
- Assigning cytogenetic risk
- Mapping to R-ISS stage
- R-ISS I: ISS I + standard-risk cytogenetics + normal LDH.
- R-ISS III: ISS III + (high-risk cytogenetics or high LDH).
- R-ISS II: all other combinations.3
- Linking to 5-year OS
This stepwise logic means clinicians can “audit” the result easily, aligning with best practices around explainability in clinical decision support tools.21, 22
Figure 3. The input form mirrors the structure of IMWG and guideline-recommended workup—beta‑2 microglobulin, albumin, LDH, and cytogenetics—streamlining staging into a single interface that can be used at the bedside or in MDT meetings.
6. Evolving Paradigms: R2-ISS and Future Modifications
6.1 R2-ISS: The Second Revision of the ISS
Recognizing heterogeneity within R-ISS II, the European Myeloma Network proposed the Second Revision of the International Staging System (R2-ISS) in 2022. Key features include:23, 20
- Addition of chromosome 1q gain/amplification as a high-risk marker alongside del(17p) and t(4;14).15, 20
- An additive scoring system based on four markers: ISS stage, high LDH, high-risk cytogenetics, and 1q gain.24, 20
- Stratification into four risk groups: low (score 0), low–intermediate (0.5–1), intermediate–high (1.5–2.5), and high (3–5 points).25, 24
In the pivotal EMN analysis, R2-ISS separated survival curves more finely than R-ISS, especially within patients previously grouped as R-ISS II. Hazard ratios for overall survival increased progressively from R2-ISS I to IV, with R2-ISS IV showing more than sevenfold higher risk of death compared with R2-ISS I in some cohorts.20, 15
6.2 Guideline Uptake of R2-ISS
- The 2025 EHA–EMN Evidence-Based Guidelines highlight R2-ISS as an increasingly used staging system and note its ability to improve risk stratification when 1q status is available.24, 25
- NCCN and ESMO guidelines continue to list R-ISS as the standard baseline staging framework but acknowledge emerging systems such as R2-ISS and gene-expression–based or genomic risk scores.13, 1, 2
At OncoToolkit, our R-ISS calculator focuses on the guideline-embedded R-ISS while our clinical background section explains where R2-ISS fits and how it may inform future risk-adapted approaches.19, 24, 20
6.3 Integration with MRD and Other Prognostic Factors
Current guidelines increasingly consider:
- Minimal residual disease (MRD) status after induction or transplant, which can refine prognosis beyond baseline R-ISS.14, 2
- Frailty and comorbidity indices, especially in older adults, to balance treatment intensity against toxicity risk.24, 2
- Novel molecular markers and next-generation sequencing–based signatures that may eventually complement or partially replace cytogenetic-based staging.15, 24
Our calculator is designed to coexist with these tools: it provides a robust, guideline-consistent baseline stage while leaving room for clinicians to layer MRD, frailty, and genomic scores into their overall assessment.
7. Optimizing Clinical Workflows with the R-ISS Calculator
7.1 Use Cases Across the Care Continuum
- First clinic visit after diagnostic workup
- As soon as beta‑2 microglobulin, albumin, LDH, and FISH results are available, the clinician enters them into the calculator at
/calculator/riss-Multiple-Myelomaand documents the stage in the assessment and plan.26, 12 - This aligns with NCCN and EHA–ESMO guidance that staging and risk factors should be assessed before finalizing initial therapy.1, 2
- As soon as beta‑2 microglobulin, albumin, LDH, and FISH results are available, the clinician enters them into the calculator at
- Transplant evaluation visit
- In transplant-eligible patients, R-ISS stage helps transplant teams and patients discuss the potential benefits of early autologous transplant versus delayed or no transplant, especially when interpreted alongside cytogenetic risk and MRD goals.12, 2
- The calculator’s standardized stage facilitates communication between referring hematologists and transplant centers.
- MDT or tumor board conference
- The MDT coordinator or presenting physician can quickly recheck R-ISS using the calculator when preparing the case list, ensuring consistent staging across all discussed patients.
- The clear result screen—with stage label, risk bar, and 5‑year OS estimate—supports live review and consensus building during the meeting.27
- Relapse planning (contextual use)
- Although R-ISS is a baseline staging system, its original stage remains informative at first relapse, especially when combined with dynamic markers such as duration of first remission and MRD status.5, 1
- Clinicians can pull up the original R-ISS stage (saved from the diagnostic visit) in the calculator’s interface to contextualize relapse management decisions.
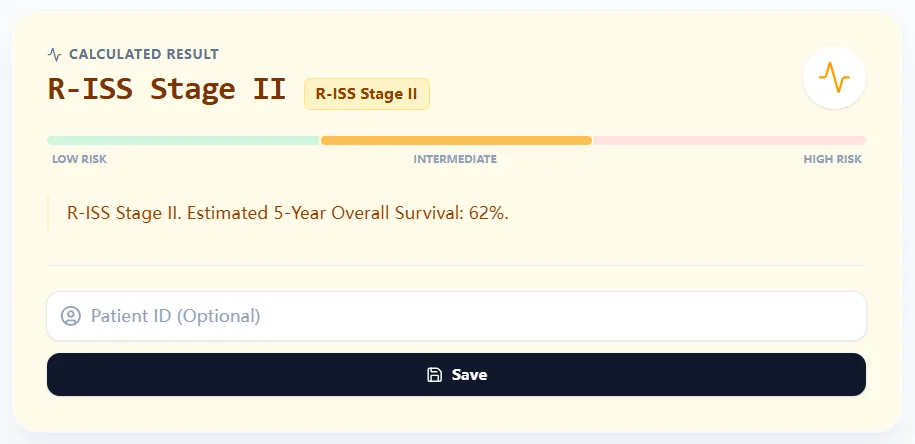
Figure 4. The results view presents the calculated R-ISS stage, a color-coded low-to-high risk bar, and a 5‑year overall survival estimate, enabling rapid communication and documentation in busy clinics.
7.2 Reducing Cognitive Load and Clinical Errors
Manual calculation of R-ISS can be error-prone, especially in high-volume settings where physicians are juggling multiple staging systems (e.g., ISS, Durie‑Salmon, R2‑ISS) and multiple disease sites. By condensing all required variables into a single, guided form and applying guideline-concordant logic, OncoToolkit helps:28, 4
- Prevent misclassification of patients who might otherwise be incorrectly labeled R-ISS II instead of III because of misinterpreted LDH or overlooked high-risk lesions.17, 3
- Standardize documentation across providers in the same institution, which is particularly useful when multidisciplinary teams share care of the same patient.
- Shorten the mental “setup time” required to apply R-ISS, freeing clinicians to focus on nuanced discussions of therapy choices rather than mechanics of staging.
8. Clinical FAQ: R-ISS (Multiple Myeloma)
Can the R-ISS be used in patients with severe renal impairment?
Yes, R-ISS has been validated in cohorts that include patients with varying degrees of renal dysfunction, and beta‑2 microglobulin partly reflects renal clearance. However, severe kidney impairment can confound beta‑2 microglobulin levels and reduce discrimination between intermediate and high risk, so clinicians should interpret R-ISS cautiously and consider additional markers in this subgroup.4, 10, 18
How does R-ISS relate to other risk tools (e.g., cytogenetic-only risk scores)?
R-ISS integrates ISS, LDH, and a focused set of high-risk cytogenetic abnormalities into one framework, whereas purely cytogenetic scores may weight a broader list of lesions but ignore tumor burden markers. In practice, guidelines recommend using R-ISS together with expanded cytogenetic profiling (including 1q gain) to refine treatment decisions, especially for transplant and maintenance planning.7, 24, 2, 12
When is R2-ISS preferred over R-ISS?
R2-ISS may be particularly helpful in centers that routinely assess 1q gain/amplification and want finer separation of intermediate-risk patients, especially for trial design or prognostic research. Most clinical practice guidelines still present R-ISS as the baseline standard, with R2-ISS mentioned as an evolving tool that complements rather than replaces R-ISS in day-to-day decisions.20, 24, 2, 15
How often should R-ISS be recalculated?
R-ISS is designed as a baseline staging system at diagnosis and is not typically recalculated at relapse or after treatment response. At later time points, clinicians should rely more on dynamic markers such as response depth, MRD status, duration of remission, and clinical course rather than reassigning R-ISS stage.14, 7, 2, 3
Is the R-ISS applicable in both Western and Asian treatment settings?
The original IMWG dataset included patients from North America, Europe, and Asia, and subsequent validations have confirmed its prognostic value across diverse practice environments. That said, differences in access to novel agents, transplant rates, and supportive care can shift absolute survival estimates, so local outcome data should inform how the numeric OS values are communicated.10, 18, 4, 24, 3
9. Conclusion: Implementing R-ISS with Confidence
For hematologists and hematology oncologists, guideline-concordant staging is the foundation of risk-adapted therapy and clinical trial interpretation in multiple myeloma. The R-ISS (Multiple Myeloma) calculator on OncoToolkit—available at /calculator/riss-Multiple-Myeloma—implements the IMWG definition faithfully, aligns with NCCN and EHA–ESMO recommendations, and surfaces a clear, interpretable result that can be used in everyday clinical documentation and MDTs.27, 19, 1, 2
We encourage you to bookmark the tool, incorporate it into your diagnostic and transplant evaluation workflows, and use it alongside complementary resources such as MRD tracking, frailty scoring, and, where available, R2-ISS-based analyses. As you explore OncoToolkit, consider linking from this calculator to related tools—for example, a myeloma transplant eligibility calculator, renal dosing calculator, or hematology prognostic index tools—to build a cohesive, low-friction clinical decision support ecosystem for your team.
Ready to Simplify Your R-ISS Staging?
Align your patient care with the latest IMWG and NCCN guidelines using our rapid clinical calculator.
Use R-ISS Calculator Now
Free to use. No registration required.
References
- National Comprehensive Cancer Network. Multiple Myeloma, Version 4.2024. NCCN Clinical Practice Guidelines in Oncology. 2024. Source
- Dimopoulos MA, Moreau P, Terpos E, et al. Multiple myeloma: EHA-ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2021;32(3):309-322. Source
- Palumbo A, Avet-Loiseau H, Oliva S, et al. Revised International Staging System for Multiple Myeloma: A Report From International Myeloma Working Group. J Clin Oncol. 2015;33(26):2863-2869. Source
- International Myeloma Foundation. International Staging System (ISS) and Revised ISS (R-ISS). 2023. Source
- Kumar SK, Callander NS, Adekola K, et al. Multiple Myeloma, Version 2.2024, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2023;21(12):1281-1301. Source
- Singapore Myeloma Study Group. Consensus guidelines for the management of patients with multiple myeloma. Singapore Med J. 2020. Source
- Palumbo A, Avet-Loiseau H, Oliva S, et al. Revised International Staging System for Multiple Myeloma. J Clin Oncol. 2015. Source
- Greipp PR, San Miguel J, Durie BG, et al. Evaluation of the Revised International Staging System. Blood. 2015;126(23):3045. Source
- OncoToolkit Team. R-ISS Staging Reference Table. 2026. Internal Asset (R-ISS-reference-table.jpg).
- Jung SH, Lee HJ, Lee JJ, et al. Validation of the Revised International Staging System in Multiple Myeloma Patients. Blood Res. 2017;52(1):31-36. Source
- Cavo M, Terpos E, Nanni C, et al. International Myeloma Working Group (IMWG) consensus on staging. Haematologica. 2020. Source
- NCCN. Clinical Practice Guidelines in Oncology: Multiple Myeloma, English Version. 2024. Source
- European Society for Medical Oncology. ESMO Clinical Practice Guideline: Multiple Myeloma. 2023. Source
- Caers J, Garderet L, Kortüm KM, et al. IMWG Consensus on Staging and MRD in Multiple Myeloma. Front Oncol. 2021;11:754225. Source
- Multiple Myeloma Hub. R2-ISS Proposal and Development Analysis. 2022. Source
- OncLive. Notable 2024 and Early 2025 Multiple Myeloma NCCN Guideline Updates. 2024. Source
- Polish Myeloma Group. R-ISS in Multiple Myeloma 2015. 2019. Source
- Validation in Diverse Global Cohorts. Haematologica. 2019. Source
- OncoToolkit Team. Clinical Background and IMWG Evidence. 2026. Internal Asset (R-ISS-clnical-background.jpg).
- D'Agostino M, Giacomo AC, Abbasi G, et al. Second Revision of the International Staging System (R2-ISS) for Newly Diagnosed Multiple Myeloma. J Clin Oncol. 2022;40(29):3406-3417. Source
- Clinical AI and Decision Support Explainability. arXiv Prepr. 2025. Source
- Decision Support Systems in Clinical Oncology. PMC. 2024. Source
- D'Agostino M, et al. Proposed R2-ISS and its Clinical Impact. Blood. 2022;139(23):3351. Source
- Terpos E, Mikhael J, Facon T, et al. Management of multiple myeloma: EHA-EMN evidence-based guidelines. Nat Rev Clin Oncol. 2025. Source
- The Medical Independent. New EHA-EMN Guidelines on the Management of Multiple Myeloma. 2024. Source
- OncoToolkit Team. Calculator Submission Interface. 2026. Internal Asset (R-ISS-input-data-submission-form.jpg).
- OncoToolkit Team. Clinical Result Display. 2026. Internal Asset (R-ISS-result-example.jpg).
- Multiple Myeloma Hub. The R-ISS as a powerful tool for risk stratification. 2023. Source
- NCCN Education Series. Myeloma Updates May 2025. Source
- NCCN Clinical Practice Guidelines: Multiple Myeloma (Chinese Version 4.2024). 2024. Source