Khorana Score (VTE Risk) Calculator: Evidence & Guidelines
An evidence-based guide and calculator for VTE risk stratification in ambulatory cancer patients using the Khorana Score criteria and ASCO/ITAC guidelines.

Quick Navigation
1. Introduction to Khorana Score (VTE Risk)
Venous thromboembolism (VTE) is a leading cause of morbidity and mortality in patients with solid tumors receiving systemic chemotherapy, rivaling infection and disease progression as causes of non‑cancer death. The challenge for oncologists is to distinguish which ambulatory patients are at sufficiently high risk to warrant pharmacologic prophylaxis, while avoiding unnecessary anticoagulation and bleeding in lower‑risk individuals.1, 2, 3
The Khorana Score (VTE Risk) provides a simple point‑based risk assessment model to estimate short‑term VTE risk in ambulatory solid‑tumor patients starting chemotherapy. At OncoToolkit, we have implemented a Khorana Score (VTE Risk) calculator that operationalizes this model in a user‑friendly interface, aligning with contemporary international guidelines and allowing clinicians to incorporate both classic and emerging score adaptations into everyday practice.4, 5, 6, 7
2. Understanding the Khorana Score (VTE Risk) Criteria
The Khorana Score was originally developed from a prospective cohort of 2,701 ambulatory cancer patients initiating chemotherapy and validated in a separate cohort of 1,365 patients. Using multivariable logistic regression, Khorana and colleagues identified tumor site, platelet count, leukocyte count, hemoglobin/erythropoiesis‑stimulating agent (ESA) use, and body mass engine (BMI) as independent predictors of early VTE, and converted their regression coefficients into an easy‑to‑apply point system.6, 7
Standard Khorana Scoring Parameters:
- 2 points: Very‑high‑risk cancer sites (stomach, pancreas).
- 1 point: High‑risk cancer sites (lung, lymphoma, gynecologic, bladder, testicular).
- 1 point: Pre‑chemotherapy platelet count ≥ 350 × 10⁹/L.
- 1 point: Hemoglobin < 10 g/dL or use of ESAs.
- 1 point: Pre‑chemotherapy leukocyte count > 11 × 10⁹/L.
- 1 point: BMI ≥ 35 kg/m².
Total scores are then grouped into:7, 6
- Low risk: 0 points.
- Intermediate risk: 1–2 points.
- High risk: ≥3 points.
Observed symptomatic VTE rates over approximately 2.5–3 months in the original cohorts were around 0.3–0.8% for low‑risk, 1.8–4.8% for intermediate‑risk, and 6.7–12.9% for high‑risk patients, supporting the model’s ability to discriminate risk levels.7, 6
Multiple subsequent cohort studies and meta‑analyses have confirmed that the Khorana Score stratifies early VTE risk in ambulatory cancer patients across diverse settings, though performance varies by tumor type and treatment era.2, 8, 9, 10
Figure 1. The clinical context panel on our platform summarizes the Khorana Score’s purpose, core variables, and primary source evidence, positioning the calculator within an evidence‑based framework.
3. Clinical Significance of Khorana Score in Practice
3.1 Direct Impact on Anticoagulation Decisions
International guidelines from ASCO, the International Initiative on Thrombosis and Cancer (ITAC), and other bodies recommend routine VTE risk assessment in ambulatory cancer patients and consideration of pharmacologic thromboprophylaxis for those at intermediate‑to‑high risk, often defined as Khorana score ≥2. For example:11, 12, 4, 1
- The 2020 ASCO guideline update on VTE prophylaxis and treatment in patients with cancer recommends that ambulatory patients receiving systemic chemotherapy who are at high risk (Khorana score ≥2) and have no significant bleeding risk may be offered prophylaxis with a direct oral anticoagulant (DOAC) such as apixaban or rivaroxaban, or low‑molecular‑weight heparin (LMWH).13, 14
- The 2022 ITAC international guidelines similarly support DOAC or LMWH prophylaxis for ambulatory patients at intermediate‑to‑high risk of VTE, identified either by very high‑risk tumor type (for example, pancreatic cancer) or by a validated risk model such as Khorana score ≥2, provided they do not have active bleeding or high bleeding risk.15, 4, 1
3.2 Specific Use Cases for Ambulatory Solid‑Tumor Patients
For oncologists focusing on solid tumors receiving intravenous or oral cytotoxic chemotherapy, common use cases of the Khorana Score (VTE Risk) calculator include:
- Baseline risk stratification before starting first‑line chemotherapy for pancreatic, gastric, lung, gynecologic, or bladder cancer, where Khorana scores ≥2 are frequent and prophylaxis decisions are highly relevant.8, 16, 2
- VTE prevention programs in infusion centers, where nursing or pharmacy teams screen new chemotherapy starts using the Khorana Score and flag high‑risk patients for clinician review of prophylaxis eligibility.5
- Shared decision‑making consultations, in which clinicians discuss the absolute VTE risk (for example, ~10% over six months for Khorana ≥2 in some cohorts) versus the risk of major bleeding and pill burden when considering DOAC prophylaxis.17, 16
- Trial eligibility and documentation, as many prophylaxis trials (for example, AVERT and CASSINI) require Khorana scores ≥2, making the calculator useful both in clinical research screening and in extrapolating trial results to individual patients.19, 18, 16, 17
- MDT case discussions, particularly for complex patients where other comorbidities, thrombocytopenia, or recent surgery complicate anticoagulation decisions; the Khorana Score provides a standardized baseline risk anchor for these conversations.5
3.3 Integrating Emerging Therapies and Regimens
Although the original cohorts were dominated by cytotoxic chemotherapy, modern practice increasingly involves immunotherapy and targeted agents. Observational data suggest that the Khorana Score continues to stratify VTE risk in some of these contexts, but calibration may differ by tumor type, biomarker profile, and combination regimen. At OncoToolkit, we emphasize that the Khorana Score should be interpreted as a starting point for risk–benefit assessment rather than a rigid rule, particularly when novel agents, recent surgery, or central venous catheters are part of the clinical picture.3, 2, 8
4. Clinical Evidence, Guidelines, and Prophylaxis Trials
4.1 Model Performance and Prognostic Impact
Systematic reviews and meta‑analyses have reported that patients with Khorana score ≥3 have 6‑month VTE incidences around 11%, compared with substantially lower rates in those with scores 0–1. Conversely, low‑risk patients (score 0) consistently demonstrate very low event rates, which supports forgoing prophylaxis in many such cases and focusing instead on education and symptom vigilance.2, 8, 5
High Khorana scores have also been associated with worse overall survival, reflecting the broader impact of thrombosis, aggressive disease biology, and comorbid factors captured by the score.10, 8, 2
4.2 Evidence from AVERT and CASSINI Trials
Two landmark randomized trials provide the foundation for guideline recommendations linking Khorana score to DOAC prophylaxis:
- AVERT (Apixaban for the Prevention of VTE in Cancer): Ambulatory solid‑tumor patients with Khorana score ≥2 starting chemotherapy were randomized to apixaban 2.5 mg twice daily versus placebo for up to 180 days. The trial demonstrated a significant reduction in VTE events (4.2% vs 10.2%) at the cost of higher major bleeding (3.5% vs 1.8%).16, 17
- CASSINI (Rivaroxaban Thromboprophylaxis in High‑Risk Ambulatory Cancer): Patients with solid tumors or lymphoma and Khorana score ≥2 were randomized to rivaroxaban 10 mg daily or placebo. Rivaroxaban significantly reduced VTE events during the on‑treatment period, again with increased—but acceptable—bleeding risk.18, 19
4.3 International Guideline Perspectives (ASCO, ITAC, NCCN)
- ASCO (2020/2023): Recommends DOACs (apixaban or rivaroxaban) or LMWH in ambulatory patients at high VTE risk (Khorana ≥2) starting systemic therapy and not at high bleeding risk.13, 14
- ITAC 2022: Provides grade 1A/1B recommendations for prophylaxis in ambulatory patients at intermediate‑to‑high risk identified by very high‑risk tumor types or Khorana score ≥2.4, 1, 15
- NCCN (2025): Highlights Khorana Score as a key tool in outpatient risk assessment, emphasizing alignment with individual bleeding risk and patient preferences.20, 21
5. How the OncoToolkit Khorana Score Calculator Works
5.1 User Workflow and Data Entry
Our Khorana Score (VTE Risk) calculator is tailored for point‑of‑care use via a 4-step workflow:
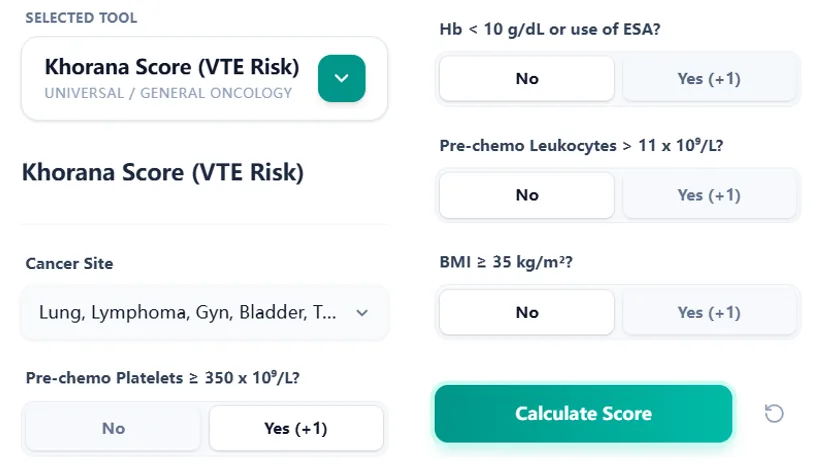
Figure 2. The structured input form maps directly to the Khorana variables, reducing recall burden and minimizing errors compared with manual pen‑and‑paper calculations.
5.2 Scoring Logic and Risk Interpretation
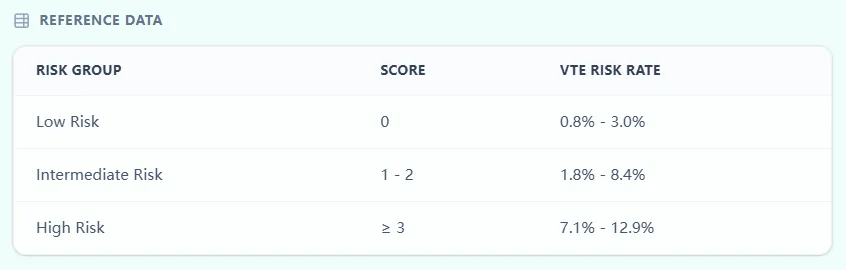
Figure 3. The embedded reference table links score ranges to risk categories and approximate event rates, supporting transparent risk communication.
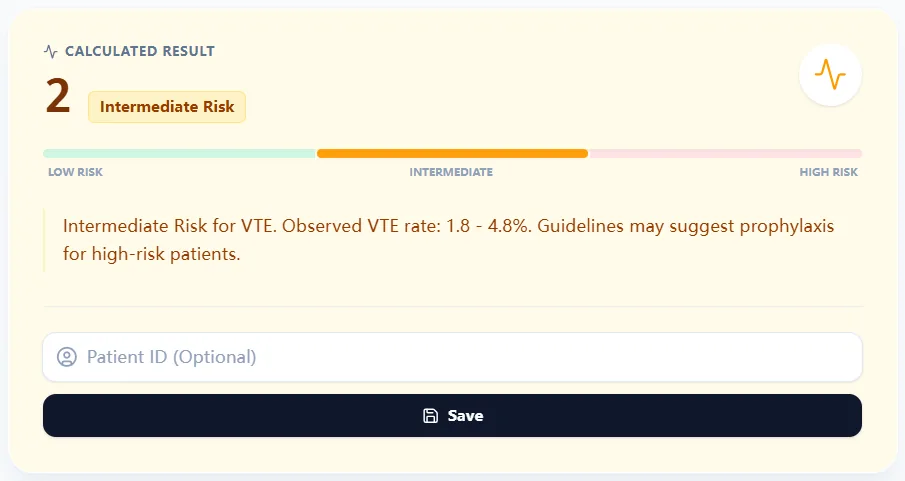
Figure 4. The result view summarizes the score, risk category, and estimated range, with space for patient identifiers for documentation workflows.
6. Advanced Use Cases and Global Applications
Consistent use of the Khorana Score via a standardized calculator facilitates prospective registry design, comparative risk‑model research, and linkage to institutional dashboards tracking VTE and bleeding outcomes to refine local prophylaxis protocols.10, 2
7. Emerging Modifications and Expansions
7.1 Tumor‑Specific Modified Khorana Scores
Recognizing heterogeneity, several groups have proposed modifications, such as adding D‑dimer levels for Stage IV lung cancer (AUC ~0.76) or histology-specific parameters for colorectal cancer.25, 23
7.2 Clinical‑Genetic and Multi‑Parameter Risk Scores
Recent research explores integrating single‑nucleotide polymorphisms (SNPs) and biomarkers. While showing superior discrimination in studies, these require specialized testing and are not yet routine recommendations.27, 28
8. Clinical FAQ on the Khorana Score (VTE Risk)
9. Conclusion: Implementing Guideline-Consistent VTE Assessment
Global guidelines now expect routine VTE risk assessment in ambulatory cancer patients, and the Khorana Score remains the most widely endorsed and practical tool for this purpose. OncoToolkit’s Khorana Score (VTE Risk) calculator translates this evidence into a rapid, intuitive workflow.4, 11
Ready to Simplify VTE Risk Stratification?
Calculate chemotherapy-associated VTE risk in seconds using the evidence-based Khorana Score.
Calculate Khorana Score Now
Free to use. No registration required.
References
- Falanga A, et al. Venous Thromboembolism in Cancer Patients. Source
- Mulder FI, et al. The Khorana score for prediction of venous thromboembolism in cancer patients. Source
- Khorana AA, et al. Role of biomarkers in cancer-associated thrombosis. Source
- International Initiative on Thrombosis and Cancer (ITAC) 2022 Guidelines. Source
- Haumschild J, et al. Implementation of a VTE Prevention Program in Ambulatory Oncology. Source
- Khorana AA, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Source
- Khorana AA. Risk assessment for venous thromboembolism in cancer patients. Source
- Ay C, et al. Prediction of venous thromboembolism in cancer patients. Source
- Bezan A, et al. The Khorana score and its parameters for prediction of venous thromboembolism. Source
- Pabinger I, et al. A clinical prediction model for cancer-associated venous thromboembolism. Source
- Farge D, et al. 2022 international clinical practice guidelines for the treatment and prophylaxis of VTE. Source
- Key NS, et al. Venous Thromboembolism Prophylaxis and Treatment in Patients With Cancer: ASCO Guideline Update. Source
- Key NS, et al. ASCO Guideline Update. Source
- ASCO 2023 VTE Update Slides. Source
- ITAC 2022 International Guidelines. Source
- Carrier M. Thromboprophylaxis in ambulatory cancer patients. Source
- Carrier M, et al. Apixaban for Thromboprophylaxis in Cancer. Source
- Khorana AA, et al. CASSINI Trial: Rivaroxaban for Thromboprophylaxis. Source
- Khorana AA, et al. Rivaroxaban for Thromboprophylaxis in High-Risk Ambulatory Patients. Source
- NCCN Guidelines: Cancer-Associated Venous Thromboembolic Disease. Source
- NCCN Guidelines 2025 version. Source
- Comparative performance of Khorana, Padua and IMPROVE models. Source
- Site-specific risk models in cancer. Source
- Risk assessment in colorectal cancer. Source
- Modified Khorana in Lung Cancer. Source
- D-dimer and Khorana Score. Source
- Clinical-genetic risk scores. Source
- Germline variants in VTE prediction. Source
- Validation of risk assessment models. Source
- Lancet Oncology VTE perspectives. Source
- Prevention strategies in oncology. Source
- Future of cancer-associated thrombosis management. Source