Oncotype DX Interpreter: Breast Recurrence Score Guide
Interpret Oncotype DX Breast Recurrence Score results for ER+, HER2- early breast cancer with age and nodal-adjusted chemotherapy guidance in seconds.

Quick Navigation
- 1. Introduction: The Role of Genomic Assays in Breast Cancer
- 2. What is the Oncotype DX Breast Recurrence Score?
- 3. Why the Oncotype DX Interpreter Matters in Clinical Practice
- 4. Clinical Evidence: TAILORx and RxPONDER Trials
- 4.1 The Math and Logic Behind the Tool
- 4.2 Key Validation Data and Typical Cohorts
- 4.3 Limitations and Clinical Caveats
- 5. How to Use the Oncotype DX Calculator
- 6. Reference Bands and Treatment-Oriented Categories
- 7. Supporting Clinical Care and Research Workflows
- 8. Frequently Asked Clinical Questions (FAQs)
- 9. References
1. Introduction: The Role of Genomic Assays in Breast Cancer
Adjuvant chemotherapy decisions in early-stage ER+, HER2- breast cancer are increasingly individualized, yet integrating genomic assays, nodal status, age, and evolving trial data at the point of care can be cognitively demanding. At OncoToolkit, we’ve built the Oncotype DX Interpreter calculator to translate the Breast Recurrence Score into clear, age- and node-adjusted guidance on chemotherapy benefit for your specific patient scenario, so you can focus on surgical planning and shared decision making rather than memorizing trial strata. [1], [2], [3]
This online tool is designed for breast surgical oncologists, breast surgeons, and multidisciplinary teams who routinely see ER+, HER2-, early breast cancer and need a fast, transparent interpretation of the Recurrence Score within contemporary TAILORx and RxPONDER evidence. The calculator is fully browser- and mobile-friendly, making it easy to use during pre-op clinics, tumor boards, or virtual MDTs when you need a quick, reproducible read on chemotherapy benefit. [4], [5], [1]
Figure 1. The clinical background panel summarizes how the Oncotype DX Breast Recurrence Score informs chemotherapy benefit in ER+, HER2- early breast cancer.
2. What is the Oncotype DX Breast Recurrence Score?
The Oncotype DX Breast Recurrence Score is a 21-gene expression assay that generates a numerical score from 0–100 to estimate distant recurrence risk and predict adjuvant chemotherapy benefit in hormone receptor–positive, HER2-negative, early breast cancer. Large prospective trials have shown that low to intermediate scores often allow safe omission of chemotherapy, whereas high scores identify patients with substantial benefit from chemo-endocrine therapy. [6], [7], [1]
TAILORx refined Recurrence Score cut-points in node-negative disease and demonstrated that many women—particularly those over 50 with scores up to 25—derive little or no benefit from chemotherapy. RxPONDER extended this paradigm to patients with one to three positive nodes, revealing node-positive subgroups, especially postmenopausal women with scores 0–25, who can often avoid chemotherapy without compromising invasive disease-free survival. The Oncotype DX Interpreter on our platform packages these nuanced trial insights into a practical, patient-level interpretation engine. [8], [5], [3], [1]
3. Why the Oncotype DX Interpreter Matters in Clinical Practice
For breast surgeons and oncologists, adjuvant chemotherapy recommendations no longer hinge on tumor size and nodal status alone; they depend on gene expression results, menopausal status, and subtle interaction effects between Recurrence Score and age. Without digital support, applying trial-based rules requires juggling multiple tables and remembering exceptions (for example, premenopausal women with intermediate scores versus postmenopausal counterparts), which increases cognitive load and the risk of misclassification. [9], [5], [1]
On our platform, the Oncotype DX Interpreter consolidates these decision rules into a single, intuitive interface that uses three clinically familiar inputs—Recurrence Score (0–100), nodal status (N0 vs N1 with 1–3 nodes), and age/menopausal category—to produce a concise, evidence-aligned statement about chemotherapy benefit. The calculator is optimized for rapid use on desktop or mobile, so you can check a case while reviewing pathology, documenting an operative note, or discussing options with a patient and medical oncology colleagues. [2], [10], [4], [6]
4. Clinical Evidence: TAILORx and RxPONDER Trials
4.1 The Math and Logic Behind the Tool
The calculator does not recompute the 21-gene Recurrence Score; instead, it acts as an interpretation layer that maps a known score to clinically meaningful categories of chemotherapy benefit. Under the hood, the algorithm performs a structured lookup based on three variables: Recurrence Score band, nodal status (N0 vs N1, 1–3 nodes), and age/menopausal group (≤50 premenopausal vs >50 postmenopausal). These lookups are explicitly anchored to cutoffs used in TAILORx and RxPONDER, such as score ranges 0–15, 16–25, and ≥26, and whether chemotherapy was non-inferior or beneficial compared with endocrine therapy alone for each subgroup. [5], [6], [1], [9]
For example, node-negative women over 50 with scores 0–25 had similar invasive disease-free survival with endocrine therapy alone versus chemo-endocrine therapy in TAILORx, so the tool returns a “No chemotherapy benefit” conclusion for that cell. In contrast, premenopausal women with node-positive disease (1–3 nodes) and scores 0–25 showed improved outcomes with chemotherapy in RxPONDER, leading to a “Chemotherapy benefit observed” interpretation. By encoding these patterns, the calculator makes the underlying trial logic transparent and repeatable across users. [3], [8], [1], [9]
4.2 Key Validation Data and Typical Cohorts
TAILORx enrolled women with HR+, HER2-negative, node-negative early breast cancer and showed that approximately 70% could safely avoid chemotherapy, including all with scores 0–15 and most over age 50 with scores up to 25. RxPONDER included women with HR+, HER2-negative breast cancer and one to three positive nodes, randomizing those with scores 0–25 to endocrine therapy alone versus chemo-endocrine therapy and demonstrating a divergence in benefit by menopausal status. Together, these trials provide high-level evidence that Recurrence Score, age, and nodal status jointly predict chemotherapy benefit rather than recurrence risk alone. [8], [4], [1], [9], [5], [3]
Additional real-world and guideline reviews have confirmed that using the Oncotype DX Recurrence Score to guide adjuvant therapy reduces chemotherapy utilization without compromising outcomes, especially for node-negative and postmenopausal node-positive patients. International consensus statements now endorse gene-expression profiling, including Oncotype DX, as a tool to select or omit chemotherapy in appropriate HR+, HER2- early breast cancer populations, acknowledging remaining uncertainties in certain premenopausal and higher-risk subgroups. [11], [7], [4], [6]
4.3 Limitations and Clinical Caveats
The Oncotype DX Interpreter is intended only for ER+, HER2-negative, early-stage breast cancer patients who meet eligibility criteria similar to those in TAILORx and RxPONDER; it should not be extrapolated to HER2-positive, triple-negative, or locally advanced disease outside the assay’s validated scope. Premenopausal women with intermediate scores present particular complexity because observed chemotherapy benefit may be partly mediated by ovarian suppression, and shared decision making about chemotherapy versus optimized endocrine therapy remains essential. [7], [6], [9], [5]
5. How to Use the Oncotype DX Calculator
The Oncotype DX Interpreter focuses on three required inputs: the numeric Recurrence Score (0–100), nodal status, and age/menopausal category. You begin by entering the Recurrence Score reported by the laboratory, either by typing the value directly or using the plus/minus controls to adjust the number. Next, you choose nodal status as “Node Negative (N0)” or “Node Positive (N1, 1–3 nodes)” and then select “Age ≤ 50 (Premenopausal)” or “Age > 50 (Postmenopausal)” to match your patient’s clinical profile. [6], [9]
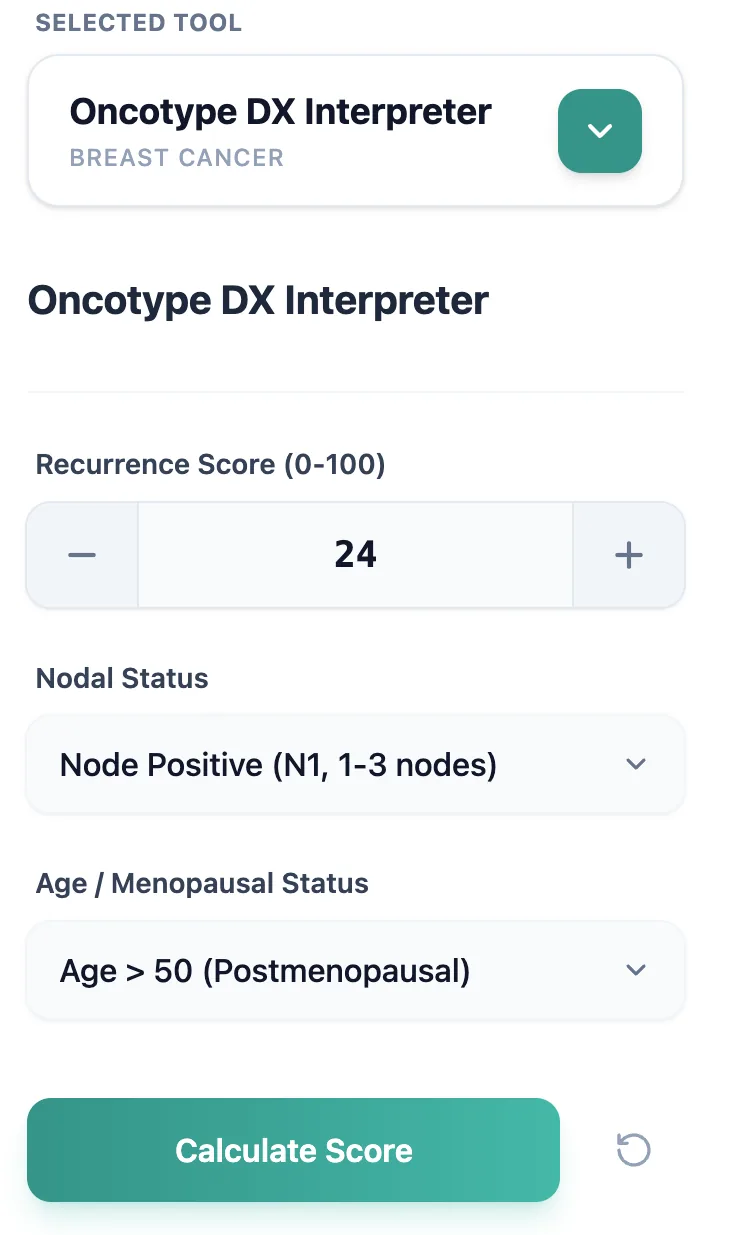
Figure 2. The streamlined input form accepts only Recurrence Score, nodal status, and age/menopausal group.
Once you click “Calculate Score,” the tool instantly generates a color-coded interpretation panel with the Recurrence Score, a genomic risk band (low, intermediate, or high), and a concise, trial-aligned statement about chemotherapy benefit. [4], [1], [2], [5], [8], [6]

Figure 3. The result screen displays the entered score, risk band, and chemotherapy benefit interpretation.
6. Reference Bands and Treatment-Oriented Categories
The calculator’s reference table summarizes how combinations of Recurrence Score ranges, nodal status, and age translate into chemotherapy benefit categories. For instance, Recurrence Scores 0–25 in node-negative women over 50 are associated with no meaningful chemotherapy benefit, while scores 26–100 are grouped under “Clear benefit” regardless of nodal status. [1], [2], [6]

Figure 4. The embedded reference table outlines Recurrence Score intervals intersecting with nodal and age categories.
7. Supporting Clinical Care and Research Workflows
In daily practice, the Oncotype DX Interpreter can be used at several key decision points: confirming adjuvant plans after definitive breast surgery, revisiting systemic therapy choices at MDT when new pathology or genomic results are available, or counseling patients who are hesitant about chemotherapy. [6], [1], [4]
For trainees, the calculator doubles as a teaching aid that reinforces how Recurrence Score, age, and nodal status interact, turning abstract trial findings into concrete patient scenarios they can experiment with. OncoToolkit’s broader platform allows these point-of-care calculations to be saved and aggregated into de-identified datasets for quality improvement. [9], [2], [5], [4]
8. Frequently Asked Clinical Questions (FAQs)
When should you not use the Oncotype DX Interpreter?
How does the Oncotype DX Interpreter compare with traditional criteria?
Ready to Simplify Your Oncotype DX Interpretations?
Get instant, trial-aligned chemotherapy benefit guidance tailored to your patient's clinical profile.
Try the Oncotype DX Interpreter Now
Free to use. No registration required.
References
- NAM Perspectives. Breast Cancer Management in the TAILORx Era. Source
- EMJ Oncology. Prediction with Precision: TAILORx and Personalised Treatment. Source
- ECOG-ACRIN. TAILORx Trial Findings on Chemotherapy Omission. Source
- PMC. Real-world utilization of Oncotype DX. Source
- The Oncologist. Integrating genomic assays in clinical practice. Source
- Nye Metoder. Molecular Gene Profile Analysis Report. Source
- ScienceDirect. Evolving paradigm in breast cancer genomics. Source
- Oncotype DX. RxPONDER Trial Results Summary. Source
- ASCO Daily News. Gene Signatures in Premenopausal Women. Source
- Oncotype DX UK. Which patients to test. Source
- GMKA. Consensus Statement on Oncotype DX Use. Source
- Annals of Oncology. Genomic assays vs clinical-pathologic criteria. Source