CAPRA Score Calculator for Prostate Cancer Risk Assessment
Estimate prostate cancer recurrence risk in seconds with the CAPRA Score calculator from OncoToolkit. Use age, PSA, Gleason, clinical stage, and biopsy cores to stratify risk, support treatment decisions, and standardize documentation.

1. Introduction to the CAPRA Score for Prostate Cancer
Localized prostate cancer remains one of the most common malignancies managed by prostate cancer specialists and urologic oncologists worldwide. Clinicians must synthesize PSA, Gleason pattern, clinical T stage, biopsy burden, and patient age while counselling men on radical prostatectomy, radiotherapy, or conservative strategies such as active surveillance. The Cancer of the Prostate Risk Assessment (CAPRA) Score was created at UCSF to condense these core variables into a concise, 0–10 pre‑treatment risk index that directly predicts the likelihood of biochemical recurrence.1, 2, 3, 4
At OncoToolkit, we’ve built a CAPRA Score calculator that operationalizes this risk model in a clean, mobile‑responsive interface. Clinicians can move from raw clinic data to a numeric score, risk group, and approximate 5‑year recurrence‑free survival in a few clicks—without paging through tables or performing mental arithmetic. By reducing cognitive load and standardizing risk language across teams, our platform helps MDTs spend less time calculating and more time discussing the nuances of treatment choice and patient preference.2, 3, 5, 1
2. Understanding the CAPRA Score System
The UCSF CAPRA Score is a validated, multivariable risk tool for men with clinically localized prostate cancer at the time of diagnosis. It assigns integer points to five routinely available clinical factors—age, PSA, biopsy Gleason pattern, clinical T category, and percentage of positive biopsy cores—and sums them to produce an overall score between 0 and 10. Higher scores reflect a greater risk of biochemical recurrence after primary treatment, as well as increased probabilities of metastasis, prostate cancer–specific mortality, and overall mortality in several cohorts.3, 4, 1, 2
The original development work used Cox proportional hazards modelling in the CaPSURE registry and then translated regression coefficients into a simplified point‑based system. Each 2‑point increase in CAPRA Score roughly doubles the risk of recurrence, providing an intuitive gradient that is easy to communicate to patients and colleagues. Five‑year recurrence‑free survival ranges from approximately 85–90% in the lowest‑risk patients (CAPRA 0–1) to below 10–30% in the highest‑risk groups (CAPRA 7–10) across different validation series.6, 2, 3
2.1 Core Components and Scoring Philosophy
CAPRA focuses deliberately on variables that are:4, 2, 3
- Routinely collected at diagnosis.
- Widely understood by urologists, radiation oncologists, and medical oncologists.
- Strongly associated with biochemical and long‑term outcomes.
The five inputs are:
- Age: Patients aged 50 or older receive 1 point, reflecting slightly higher risk independent of other factors.
- PSA: Rising PSA strata (e.g., 0–6, 6–10, >10 ng/mL in many implementations) contribute 0–3 points, capturing tumour burden and biology.
- Biopsy Gleason pattern: Presence and dominance of Gleason pattern 4 or 5 are major determinants of risk and can contribute up to 3 points.
- Clinical T stage: Higher T categories (beyond T1–T2) add points to the score, reflecting adverse local tumour extent.
- Percent positive cores: Increasing proportions of involved biopsy cores add 0–3 points, integrating volume of disease into the risk estimate.
On our platform, these elements are presented as intuitive categories that mirror the original scoring scheme, so clinicians do not need to remember precise point allocations at the point of care.5, 7
3. Clinical Utility: Why the CAPRA Score Matters in Practice
3.1 Linking Baseline Risk to Treatment Decisions
Management of localized prostate cancer increasingly emphasizes individualized risk stratification. CAPRA supports several key clinical tasks:1, 2, 3, 4
- Treatment selection: Men with low CAPRA Scores (0–2) are often suitable for active surveillance or single‑modality local therapy, whereas those with higher scores are more likely to benefit from aggressive approaches such as dose‑escalated radiotherapy, combined androgen‑deprivation therapy, or extended lymph node dissection.
- Counselling on recurrence risk: A single 0–10 scale allows urologists to discuss the probability of biochemical recurrence in concrete terms, using 5‑year recurrence‑free survival ranges as an anchor.
- Multidisciplinary planning: CAPRA provides a shared language across urology, radiation oncology, and medical oncology, helping MDTs quickly prioritize complex cases.
Compared with traditional three‑tier systems such as D’Amico, CAPRA offers finer gradations of risk and has shown comparable or superior discrimination in several studies. This additional granularity is particularly helpful for “borderline” cases, such as favourable intermediate‑risk disease, where small differences in PSA, Gleason, or biopsy burden may meaningfully influence management.8, 9, 2, 1
3.2 Reducing Complexity and Cognitive Load for Clinicians
In daily practice, clinicians often juggle multiple nomograms, guideline algorithms, and genomic tests. Manually calculating CAPRA requires referencing point tables, remembering how each variable contributes, and then mapping the total score to risk groups and survival estimates. Under time pressure, this workflow can be error‑prone and cognitively demanding.
OncoToolkit’s CAPRA calculator is intentionally designed to reduce this friction:5, 1
- Single‑screen data entry avoids multi‑page forms.
- Dropdowns and toggle buttons minimize typing and reduce mis‑keyed values.
- Automated mapping to risk groups simplifies interpretation.
4. Clinical Evidence and Validation of the CAPRA Model
4.1 Technical Breakdown: The Underlying Model and Formula
CAPRA was developed using Cox proportional hazards regression on a large, prospective registry of men with localized prostate cancer, most of whom underwent radical prostatectomy. Variables were chosen based on clinical plausibility and univariable performance, then retained in a multivariable model. The final coefficients were scaled and rounded to produce integer point contributions that approximate the log‑hazard for biochemical recurrence.2, 3, 4
4.2 Key Validation Studies and Outcomes
Multiple independent groups have evaluated CAPRA across diverse settings:11, 12, 6, 3, 2
4.3 CAPRA in Active Surveillance and Contemporary Care
In modern practice, many men with low‑ or selected intermediate‑risk disease are managed with active surveillance. CAPRA has been used to track “risk drift” over time and to investigate whether changes in the score predict upgrading or adverse pathology at delayed prostatectomy. Increasing CAPRA during surveillance correlates with higher odds of unfavourable pathology, suggesting that dynamic changes in the score may help identify men who should transition from surveillance to definitive therapy.10, 3
4.4 Comparison with Alternative Stratification Systems
CAPRA has been compared with several other frameworks:14, 9, 8, 1, 2
4.5 Limitations and Thoughtful Use
Despite its strengths, CAPRA has recognized limitations. For these reasons, OncoToolkit presents CAPRA as a transparent, evidence‑based decision support aid, not a stand‑alone directive. Clinicians should integrate CAPRA with pathology review, imaging, genomic tests, comorbidity indices, and patient preferences before finalizing management.6, 15, 14, 2
5. How the CAPRA Score calculator on OncoToolkit works
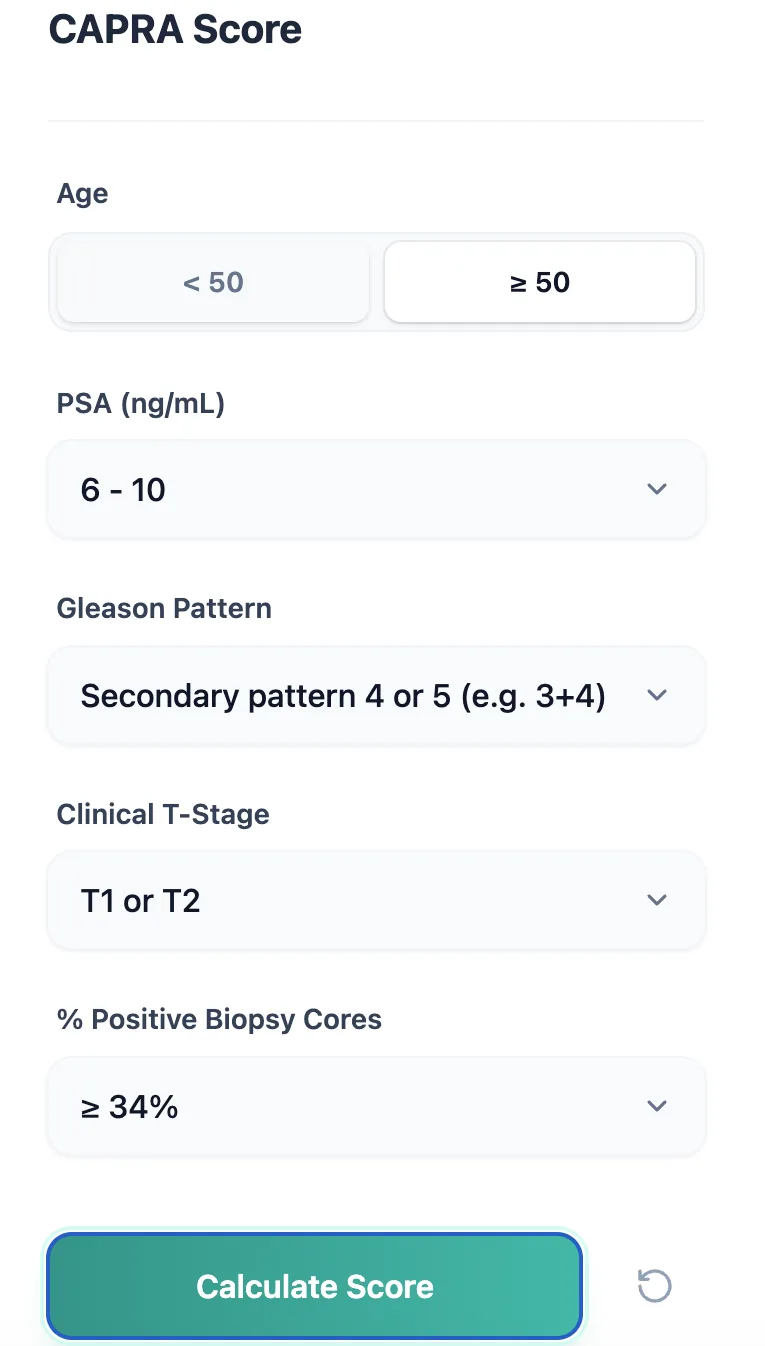
Figure 1. The CAPRA calculator input form captures age, PSA band, Gleason pattern, clinical T stage, and percentage of positive biopsy cores using simple toggles and dropdowns.
5.1 Input workflow
- Age: Select “< 50” or “≥ 50”, which automatically supplies the appropriate age‑related point contribution.
- PSA (ng/mL): Choose the PSA band that matches the diagnostic value—e.g., 0–6, 6–10, or higher categories.7, 5
- Gleason pattern: Use the dropdown to specify pattern burden.7, 5
- Clinical T stage: Choose T1/T2 for organ‑confined disease or higher categories when there is clinical extension.1, 7
- Percentage of positive cores: Select the bracket that matches the proportion of biopsy cores containing carcinoma.5, 7
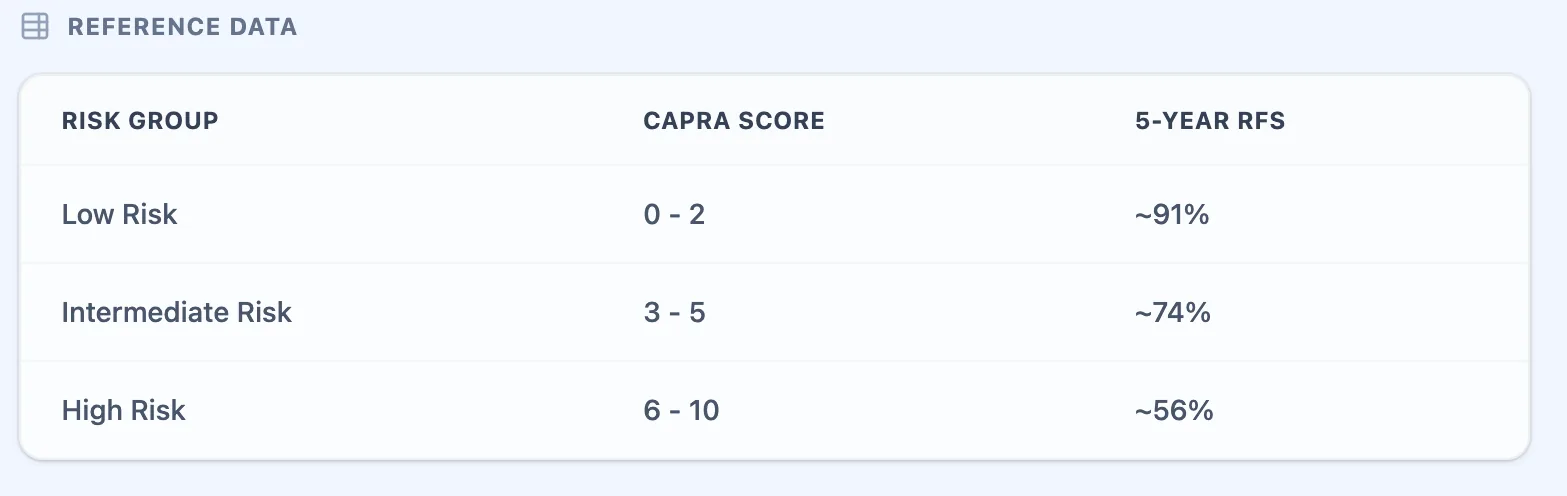
Figure 2. The embedded reference table maps total CAPRA Scores to low, intermediate, and high risk categories.

Figure 3. The clinical background panel summarizes the purpose of CAPRA and the point‑based formula.
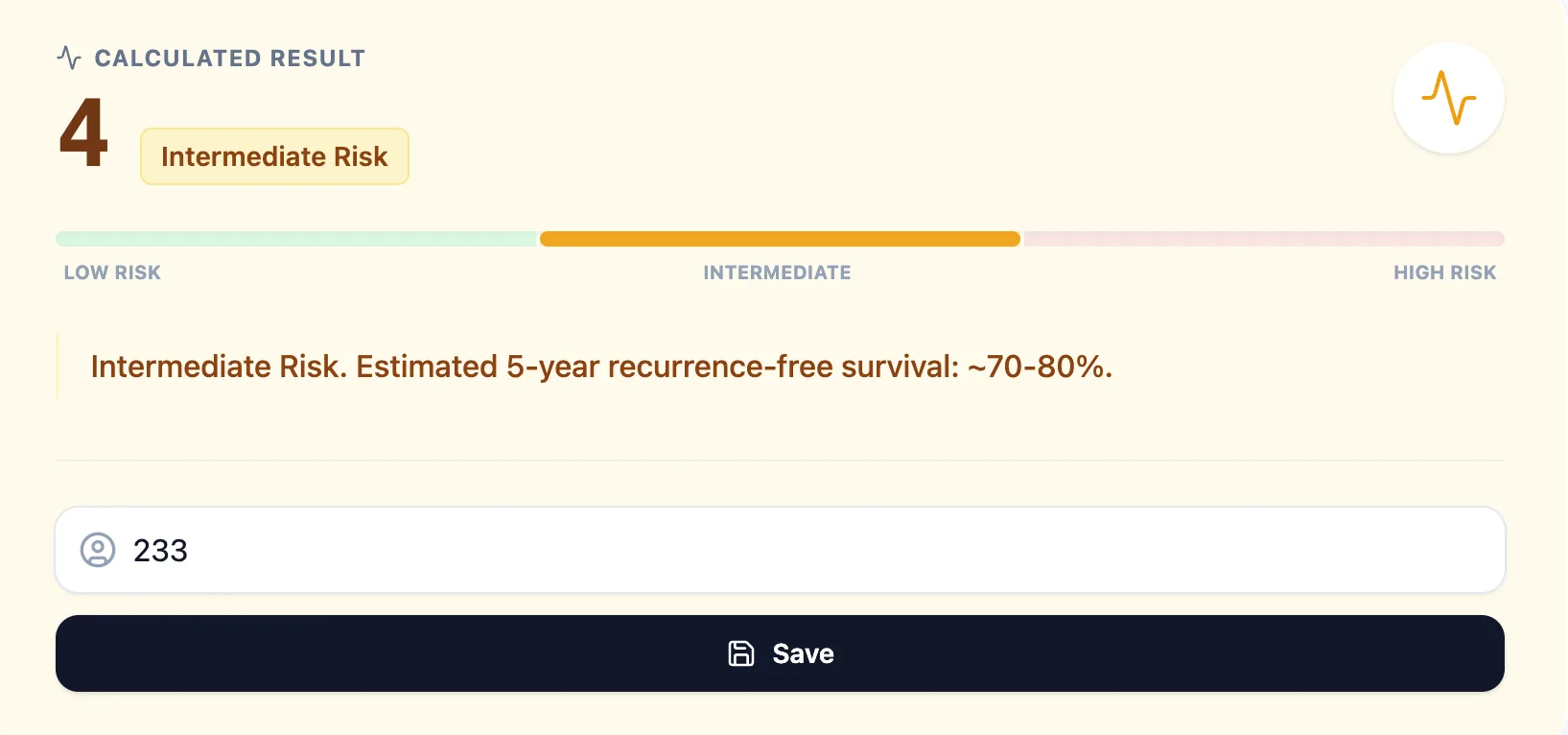
Figure 4. Example output showing an intermediate‑risk CAPRA Score with a visual risk bar.
5.2 Designed for day‑to‑day usability
Several design choices make the OncoToolkit CAPRA calculator practical in real workflows.
6. How the platform supports both clinical care and research
6.1 Routine clinical decision support
For prostate cancer specialists and urologic oncologists, the OncoToolkit CAPRA calculator supports multiple stages of the care pathway:3, 1, 2
6.2 Education and training
The CAPRA calculator is a powerful educational tool for case‑based teaching and understanding parameter sensitivity.1, 2, 3
6.3 Research, registries, and quality improvement
Because CAPRA is simple, validated, and widely cited, it is well suited as a stratification variable in clinical research and QI.15, 9, 12, 6, 3
7. Clinical FAQ
When should you not use the CAPRA Score? The CAPRA Score is intended for men with clinically localized, previously untreated prostate adenocarcinoma. It is not validated for: de novo metastatic disease; patients who have already undergone radical prostatectomy, radiotherapy, or focal therapies; or situations with incomplete core variables.4, 15, 2
How does the CAPRA Score compare with D’Amico and NCCN criteria?
D’Amico and NCCN systems remain central to guidelines. CAPRA complements them by offering more individualized risk estimates by incorporating biopsy core burden and age.9, 8, 2, 1
Can CAPRA be used for active surveillance decisions?
Increasing CAPRA during surveillance is associated with higher rates of adverse pathology. However, it should be used alongside MRI and genomic tests.13, 10, 3
Ready to Simplify Your Prostate Cancer Risk Assessment?
Calculate the CAPRA Score in seconds with our validated clinical tool.
Use CAPRA Calculator Now
FREE TO USE • NO REGISTRATION REQUIRED