Prostate Cancer Risk Stratification: NCCN & CAPRA Integration
A comprehensive clinical guide on integrating NCCN risk groups, CAPRA scores, and Epstein criteria for localized prostate cancer management.

Quick Navigation
- 1. Introduction to Prostate Cancer Risk Stratification
- 2. Core Frameworks for Risk Stratification
- 3. The Clinical Importance of Accurate Stratification
- 4. Clinical Evidence and Mathematical Validation
- 5. Operationalizing the Prostate Cancer Risk Calculator
- 6. Supporting Clinical Care, Education, and Research
- 7. Clinical FAQ: Navigating Stratification Dilemmas
- 8. Integrating Risk Stratification into Multidisciplinary Care
1. Introduction to Prostate Cancer Risk Stratification
Prostate cancer remains one of the most common malignancies in men worldwide, yet it represents a spectrum of disease ranging from indolent tumors requiring no immediate intervention to aggressive cancers demanding multimodal therapy. For oncologists and urologists, the challenge lies not in the diagnosis itself, but in accurately stratifying patients to match treatment intensity with disease biology. Risk stratification systems help answer the fundamental question: which patients benefit from active surveillance, and which require definitive treatment or intensified therapy?
At OncoToolkit, we've built a master calculator that integrates three essential decision support tools—the NCCN Risk Stratification (v4.2024), the CAPRA score, and the Epstein criteria—into a single, mobile-responsive interface. This unified platform eliminates the cognitive burden of manually cross-referencing multiple staging systems during multidisciplinary team (MDT) meetings, point-of-care rounds, and treatment planning discussions. You can access our comprehensive calculator at /calculator/panel-prostate-risk.
2. Core Frameworks for Risk Stratification
Risk stratification in localized prostate cancer refers to the systematic classification of patients into prognostic categories based on clinical, biochemical, and pathological parameters collected at diagnosis. This process informs critical treatment decisions, from selecting candidates for active surveillance to determining the need for androgen deprivation therapy (ADT) alongside radiation or the extent of lymph node dissection during radical prostatectomy.
2.1 NCCN Risk Stratification (v4.2024)
The National Comprehensive Cancer Network (NCCN) guidelines provide the most widely adopted framework in North America for prostate cancer risk grouping. The 2024 version stratifies patients into six tiers: Very Low, Low, Favorable Intermediate, Unfavorable Intermediate, High, and Very High risk. These categories are determined by a hierarchical assessment of clinical T-stage, PSA level, Gleason Grade Group, and biopsy core involvement.[1], [2]
The distinction between favorable and unfavorable intermediate-risk disease is particularly important. Favorable intermediate-risk patients have only one intermediate-risk factor (Gleason Grade Group 2 OR PSA 10-20 OR clinical stage T2b-c) and fewer than 50% positive cores, while unfavorable intermediate patients have either Gleason Grade Group 3 disease, multiple intermediate-risk factors, or greater core involvement. This subdivision has been validated across multiple treatment modalities and significantly influences whether patients receive monotherapy or combination approaches.[3], [4]
2.2 CAPRA Score (UCSF)
The Cancer of the Prostate Risk Assessment (CAPRA) score, developed at the University of California San Francisco, offers a more granular, continuous risk assessment on a 0-10 point scale. The CAPRA score incorporates patient age at diagnosis, PSA, Gleason Grade Group, clinical T-stage, and the percentage of positive biopsy cores. Unlike categorical systems, the CAPRA score provides individualized risk estimates for biochemical recurrence, metastasis-free survival, and cancer-specific mortality across all treatment modalities—radical prostatectomy, radiation therapy, brachytherapy, and active surveillance.[5], [6], [7], [8]
Multiple international validation studies have confirmed the CAPRA score's discriminatory ability, with concordance indices ranging from 0.63 to 0.81 for predicting biochemical recurrence and higher values (0.85-0.88) for metastasis and cancer-specific mortality. Risk approximately doubles with every two-point increase in CAPRA score, making it particularly useful for patient counseling and shared decision-making.[6], [7]
2.3 Epstein Criteria for Active Surveillance
Originally described in 1994, the Epstein criteria define "clinically insignificant" prostate cancer suitable for active surveillance: clinical stage T1c, PSA density less than 0.15 ng/mL/cc, Gleason score 6 or less, two or fewer positive cores, and less than 50% involvement of any single core. While these stringent "very low-risk" criteria have historically guided active surveillance enrollment, contemporary evidence supports expanded inclusion criteria—particularly allowing up to four positive cores and 60% maximum core involvement—without compromising oncologic safety.[9], [10], [11]
Figure 1: Pathological and clinical landscape of prostate cancer risk assessment.
Clinical Pearl: The distinction between favorable and unfavorable intermediate-risk disease is a key pivot point for ADT escalation. Favorable cases may often avoid ADT, while unfavorable cases typically benefit from short-term hormone therapy.
3. The Clinical Importance of Accurate Stratification
Accurate risk stratification is not an academic exercise—it directly determines treatment recommendations that carry profoundly different quality-of-life implications. Recommending radical prostatectomy to a man with very low-risk disease exposes him unnecessarily to erectile dysfunction and incontinence risks, while misclassifying unfavorable intermediate-risk disease as favorable may result in undertreatment and early biochemical recurrence.
In busy clinical settings, manually calculating these scores is error-prone and time-consuming. During MDT meetings where multiple cases are reviewed sequentially, the ability to rapidly input parameters and receive simultaneous NCCN risk group, CAPRA score, and Epstein eligibility dramatically reduces "calculator fatigue." Studies have demonstrated that MDT discussions improve overall survival in metastatic castration-resistant prostate cancer patients, and the same multidisciplinary approach benefits localized disease management by ensuring all relevant specialists—urologic oncologists, radiation oncologists, radiologists, and pathologists—contribute to a unified treatment plan.[12]
The calculator's mobile-responsive design means it can be used at the point of care during clinic visits or on ward rounds, not just during scheduled MDT meetings. This immediacy supports real-time shared decision-making with patients, allowing clinicians to demonstrate how different clinical parameters influence risk stratification and discuss treatment trade-offs with transparent, evidence-based data.
4. Clinical Evidence and Mathematical Validation
4.1 NCCN Risk Groups: Hierarchical Categorization
The NCCN system employs a rule-based hierarchical logic rather than a mathematical formula. Patients are classified into the highest risk category for which they meet criteria. For example, a patient with clinical stage T3b disease automatically qualifies as Very High risk regardless of PSA or Gleason score. The key thresholds include:
- Very Low: T1c, Grade Group 1, PSA < 10, PSA density < 0.15, < 3 positive cores, ≤ 50% involvement per core
- Low: T1-T2a, Grade Group 1, PSA < 10 (not meeting Very Low criteria)
- Favorable Intermediate: One intermediate factor (Grade Group 2 OR PSA 10-20 OR T2b-c) AND < 50% positive cores
- Unfavorable Intermediate: Grade Group 3 OR ≥ 2 intermediate factors OR ≥ 50% positive cores
- High: T3a OR Grade Group 4-5 OR PSA > 20
- Very High: T3b-T4 OR Primary Gleason pattern 5 OR > 4 cores with Grade Group 4-5
This stratification has been prospectively validated in phase III randomized trials, demonstrating clear separation in biochemical recurrence-free survival, distant metastasis, and prostate cancer-specific mortality across risk groups. Validation studies confirm that 5-year biochemical recurrence-free survival ranges from approximately 95% in low-risk patients to less than 55% in high-risk patients.[13], [14], [1]
4.2 CAPRA Score: Weighted Point-Based System
The CAPRA score uses a simple additive model with integer weights derived from Cox proportional hazards regression analysis of the CaPSURE database:
- Age: 0 points (< 50 years), 1 point (≥ 50 years)
- PSA (ng/mL): 0 points (< 6), 1 point (6-10), 2 points (10.1-20), 3 points (20.1-30), 4 points (> 30)
- Gleason Grade Group: 0 points (Grade Group 1), 1 point (Grade Group 2-3), 3 points (Grade Group 4-5)
- Clinical T-stage: 0 points (T1-T2), 1 point (T3a)
- Percentage of positive cores: 0 points (< 34%), 1 point (≥ 34%)
The resulting score (0-10) stratifies patients into low-risk (0-2 points), intermediate-risk (3-5 points), and high-risk (6-10 points) categories. European multicenter validation in 1,296 patients demonstrated that 5-year progression-free survival declined from 100% in CAPRA 0-1 patients to 40% in CAPRA 7-10 patients, with a concordance index of 0.764. Importantly, the CAPRA score maintains predictive accuracy across diverse treatment modalities—a key advantage over nomograms developed for single treatment types.[15], [7], [5], [6]
4.3 Epstein Criteria: Binary Eligibility Assessment
The Epstein criteria represent a binary classification (meets criteria vs. does not meet criteria) rather than a scoring system. The underlying principle is that tumors meeting all five criteria have an extremely low probability of harboring occult high-grade disease or extraprostatic extension. Retrospective analyses of radical prostatectomy specimens have shown that patients meeting Epstein criteria have organ-confined disease in approximately 98% of cases.[9]
Contemporary data support expanding these criteria while maintaining oncologic safety. Studies examining active surveillance cohorts with up to four positive cores or 60% maximum core involvement (with preserved PSA density < 0.15 and Gleason 6) show no significant difference in biochemical recurrence-free survival compared to strict Epstein criteria patients. However, PSA density and Gleason score thresholds should not be liberalized—these parameters remain the strongest independent predictors of reclassification during surveillance.[11], [9]
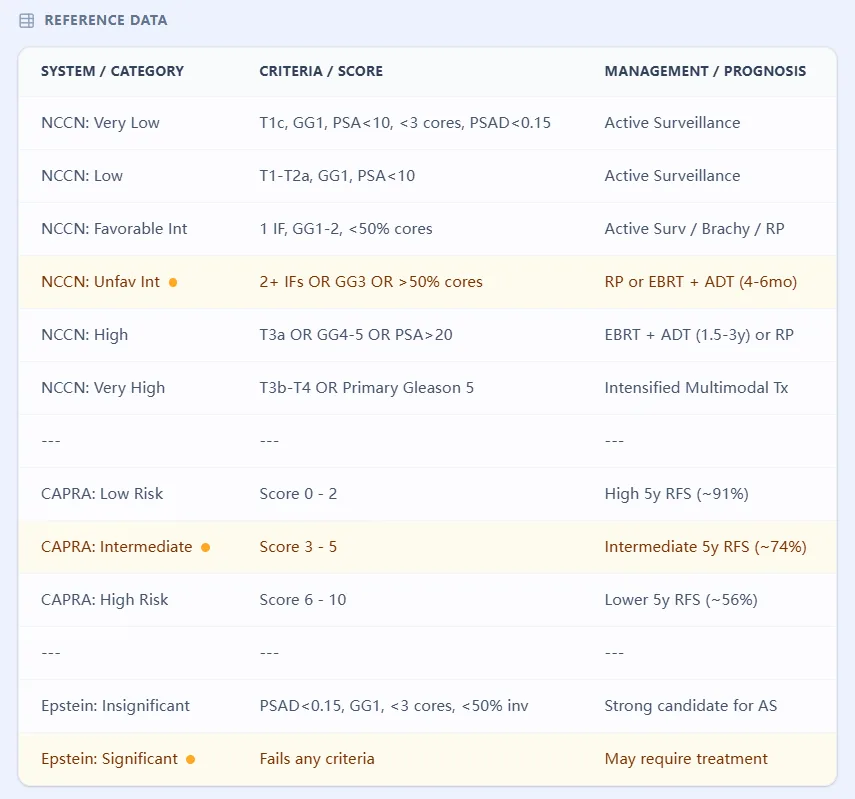
Figure 2: Summary reference for clinical risk thresholds across NCCN and CAPRA systems.
5. Operationalizing the Prostate Cancer Risk Calculator
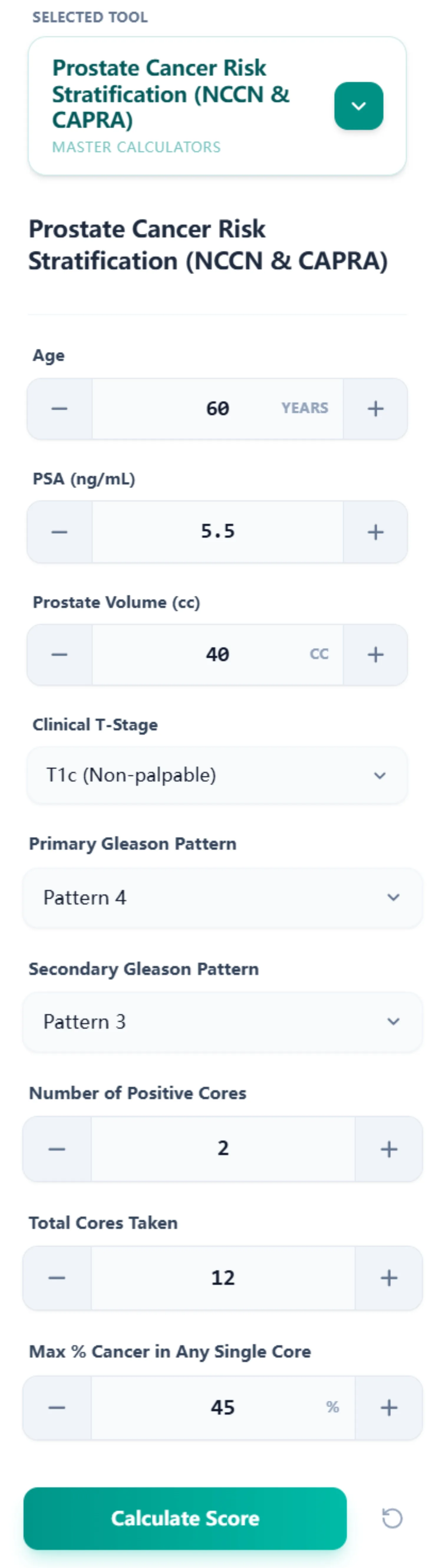
Our prostate cancer risk stratification calculator is designed for rapid data entry and transparent result interpretation. The input form captures eight discrete parameters:
- Age (years)
- PSA (ng/mL)
- Prostate Volume (cc) – for PSA density calculation
- Clinical T-Stage (T1c through T3b-T4)
- Primary Gleason Pattern (Pattern 3, 4, or 5)
- Secondary Gleason Pattern (Pattern 3, 4, or 5)
- Number of Positive Cores
- Total Cores Taken
- Maximum % Cancer in Any Single Core
These inputs are entered through an intuitive interface with increment/decrement buttons for numeric values and dropdown menus for categorical variables, minimizing data entry errors common with manual typing.
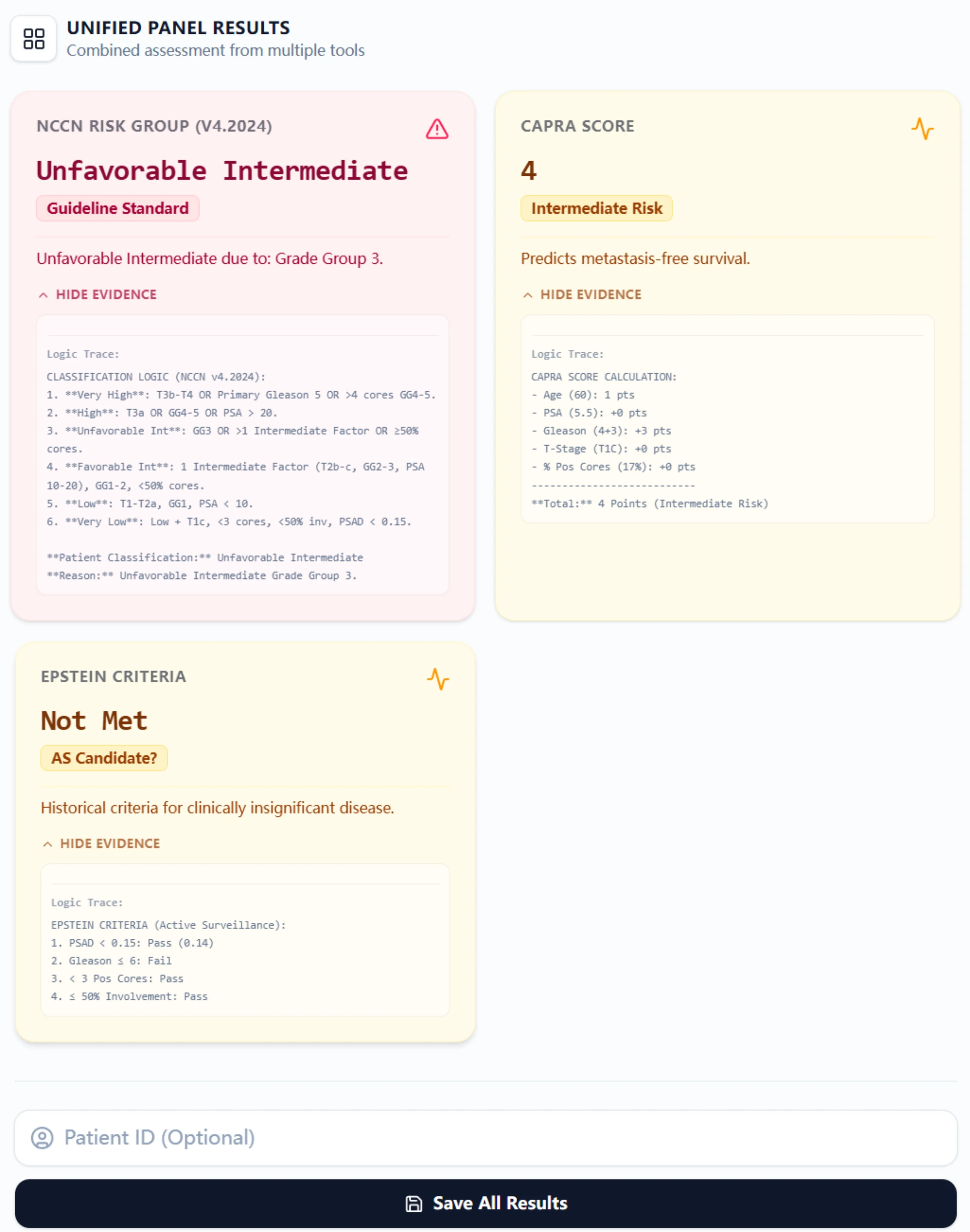
Upon clicking "Calculate Score," the platform generates a unified results panel displaying three color-coded cards:
5.1 NCCN Risk Group Interpretation
This card displays the assigned NCCN risk group (e.g., "Unfavorable Intermediate") with the specific reason for classification. For example, "Unfavorable Intermediate due to: Grade Group 3." An expandable "Logic Trace" section reveals the complete decision tree, showing how the algorithm evaluated each NCCN criterion step-by-step. This transparency is invaluable for teaching fellows and residents, as they can see precisely why a patient fell into a particular category.
5.2 CAPRA Score Evaluation
The CAPRA card shows the total point score (0-10) and the corresponding risk category (Low, Intermediate, or High). The "Logic Trace" breaks down point allocation by category: "Age (60): 1 pts; PSA (5.5): +0 pts; Gleason (4+3): +3 pts; T-Stage (T1c): +0 pts; % Pos Cores (17%): +0 pts; Total: 4 Points (Intermediate Risk)." This itemization helps clinicians identify which factors are driving risk and which parameters, if modified on follow-up assessments, might result in reclassification.
5.3 Epstein Criteria Analysis
The third card indicates whether Epstein criteria are met ("Pass" or "Fail"), with a secondary annotation for Active Surveillance candidacy. The logic trace specifies which criteria passed and which failed (e.g., "PSA < 0.15: Pass (0.14); Gleason ≤ 6: Fail; < 3 Pos Cores: Pass; ≤ 50% Involvement: Pass"). This granular feedback is particularly useful when patients narrowly miss active surveillance eligibility—it directs attention to parameters that could be reassessed (e.g., confirming PSA density with repeat imaging or considering MRI-targeted biopsies to clarify grade).
6. Supporting Clinical Care, Education, and Research
Routine Clinical Decision Support
The calculator's primary function is to provide rapid, accurate risk stratification at the point of care. During initial consultation following a prostate cancer diagnosis, clinicians can input biopsy and staging data in real time, generating immediate treatment pathway recommendations aligned with NCCN guidelines. The platform eliminates manual lookup of complex flowcharts and reduces the risk of miscalculation—particularly important for the CAPRA score, where multiple weighted variables must be summed correctly.
For MDT preparation, the calculator enables batch processing of multiple cases. Clinicians can quickly stratify an entire week's new diagnoses before the meeting, arriving with pre-populated risk assessments that accelerate discussion. The ability to save and export results (via the "Save All Results" button shown in the interface) facilitates documentation in electronic health records and supports quality assurance audits.
Education and Simulation for Trainees
Medical education increasingly emphasizes competency-based learning with real-time feedback. Our platform serves as an interactive teaching tool during didactic sessions and case-based learning. Fellows and residents can adjust individual parameters (e.g., incrementing the number of positive cores from 2 to 4) and immediately observe how the NCCN classification and CAPRA score change. This hands-on approach builds intuition about which variables exert the greatest influence on risk stratification.
The expandable logic traces function as a "show your work" feature, analogous to step-by-step solutions in mathematics education. Trainees learn not just the final risk category, but the reasoning pathway that led there. This transparency demystifies risk stratification systems and empowers junior clinicians to confidently explain classifications to patients.
Clinical Research and Quality Improvement
Consistent, reproducible risk stratification is essential for clinical trials and quality improvement initiatives. Many prostate cancer trials stratify randomization by NCCN risk group or CAPRA score; using a standardized calculator ensures uniform classification across participating institutions. The platform's ability to export results supports retrospective cohort analyses, enabling researchers to correlate risk stratification with treatment patterns and outcomes.
At OncoToolkit, we recognize that point-of-care decision support and population-level analytics are complementary needs. While this calculator focuses on individual patient assessment, our broader platform includes tools for aggregated survival analysis and outcomes dashboards. Clinicians can transition seamlessly from calculating a single patient's CAPRA score to analyzing recurrence-free survival curves across their entire practice cohort, stratified by risk group.
7. Clinical FAQ: Navigating Stratification Dilemmas
Can the NCCN Risk Stratification Be Used in Patients with Multiple Comorbidities?
Yes, the NCCN risk stratification is based solely on cancer-specific parameters and does not directly incorporate comorbidity burden. However, comorbidities profoundly influence treatment decisions within each risk group. NCCN guidelines explicitly recommend observation (rather than active treatment) for patients with life expectancy less than 5-10 years, regardless of risk group.[16]How Does the CAPRA Score Differ from the D'Amico Classification?
The D'Amico classification is a simple three-tier system, while the CAPRA score provides a 0-10 point scale with weighted variables. This allows for superior discrimination, particularly within the broad "intermediate-risk" category. Two patients classified as D'Amico intermediate might have CAPRA scores of 2 and 5, respectively, indicating significantly different prognoses.[17], [14], [5], [6]What is the Most Common Mistake When Calculating CAPRA Score?
The most frequent error is miscalculating the percentage of positive cores. The CAPRA score uses: (number of positive cores / total cores taken) × 100. Confusion often arises with "maximum percent involvement of a single core," which is relevant for Epstein criteria but NOT for CAPRA.Is the CAPRA Score Validated for Western or Asian Populations?
The CAPRA score is highly validated in North American and European cohorts with concordance indices of 0.78-0.81.[5], [6] Data in Asian populations are more limited; clinicians should interpret results in the context of local validation data, as prostate cancer presentation may differ ethnically.[13]When Should You Not Use the NCCN Risk Stratification?
It should not be applied to metastatic (M1) disease, where treatment is driven by metastatic burden and molecular biomarkers.[19] Furthermore, standard clinical stratification may be insufficient where molecular testing (e.g., Decipher, Prolaris) is indicated to clarify risk in borderline cases.[4]8. Integrating Risk Stratification into Multidisciplinary Care
The modern standard of care for prostate cancer involves multidisciplinary collaboration. Effective MDT meetings depend on shared data and common language—precisely what standardized risk stratification provides.
Beyond the MDT setting, risk stratification supports care coordination across the treatment continuum. A patient managed with active surveillance will undergo serial testing; pre-specified thresholds for intervention (e.g., upgrading from Grade Group 1 to Grade Group 2) trigger recalculation of NCCN risk group and reconsideration of treatment. Having a consistent, accessible tool throughout the patient journey reduces fragmentation and ensures guideline-concordant care.
Ready to Simplify Your Prostate Cancer Risk Stratification?
Calculate NCCN, CAPRA, and Epstein criteria simultaneously for more precise clinical decisions.
Calculate Risk Now
Free to use. No registration required.
References
- Duke University Scholars. NCCN Risk Categorization Validation. Source
- PubMed. NCCN Guidelines 2024 Updates. Source
- ScienceDirect. Favorable vs Unfavorable Intermediate Risk. Source
- ASCO Pubs. Molecular Testing in Intermediate Risk. Source
- UCSF Urology. CAPRA Score Overview. Source
- AUA Journals. CAPRA Validation Studies. Source
- AUA Journals. Longitudinal CAPRA Outcomes. Source
- European Urology. CAPRA Score and Mortality. Source
- PMC. Epstein Criteria and Active Surveillance. Source
- AUA Journals. MRI and Epstein Criteria. Source
- TAU Journal. Contemporary Active Surveillance. Source
- PMC. Impact of MDT on Prostate Cancer Survival. Source
- ASCO PO. Prostate Risk in Diverse Populations. Source
- Evidencio. Hierarchical Risk Modeling. Source
- ScienceDirect. Multicenter CAPRA Validation. Source
- Grand Rounds Urology. Life Expectancy and NCCN. Source
- Nature. Comparison of Risk Nomograms. Source
- Frontiers. Favorable vs Unfavorable Intermediate Risk Subtypes. Source
- Duke University. Metastatic Prostate Staging. Source
- PMC. Molecular and Clinical Staging in Prostate Cancer. Source
- Scribd. NCCN Guidelines Prostate Cancer V3.2024. Source
- ASCO Pubs. Long-term outcomes of NCCN Risk Groups. Source
- PMC. The Evolution of PSA Density in Risk Assessment. Source
- eCancer. CAPRA-S vs Minimal Residual Disease Subtypes. Source
- PMC. Validation of the Epstein Criteria in Radical Prostatectomy. Source
- MSKCC. Active Surveillance for Localized Prostate Cancer. Source
- Medical Independent. National Guidelines on Active Surveillance. Source
- BAUS. Patient Information: Active Surveillance Leaflet. Source
- Mayo Clinic. Active Surveillance for Prostate Cancer: What to Expect. Source
- OHA. HERC Evidence-based Review: NCCN Prostate 3.2024. Source
- PubMed. Gleason Score Trends in Localized Disease. Source
- Prostate Cancer UK. Active Surveillance Evidence-based Hub. Source
- The Focal Therapy Clinic. Understanding MDT Meetings for Prostate Care. Source
- King's College Hospital. The Role of the Multidisciplinary Team. Source
- PCF. Prostate Cancer Diagnosis and Risk Groups. Source
- National Cancer Institute. D'Amico Criteria Definition. Source
- BAUS. MDT Prostate Cancer Guidance. Source
- PMC. Risk Stratification in the Era of Genomic Testing. Source
- PMC. Clinical Utility of Prostate Cancer Nomograms. Source
- Uroweb. EMUC23: Why MDTs are Paramount to Optimal Care. Source
- CancerNetwork. Review of Favorable vs Unfavorable Intermediate Risk. Source